1. Alkaloidal analysis of the opium from Papaver setigerum DC.
2. A natural hybrid of Papaver setigerum DC. and P. somniferum L.
3. Examination of poppies of various species for the presence of morphine
4. Examination of poppy varieties of Papaver somniferum L. for opium of the highest morphine yield
5.The porphyroxine content of Japanese opium and of opium from successive lancings
The morphine content of Japanese opium
Author: Haruyo Asahina,, Toyohiko Kawatani,, Miss Masako Ono,, Sanaenosuke Fujita,
Pages: 20 to 33
Creation Date: 1957/01/01
Introduction
Since the promulgation of the Japanese Opium Act in 1954, which allows the cultivation of the opium poppy under licence, we have grown the opium poppy and other poppies for study and experiments, and the opiums or juices therefrom have been analysed.
Papaver setigerum DC. and its hybrid with P. somniferum L. have been grown. The main alkaloids in opiums from these different poppies were estimated and the percentages compared.
On the opium poppy varieties and other poppies, we tested for the presence or absence of morphine by our method of paper chromatography. The chromosome numbers of various species were counted. Further, for varieties or strains of P. somniferum, the morphine content was determined for the purpose of the plant breeding, chiefly by our method of paper chromatography and spectrophotometry.
The content of "porphyroxine-meconidine ", a red-turning alkaloid when heated in diluted mineral acid, of Japanese opium and of opium from successive lancings of P. somniferum was measured spectrophoto-metrically.
The morphine content of commercial opiums produced by the Japanese cultivators in the crop seasons of 1953/54 and 1954/55 was assayed by us by the method of the Japanese Pharmacopoeia, and this information is summarized in the paper.
Our paper is divided into the following sections:
Alkaloidal analysis of the opium from Papaver setigerum DC.
A natural hybrid of Papaver setigerum DC. and P. Somniferum L.
Examination of poppies of various species for the presence of morphine.
Examination of poppy varieties of Papaver somniferum L. for opium of the highest morphine yield.
The porphyroxine content of Japanese opium and of opium from successive lancings.
The morphine content of Japanese opium.
Acknowledgements
It is a pleasure to acknowledge our indebtedness to Dr. Charles G. Farmilo, Organic Chemistry and Narcotic Section, Food and Drug Directorate, Canada, for the seed of P. setigerurn DC. and to the chemists of the laboratory of the Division of Narcotic Drugs, United Nations, for their invaluable and continuous help and guidance.
We wish to express our thanks to S. Mizumachi and Y. Shiuchi, National Hygienic Laboratory, for their assistance in making many of the analyses during this investigation. Thanks are also due to T. Ohno, Kasukabe Experiment Station, for his aid in cytogenetical studies.
The presence of morphine in Papaver setigerum DC., previously suspected, was confirmed by Farmilo et al.[1] in 1953. Morphine was detected in the pod, bud, and leafy parts of this poppy plant.
The Japanese Opium Act (Act No. 71) promulgated in 1954, prohibits the cultivation of the opium poppy, including P. setigerum, except under licence.
For actual production of opium only P. somniferum has been cultivated, and there is no history of the cultivation of P. setigerum even experimentally, in Japan. No estimation of the alkaloids in opium from this poppy plant has been reported prior to the writing of the present paper. The capsules of P. setigerum are small and dehiscent, and this species can scarcely have any commercial use. Besides our desire to clarify the legal point of its inclusion in the Opium Act, it was studied for scientific reasons and as a possible parent in hybridization.
This investigation was undertaken to grow and identify botanically P. setigerum, and to determine quantitatively the content of alkaloids in the opium from this poppy plant.
With this in mind, the following two experiments, experiment I and experiment II, were made in successive years. The seeds were sown in October 1954 and 1955. The flowering continued almost throughout May of the following year.
There were plants with violet flowers and also plants with mauve flowers in the strain. In experiment I both kinds were used without separating them, whereas in experiment II only plants with violet flowers were used.
Three or four incisiom, each covering a quarter of the capsule, were made at intervals of one day, till no more latex was yielded. The first incision began ten days after the petals had fallen. In experiment I it began at different dates in the sequence of flowering, irrespective of the strains, whereas in experiment II all sets of incisions made began on the same date. Each incision consisted of four longitudinal slashes.
The capsules were incised in the evening and the latex was gathered just after incision (" Kiritori" collection) and the following morning (" Oigaki" collection). The opiums thus obtained were mixed, as is the usual practice in collecting commercial opium from P.somniferum.
|
Experiment |
Seeds sown |
Strains used |
Capsules incised |
Opium per capsule (air.dried) (Mg) |
Period o/ incision |
Plant height (cm) |
|
I |
20.X.1954 |
Violet and mauve-flowered |
90 |
15.56 |
16-31.V.1955 |
65 |
|
II |
25.X.1955 |
Violet-flowered |
130 |
15.88 |
18-24.V.1956 |
67 |
The presence of alkaloids, at the proper Rf values for morphine, codeine, and other opium alkaloids in the opium from P. setigerum was readily demonstrated by using paper chromatography [2]
Opium was rubbed thoroughly with dilute hydrochloric acid, and the solution containing the alkaloidal salts was centrifuged. The supernatant solution was used for the paper chromatography.
The paper chromatography was carried out as follows:
Solvent: n-butanol 50, 28 % NH4OH 9, distilled water 15 parts by volume. In the preparation of the solvent, the constituents were mixed, the mixture allowed to stand for some time, and the top layer of the mixture used for chromatography.
Paper: No. 51 manufactured by Toyo Filter Paper Co., Ltd., 2.5 x 40 cm.
Movement of solvent: Ascending.
Spot-developing reagent: Potassium iodoplatinate prepared according to Munier: 1 cc of 10% platinic chloride and 25 cc of 4% potassium iodide were mixed and the resulting solution made up to 50 cc with H 20.
The following spots were obtained. The Rf values and colouration of the spot are given.
|
Opium |
R/ values | ||
|---|---|---|---|
|
Experiment I |
0.73 |
0.87' |
0.92 |
|
Experiment II |
0.72 | 0.88 | 0.93 |
|
(Opium tincture) |
0.71 | 0.87 |
0.93 for comparison; P. somniferum) |
|
Colouration |
Dark blue |
Violet |
Violet |
The characteristic dark blue colouration of the morphine spot on a filter paper, when treated with potassium iodoplatinate solution, and occurring at the proper Rf value for morphine, near 0.71 with this solvent, was easily recognized.
The other alkaloidal spots were coloured violet. The Rf value of the codeine spot was 0.87 and the spot of Rf 0.93 corresponded to other by-alkaloids. Thebaine, papaverine, and narcotine were inseparable from each other by this solvent.
Opium tincture was used for comparison to fix the position of the morphine spot more precisely under the particular conditions.
The morphine content of the opium from P. setigerum was then determined by paper chromatography and spectrophotometry. [3] [4]
The contents of codeine, thebaine, papaverine, and narcotine were also assayed by our unified method.[5]
The results, as determined on the air-dried opium, are given below:
|
Opium |
Morphine |
Codeine |
Thebaine |
Papaverine |
Narcotine |
|
Experiment I |
5.1 | 0.9 | 2.1 | 1.9 | 0.1 |
|
Experiment II |
7.3 | 0.8 | 1.6 | 2.6 | 0.1 |
The values were quite similar for the two crop seasons. Perhaps this is due to a dose control of growing and harvesting methods and use of a particular strain of the species, but it is naturally not known whether this species may vary much in the alkaloids present in the opium or not. We hope later to examine one or more other strains of P. setigerum.
For comparison, the alkaloidal analyses of a few opium samples from the United Nations Opium Distribution Centre assayed by our unified method are given below. These relate, of course, to opium from P. somniferum.
|
Origin of opium |
Morphine |
Codeine |
Thebaine |
Papaverine |
Narcotine |
|
Turkey export U.N.15 |
13.5 | 1.7 | 0.8 | 0.8 | 4.9 |
|
India excise U.N.36 |
11.6 | 4.2 | 1.8 | 0.4 | 6.3 |
|
Iran Fars U.N.47 |
12.8 | 4.0 | 3.5 | 1.5 | 7.1 |
The results show that opium from P. setigerum is comparatively low in morphine and codeine, but high in thebaine and papaverine, and very low in narcotine. While morphine is still the principal alkaloid, narcotine is not the second one, but either thebaine or papaverine, and in general the picture of alkaloidal composition is almost opposite to that of somniferum opium.
The chromosome number was confirmed to be n = 22 in the haploid; 2n = 44 in the diploid (fig. 1 and 2).
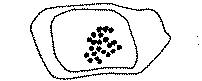
FIG.1. The Chromosomes in the Pollen Mother Cell of Papaver setigerum DC., showing n = 22 chromosomes, x 1,500

FIG. 2. The Chromosomes in the Root Tip Cell of Papaver setigerum DC., showing 2n = 44 chromosomes (x 2,500)
Summary:
Morphine, codeine, and other opium by-alkaloids were detected in the opium from Papaver setigerum DC.
The content of these alkaloids was determined quantitatively.
The chromosome number was confirmed to be n = 22 in the haploid, 2n = 44 in the diploid.
Vesselovskaya mentioned in 1933 that P. setigerum, although distinct, could be crossed readily with P. somniferum, indicating their close genetic relationship, and that the crosses were perfectly normal fertile hybrids with viable seed.[6] However, we do not know of either a chemical or a detailed botanical investigation of such a hybrid before our own.
It was observed in 1955 that the violet- and mauve-flowered strains of P. setigerum were almost pure phenotypically, that is, the violet- or mauve-flowered strain consisted almost exclusively of plants with violet or mauve flowers respectively.
An F 1 hybrid was found to grow in the violet-flowered strain in 1956. The chromosome number in the root-tip cell was 2n= 33, which was the stun of the gametic chromosome numbers of P. setigerum n = 22, and P. somniferum n = 11 (fig. 3). At the first meiotic metaphase in the pollen mother cell (PMC), 11 bivalent and 11 univalent chromosomes were observed (fig. 4). The chromosome behaviour in meiosis of PMCs of the F 1 hybrid was of the type of Pilosella.
Apparently the female parent is the violet-flowered strain of P. setigerum, whereas the male parent is considered to be P. somniferum, "Ikkanshu" variety, a strain commonly cultivated for actual production of opium in Japan. In 1955, no species with a chromosome number 2n = 22 or its multiple other than P. somniferum and P. setigerum was grown by us, and "Ikkanshu" was cultivated in the adjoining field to the female parent at our experiment station.
The hybrid was 125 cm in height, intermediate between the parents: P. setigerum 67 cm, P. somniferum 135 cm.
External characteristics as well as habits of the hybrid were observed in detail. It was matroclinous. The leaves, stems, flowers and capsules were like those of the female parent but were larger in size.
Seven capsules of the hybrid were incised in the usual way, as described in section 1, and 0.24 g of opium was obtained.

FIG. 3. Equatorial Plate in a Root Tip Cell of the F1 Hybrid, showing 33 somatic chromosomes (ca. x 3,700)
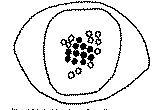
TABLE I
|
|
Female parent (P. setigerum a |
Fl hybrid |
Male parent (P. somni/erum b) |
|
Flower, colour |
violet |
violet |
white |
|
Pollen grain, length and width µ |
42.9 x 26.8 |
34.3 x 19.6 |
41.8 x 21.91 |
|
length: width |
1.6:1 |
1.75: 1 |
1.9:1 |
|
Stoma, length and width (µ) |
69.0 x 45.2 |
60.7 x 42.6 |
46.5 x 33.5 |
|
Capsule, character |
dehiscent |
dehiscent |
indehiscent |
|
length and diameter (mm) |
25 x 13 |
35 x 19 |
82 x 44 |
|
Disc, diameter (ram) |
12 | 18 | 27 |
|
Ray, range of number |
7-10 |
9-11 |
10 - 14 |
|
average number |
8.4 | 9.9 | 11.7 |
a Violet-Flowered strain.
b "Ikkanshu" strain.
The analytical values for alkaloides assayed by our unified method[7] on the opiums from the hybrid and its parents are listed as follows. The parents were grown again in the same year as the hybrid(1956).
|
Opium |
Morphine |
Codeine |
Thebaine |
Papaverine |
Narcotine |
|
P. setigerum (?) |
7.3 | 0.8 | 1.6 | 2.6 | 0.1 |
|
F 1 Hybrid |
13.2 | 3.6 | 1.5 | 2.6 | 0.2 |
|
P. somniferum (?) |
|
|
|
|
|
|
"Ikkanshu" |
16.0 | 3.7 | 1.0 | 0.9 | 1.1 |
By comparison with the result for opium from the female parent, P.setigerum, the opium of the hybrid showed a fairly considerable increase in morphine content (almost doubled) and a large increase in codeine content (four times as great). Thebaine and papaverine, already high, were not changed.
FIG. 4. First Meiotic Metaphase of a Pollen Mother Cell of the F 1 Hybrid, showing 11 bivalent (in black) and 11 univalent (in black) chromosomes(ca. x 2,860)
The narcotine content increased from 01% to 0.2% by our determination, but thus remained remarkably low.
As compared with the opium from the male parent. P. somniferum, the opium of the hybrid was almost equal in morphine and codenine, very low in narcotine, but quite high in thebaine and papaverine.
It is very interesting to observe that in the production of some alkaloids the hybrid became almost equal to the female parent, but in others it remained like like the female parent, whether the production was high, as with thebaine and papaverine, or low, as with narcotine.
Summary :
A natural hybrid of P. setigerum DC (?) and P. somniferum L. (?) was found. Connecting the external characteristics, it was matroclinuos.
The chromosome number was 2n=33. At the first meiotic metaphase in PMCs ,11 bivalent and 11 univalent chromosomes were observed.
The chromosome behaviour in meiosis of PMCs was of the type of Plosella.
Morphine was detected in the opium from the hybrid. The content was 13.2%.
The opium of the hybrid was high in thebaine and papaverine, but very low in narcotine. In morphine and codeine production, the hybrid was superior to the female parent and was almost equal to the male parent; but in production of other alkaloids, thebaine, papaverine, and narcotine, it corresponded to the female parent.
During 1955 and 1956 a considerable number of varieties and strains of Papaver somniferum, P. orientale, and P. rhoeas, together with 29 other species grown at the Kasukabe Experiment Station from seeds sent by foreign botanical gardens, were examined for the presence of morphine by our method of paper chromatography. The method has been given in a preceding section of this paper and was previously published in a United Nations document.[8]
The results are set out in Table II. In nearly all cases the latex was used for the test. In no poppy but P. somniferum and P. setigerum was any clear evidence of morphine found. In five other cases (Nos. 36 to 39, and No. 45) there was a spot at about the right Rf value, but the colouration was different from that of a morphine spot.
Examination was made by the ordinary paraffin method to determine or confirm the chromosome numbers of 31 of these species of Papaver. The results are set out in Table III -This table also shows in which cases previous determinations are known to us. It will be noted that only P.somniferum and P. setigerum were found to have chromosome numbers which are multiples of 11.
|
L: Latex |
B: Blue |
|
C: Capsule |
DB: Dark blue |
|
S: Straw |
BG :Blue green |
|
|
V: Violet |
|
|
DV: Dark violet |
|
|
Italics: Morphine spot |
|
No. |
Poppy |
Rf relative to 100 and Colouration |
Origin | ||||||
|---|---|---|---|---|---|---|---|---|---|
|
|
Papaver |
|
|
|
|
|
|
|
|
|
1 |
somniferum . |
L |
|
|
|
71DB |
88V | 94V |
Japan, perhaps originally |
|
|
|
|
|
|
|
69DB |
86V |
|
from North China |
|
|
|
|
|
|
|
69DB |
87V | 92V |
|
|
|
|
|
|
|
|
71DB |
86V | 91V |
|
|
2 |
Somniferum |
L |
|
|
|
72DB |
|
95V |
Saitama, Japan |
|
|
|
|
|
|
|
73DB |
|
95V |
|
|
|
|
|
|
|
|
70DB |
|
92V |
|
|
3 |
Rhoeas |
L |
none |
|
|
|
|
|
Tokyo, Japan |
|
4 |
Rhoeas |
L |
|
|
|
|
|
91V |
Tokyo, Japan |
|
|
|
|
|
|
|
|
|
93V |
|
|
5 |
orientale "Mahoney" |
L |
15DB |
|
|
|
|
96V |
Tokyo, Japan |
|
6 |
orientale bracteatum |
L |
18DB |
|
36V |
|
|
94V |
Tokyo, Japan |
|
7 |
orientale "Queen Alexandra" |
L |
18DB |
|
|
|
|
95V |
Tokyo, Japan |
|
8 |
orientale "Feltham" |
L |
17BG |
|
25V |
|
|
94V |
Tokyo, Japan |
|
9 |
orientale "Beauty of Livermere" |
L |
18BG |
|
25V |
|
|
96V |
Tokyo, Japan |
|
10 |
orientale "Salmon Queen" |
L |
|
|
|
|
|
95V |
Tokyo, Japan |
|
11 |
orientale "Apricot Queen" . |
L |
18DB |
|
26V |
|
|
92V |
Tokyo, Japan |
|
12 |
orientale "Princess Victoria Louise" |
L |
19BG |
|
27V |
|
|
96V |
Tokyo, Japan |
|
|
|
|
16BG |
|
23V |
|
|
96V |
|
|
|
|
|
17BG |
|
24V |
|
|
96V |
|
|
|
|
|
17BG |
|
25V |
|
|
92V |
|
|
13 |
orientale "Rembrandt" |
L |
19BG | 20DB | 28V |
|
|
91V |
Tokyo, Japan |
|
14 |
orientale "Pink Beauty" . |
L |
18BG |
|
27V |
|
|
91V |
Tokyo, Japan |
|
15 |
orientale "Mrs. Perry" |
L |
19DB |
|
29V |
|
|
93V |
Tokyo, Japan |
|
16 |
orientale "Columbia (Olympia fl. pl.)" |
L |
19DB |
|
|
|
|
94V |
Tokyo, Japan |
|
17 |
orientale "Perry's White" |
L |
18BG | 26DB | 28V |
|
|
93V |
Tokyo, Japan |
|
|
|
|
18BG |
|
26V |
|
|
89V |
|
|
18 |
somniferum |
L |
|
|
|
77DB |
|
94V |
Tokyo, Japan |
|
19 |
somniferum . |
L |
|
|
|
78DB |
|
96V |
Tokyo, Japan |
|
20 |
somniferum |
L |
|
|
|
72DB |
|
94V |
Tokyo, Japan |
|
|
|
|
|
|
|
70DB |
|
92V |
|
|
21 |
somniferum |
L |
|
|
|
71DB |
88V | 93V |
Tokyo, Japan |
|
22 |
setigerum |
L |
|
|
|
74DB |
89V | 93V |
Saitama, Japan |
|
|
|
|
|
|
|
73DB |
87V | 92V |
|
|
|
|
|
|
|
|
67DB |
88V | 94V |
|
|
23 |
somniferum |
L |
|
|
|
72DB |
|
94V |
Tokyo, Japan |
|
24 |
somniferum |
L |
|
|
|
76DB |
|
95V |
Tokyo, Japan |
|
25 |
somniferum |
L |
|
|
|
75DB |
91V | 96V |
Tokyo, Japan |
|
26 |
somniferum |
L |
|
|
|
77DB |
|
96V |
Tokyo, Japan |
|
27 |
somniferum . |
L |
|
|
|
75DB |
91V | 95V |
Tokyo, Japan |
|
28 |
somniferum . |
L |
|
|
|
70DB |
|
92V |
Tokyo, Japan |
|
29 |
somniferum . |
S |
|
|
|
73DB |
|
|
Tokyo, Japan |
|
30 |
somniferum |
S |
|
|
|
70DB |
84V |
|
Japan, perhaps originally from North·China |
|
|
|
C |
|
|
|
73DB |
88V |
|
|
|
31 |
rhoeas |
C |
none |
|
|
|
|
|
Tokyo, Japan |
|
32 |
orientale |
L |
20BG | 28DB |
|
|
92V |
|
Kassel, Germany |
|
|
|
|
22BG | 30DB |
|
|
95V |
|
|
|
|
|
|
20BG | 29 DB |
|
|
94V |
|
|
|
33 |
argemone |
L |
none |
|
|
|
|
|
Louvain Belgium |
|
34 |
rhoeas |
L |
|
|
|
|
|
93V |
Rome, Italy |
|
35 |
pyrenaicum |
L |
|
|
|
|
|
90V |
Hamburg, Germany |
|
36 |
polare |
L |
17V | 21DB |
|
75DV |
|
95V |
Hamburg, Germany |
|
37 |
atlanticum |
L |
16V | 21DB |
|
74DV |
|
94V |
Vienna, Austria |
|
38 |
rubriaurantiacum |
L |
16V | 20DB |
|
72DV |
|
92V |
Helsinki, Finland |
|
No. |
Poppy |
Rf relative to 100 and Colouration |
Origin | ||||||
|---|---|---|---|---|---|---|---|---|---|
|
|
Papaver |
|
|
|
|
|
|
|
|
|
39 |
rupifragum var. atlanticum |
L |
|
18V | 21DB | 41DB | 72DV | 96V |
Helsinki, Finland |
|
|
|
|
|
17V | 23DB |
|
69DV | 91V |
|
|
40 |
rhoeas |
L |
|
|
|
|
|
92V |
Helsinki, Finland |
|
41 |
alpinum |
L |
|
none |
|
|
|
|
Uppsala, Sweden |
|
42 |
apulum |
L |
|
none |
|
|
|
|
Uppsala, Sweden |
|
43 |
glaucum |
L |
|
|
|
|
|
86V |
Uppsala, Sweden |
|
44 |
lecoqii |
L |
|
|
|
|
|
95V |
Uppsala, Sweden |
|
45 |
pilosum |
L |
|
17V | 22DB |
|
70DV | 93V |
Uppsala, Sweden |
|
46 |
rhoeas "Shirley Wilks" |
L |
|
|
|
|
|
89V |
Wageningen, Netherlands |
|
47 |
lateritium |
L |
|
18V | 23DB | 40V |
|
89V |
Berlin-Dahlem, Germany |
|
48 |
rubro-aurantiacum |
L |
|
18V | 24DB | 41V |
|
91V |
Basle, Switzerland |
|
49 |
pavoninum |
L |
|
none |
|
|
|
|
London, United Kingdom |
|
50 |
umbrosum |
L |
|
|
|
|
|
94V |
Klagenfurt, Austria |
|
51 |
aurantiacum |
L |
|
16V | 19DB | 37DB |
|
90V |
Munich, Germany |
|
52 |
mairei |
L |
|
|
21DB |
|
|
88V |
Munich, Germany |
|
53 |
monanthum |
L |
|
|
|
|
|
93V |
Munich, Germany |
|
54 |
persicum |
L |
|
17V | 20DB |
|
|
94V |
Munich, Germany |
|
55 |
oreophilum |
L |
|
|
|
|
|
92V |
Munich, Germany |
|
56 |
argemone |
L |
|
|
|
|
|
93V |
Cologne, Germany |
|
57 |
bracteatum |
L |
|
|
22DB | 30DB |
|
94V |
Cologne, Germany |
|
58 |
nudicaule |
L |
|
none |
|
|
|
|
Cologne, Germany |
|
59 |
californicum |
L |
|
|
|
|
|
92V |
Dublin, Eire |
|
60 |
hybridum |
L |
|
none |
|
|
|
|
Dublin, Eire |
|
61 |
pyrenaicum |
L |
|
|
|
|
|
92V |
Dublin, Eire |
|
62 |
triniaefolium |
L |
|
|
|
|
|
91V |
Dublin, Eire |
|
63 |
lateritium |
L |
|
16V | 19DB |
|
|
90V |
Dublin, Eire |
|
64 |
nudicaule |
L |
|
12V |
|
|
|
86V |
Heidelberg, Germany |
|
65 |
monanthum |
L |
|
12DV | 14DB | 3lB | 67V | 86V |
Sofia, Bulgaria |
|
66 |
dubium |
L |
3V |
|
|
|
|
|
Toulouse, France |
|
67 |
collinum |
L |
2V |
|
|
|
|
|
Nantes, France |
|
68 |
hybridum L |
L |
|
none |
|
|
|
|
Uppsala, Sweden |
|
69 |
rhoeas var. latifolium |
L |
|
11DB |
|
|
|
91V |
London, United Kingdom |
|
70 |
schinzianum |
L |
|
14DB |
|
|
|
91V |
Braumchweig, Germany |
|
71 |
rupifragum |
L |
|
13DB |
|
|
|
88V |
Warsaw, Poland |
|
72 |
orientale |
L |
3V | 12BG | 18B |
|
|
88V |
Graz, Austria |
|
Species |
Chromosome number 2n |
Previous determinations a b 2n |
species |
Chromosome number 2n |
Previous determinations a b 2n |
|
Papaver |
|
|
Papaver |
|
|
|
alpinum |
14 |
14 (Ljungdahl, 1922) (Sugiura, 1936, |
hybridum |
14 |
14 (Ljungdahl, 1922) |
|
1940) (Faberge, 1944) |
(Sugiura, 1937, 1940) | ||||
|
apulum |
14 |
12 (Sugiura, 1936) |
lateritium |
14 |
14 (Ljungdahl, 1922) Yasui, 1941) |
|
argemone |
42 |
12 (Beale, 1939); |
lecoqii |
14 | |
|
42 (Sugiura, 1936, 1940) |
mairei |
14 | |||
|
atlanticum |
14 |
14 (Ljungdahl, 1922) |
monanthum |
14 | |
|
12, 14 (Sugiura, 1940) |
nudicaule |
14 |
14 (Ljungdahl, (1922)(Fabere, 1944) | ||
|
aurantiacum |
14 |
28 (Horn, 1938) | |||
|
bracteatum |
42 |
14 (Yasui, 1936) |
oreophilum |
14 |
14 (Sugiura, 1936) |
|
californicum |
14 |
orientale |
42 |
42 (Yasui, 1921) (Ljungdahl, 1922) | |
|
collinum |
28 |
28 (Snoad, 1952) 42 (Yamazzaki, 1936) | |||
|
glaucum |
14 |
14 (Sugiura, 1931, 1944) |
pavoninum |
12 |
12 (Sugiura, 1931, 1936) |
|
(Kuzmina, 1935) |
persicum |
14 |
14 (Ljungdahl, 1922) |
|
Species |
Chromosome number 2n |
Previous determinations a b 2n |
Species |
Chromosome number 2n |
Previous determinations a b 2n |
|
Papaver |
|
|
Papaver |
|
|
|
pilosum |
14 |
28 (Ljungdahl, 1922) |
rupifragum var. |
||
|
polare |
14 |
atlanticum |
14 | ||
|
pyrenaicum |
14 |
schinzianum |
14 | ||
|
rhoeas |
14 |
14 (Tahara, 1915) (Vilcins & Abele, |
setigerum |
44 |
22 (Sugiura, 1940); 44 (Kuzmina, 1935) |
|
1927) (Yamagaki, 1936) (Sugiura, |
44 (Ljungdahl, 1922) | ||||
|
1940) (Lawrence, 1930) (Felfoldy: |
44 (Heimburger, Moore, 1953) c | ||||
| 1947) |
somniferum |
22 |
22 (Furusato, 1940) (Tahara, 1915) | ||
|
rhoeas var. |
22 (Yasui, 1921) (Ljungdahl, 1922) | ||||
|
latifolium |
14 |
22 (Kuzmina, 1935) 20 (Ghimpu, 1933) | |||
|
rubriaurantiacum |
14 |
triniaefolium |
14 |
28 (Sugiura, 1940) | |
|
rubro-aurantiacum |
14 |
umbrosum |
14 |
14 (Sugiura, 1931, 1936) |
a C. D. Darlington and A. P. Wylie. Chromosome Atlas of Flowering Plants, 2nd. ed. 33-34, London: George Allen & Unwin Ltd. 1955.
Georg Tischeer, "Pflanzliche Chromosomen-Zahlen" in Tabulae Biologicae
4, 24 (1927); 12, 65 (1936); 16, 172-3 (1958); also Die Chromosomen-Zahlen der Gefassp flanzen Mitteleuropas ('s-Gravenhage: Dr. W. Junk, 1950).
c See footnote I, p. 20.
For 98 varieties or strains of Papaver somniferum, using in most cases seeds from foreign botanical gardens to obtain the plants, the morphine content of the opium was determined and compared with that of opium poppy plants of ordinary Japanese origin.
When sufficient opium could be collected from plants of one strain, the method of assay of the Japanese Pharmacopoeia VI was used. When the amount of opium was smaller, this method was modified to use only 1 gramme of opium. When less than 1 gramme was the most that could be collected, our method of paper chromatography and spectrophotometry was used).[9]
The results are given in Table IV.
The poppies were sown in October 1954 and 1955 and grown under uniform cultural conditions at the Kasukabe Experiment Station (numbers 1 to 26, 58 to 98) and the Wakayama Experiment Station (numbers 27 to 57). The method of incision was the usual practice described in section 1.
It is remarkable that the ten highest results for morphine percentage are on opium of poppies of which the seeds came from certain quite northern botanical gardens, and they are above 20%; but the quantity of opium was low.
|
Variety No. |
Morphine percentage |
Seed origin |
|
25 |
30.0 |
Copenhagen, Denmark |
|
64 |
27.5 |
Tabor, Czechoslovakia |
|
11 |
24.9 |
Cologne, Germany |
|
22 |
24.0 |
Montreal, Canada |
|
19 |
23.6 |
Cologne, Germany |
|
20 |
23.0 |
Munich, Germany |
|
67 |
22.1 |
Nantes, France |
|
90 |
21.2 |
Li?, Belgium |
|
91 |
21.0 |
Seattle, United States of America |
|
63 |
20.7 |
Hamburg, Germany |
In these cases the determinations were all made by our paper chromatographic method. This method possibly yields somewhat higher results than the precipitation method of the Japanese Pharmacopoeia, on the same opium.
However, it is to be noted that the highest results by the modified J.P. VI method were the following:
|
Variety No. |
Morphine percentage |
Seed origin |
|
36 |
20.04 |
Copenhagen, Denmark |
|
47 |
19.82 |
Cologne, Germany |
It is very interesting to observe that two samples of variety monstrosum, No. 25 and 67, gave very high results; 30% and 22.1%.
Contrary to the high morphine percentage in some opiums of foreign seeds, the ordinary Japanese opium "Ikkanshu" gave medium results.
However, opium weight per capsule was also calculated. The poppies of Japanese origin, "Ikkanshu", produced a large quantity of opium. As can be seen in Table IV, the poppies which gave more opium than 150 mg per capsule were all of the "Ikkanshu" strain. Therefore the Japanese strain "Ikkanshu" is superior in productivity of opium to all other poppies that we have grown in Japan from foreign seeds; but in these trials so far we have used seeds from botanical gardens and not seeds from poppies used for commercial opium production in other countries.
The poppies producing opium of exceptionally high morphine percentage, such as Danish and Czech poppies, produced a very small quantity of opium per capsule; but such poppies have great interest for trials as parent stock for commercial production, and our paper chromatographic method is a very convenient means for selecting plants of high productivity.
|
Method used: |
Period of incision: |
|
A. Japanese Pharmacopoeia VI method. |
|
|
B. Slightly modified J.P.VI method using 1 g of opium. |
Nos. 1-26, 27-57, 92-98 in 1955. |
|
C. Our paper chromatographic method. |
Nos. 58-91 in 1956. |
Poppies grown at the Kasukabe Experiment Station
|
No. |
Morphine percentage |
Determination method used |
Opium weight per capsule |
Capsules incised |
Period of. incision |
Plant height |
Seed origin |
|
|
|
|
mg |
|
|
cm |
|
|
1c |
9.26 |
B |
170.2 | 23 |
23 - 30. V |
125 |
Wakayama, Japan |
|
2c |
15.48 |
B |
232.5 | 12 |
23 - 30. V |
120 |
Wakayama, Japan |
|
3c |
11.97 |
B |
115.5 | 27 |
23 - 30. V |
132 |
Wakayama, Japan |
|
4c |
7.90 |
B |
166.1 | 24 |
23 - 30. V |
122 |
Wakayama, Japan |
|
5c |
12.45 |
B |
159.1 | 23 |
23 - 30. V |
125 |
Wakayama, Japan |
|
6 |
16.71 |
B |
60.5 | 30 |
23 - 30. V |
120 |
Aichi, Japan |
|
7 |
12.98 |
B |
116.0 | 27 |
23 - 30. V |
125 |
Aichi, Japan |
|
8 |
13.43 |
B |
72.2 | 25 |
27.V - 1. VI |
133 |
Utrecht (Baarn), Netherlands |
|
9 |
12.31 |
B |
44.6 | 53 |
1 - 6. VI |
125 |
Louvain, Belgium |
|
10 |
15.6 |
C |
42.3 | 17 |
1 - 6. VI |
135 |
Turin, Italy |
|
11 |
24.9 |
C |
2.9 | 24 |
7 - 12. VI |
150 |
Cologne, Germany |
|
12 |
19.4 |
C |
6.9 | 25 |
1 - 6. VI |
142 |
Dublin, Eire |
|
13a |
18.4 |
C |
5.3 | 29 |
1 - 6. VI |
147 |
Rome, Italy |
|
14 |
15.2 |
C |
4.3 | 30 |
1 - 6. VI |
151 |
Li?, Belgium |
|
15 |
12.7 |
C |
4.6 | 34 |
1 - 6. VI |
125 |
Basle, Switzerland |
|
16 |
16.1 |
C |
2.3 | 26 |
1 - 6. VI |
143 |
Berlin-Dahlem, Germany |
|
17 |
10.76 |
A |
89.9 | 120 |
23 - 30. V |
128 |
Japan, perhaps originally from North China |
|
18 |
17.8 |
C |
2.9 | 30 |
1 - 6. VI |
133 |
Bremen, Germany |
|
19 |
23.6 |
C |
1.4 | 33 |
1 - 6. VI |
147 |
Cologne, Germany |
|
20 |
23.0 |
C |
1.5 | 27 |
1 - 6. VI |
140 |
Munich, Germany |
|
21 |
14.6 |
C |
4.4 | 37 |
1 - 6. VI |
134 |
Delft, Netherlands |
|
22 |
24.0 |
C |
1.3 | 22 |
1 - 6. VI |
133 |
Montreal, Canada |
|
23 |
15.1 |
C |
0.3 | 33 |
1 - 6. VI |
100 |
Edinburgh, United Kingdom |
|
24 |
15.4 |
C |
0.2 | 39 |
1 - 6. VI |
135 |
Copenhagen, Denmark |
|
25b |
30.0 |
C |
0.9 | 44 |
7 - 12. VI |
144 |
Copenhagen, Denmark |
|
26a |
10.4 |
C |
0.2 | 28 |
1 - 6. VI |
120 |
Naples, Italy |
|
58 |
11.0 |
C |
47.4 | 17 |
15 - 19. V |
65 |
Formosa, China |
|
59c |
11.14 |
B |
157.4 | 25 |
28. V- 3. VI |
114 |
Wakayama, Japan |
|
60 |
15.41 |
B |
83.2 | 15 |
28.V - 3. VI |
126 |
Utrecht (Baarn), Netherlands |
|
61 |
10.3 |
C |
38.1 | 17 |
6 - 10. VI |
102 |
Parma, Italy |
|
62a |
18.8 |
C |
10.9 | 23 |
6 - 10. VI |
91 |
Adelaide, Australia |
|
63 |
20.7 |
C |
51.5 | 12 |
11 - 15. VI |
126 |
Hamburg, Germany |
|
64 |
27.5 |
C |
22.9 | 8 |
11 - 15. VI |
141 |
Tabor, Czechoslovakia |
|
65 |
16.4 |
C |
34.9 | 4 |
11 - 15. VI |
137 |
Delft, Netherlands |
|
66 |
15.56 |
B |
60.4 | 19 |
6 - 12. VI |
135 |
Strasbourg, France |
|
67b |
22.1 |
C |
32.0 | 5 |
11 - 15. VI |
146 |
Nantes, France |
|
68a |
11.7 |
C |
40.5 | 6 |
11 - 15. VI |
137 |
Bratislava, Czechoslovakia |
|
69 |
14.5 |
C |
51.4 | 18 |
17 - 22. V |
51 |
Formosa, China |
|
70 |
17.34 |
B |
102.0 | 23 |
3- 9. VI |
128 |
Louvain, Belgium |
|
71c |
13.30 |
B |
184.9 | 12 |
28.V- 3. VI |
121 |
Wakayama, Japan |
|
72 |
19.2 |
C |
38.0 | 18 |
11 - 15. VI |
151 |
Seattle, United States of America |
|
73 |
19.8 |
C |
25.5 | 9 |
11 - 15. VI |
151 |
Braunschweig, Germany |
|
74 |
17.1 |
C |
25.6 | 9 |
11 - 15. VI |
154 |
Lodz, Poland |
|
75 |
14.6 |
C |
53.8 | 15 |
3 - 9. VI |
130 |
Florence, Italy |
|
76 |
16.0 |
C |
26.0 | 12 |
6 - 12. VI |
121 |
Florence, Italy |
|
77 |
17.82 |
B |
84.2 | 15 |
3 - 9. VI |
135 |
Louvain, Belgium |
|
78c |
14.13 |
B |
205.5 | 12 |
28.V- 3. VI |
116 |
Wakayama, Japan |
|
79 |
18.6 |
C |
16.8 | 9 |
6- 10. VI |
145 |
Lodz, Poland |
|
80 |
17.5 |
C |
31.2 | 12 |
6 - 10. VI |
136 |
Bonn, Germany |
|
81a |
13.9 |
C |
21.4 | 12 |
14- 16. VI |
127 |
Nantes, France |
|
82 |
17.7 |
C |
56.9 | 16 |
11 - 15. VI |
108 |
Besancon, France |
|
83 |
18.2 |
C |
47.2 | 10 |
11 - 15. VI |
94 |
Dublin, Eire |
|
84 |
14.70 |
B |
98.3 | 12 |
3 - 9. VI |
131 |
Turin, Italy |
|
No. |
Morphine percentage |
Determination method used |
Opium weight per capsule |
Capsules incised |
Period of. incision |
Plant height |
Seed origin |
|
|
|
|
mg |
|
|
cm |
|
|
85c |
15.97 |
B |
184.2 | 23 |
28.V - 3.VI |
116 |
Wakayama, Japan |
|
86 |
14.9 |
C |
63.4 | 12 |
6 - 10.VI |
120 |
Besancon, France |
|
87 |
17.1 |
C |
45.2 | 6 |
11 - 15.VI |
138 |
Bremen, Germany |
|
88 |
15.2 |
C |
101.6 | 4 |
3 - 9.VI |
125 |
Taranto, Italy |
|
89a |
14.3 |
C |
16.1 | 52 |
3 - 7.VI |
97 |
Lisbon, Portugal |
|
90 |
21.2 |
C |
36.5 | 4 |
11 - 13.VI |
134 |
Li?, Belgium |
|
91 |
21.0 |
C |
12.9 | 8 |
11-13.VI |
143 |
Seattle, United States of America |
|
92 |
9.0 |
C |
36.6 | 29 |
7 - 9.VI |
162 |
Rome, Italy |
|
93a |
12.9 |
C |
11.4 | 65 |
7 - 12.VI |
110 |
Adelaide, Australia |
|
94 |
17.6 |
C |
39.2 | 32 |
7 - 12.VI |
132 |
Braunschweig, Germany |
|
95 |
15.8 |
C |
8.0 | 57 |
7 - 12.VI |
125 |
London, United Kingdom |
|
96 |
16.0 |
C |
1.9 | 48 |
7 - 12. VI |
145 |
Amsterdam, Netherlands |
|
97 |
14.3 |
C |
18.4 | 33 |
7 - 12.V1 |
140 |
Amsterdam, Netherlands |
|
98 |
14.8 |
C |
1 7 | 64 |
1 - 6.VI |
160 |
Turin, Italy |
a Var. plenum
bVar. monstrosum
c"Ikkanshu"
|
No. |
Morphine percentage |
Determination method used |
Seed origin |
No. |
Morphine percentage |
Determination method used |
Seed origin |
|
27 |
10.25 |
A |
Wakayama, Japan |
42 | 16.0 |
C |
Poznan, Poland |
|
28 |
9.85 |
A |
Wakayama, Japan |
43 | 14.7 |
C |
Louvain, Belgium |
|
29 |
10.42 |
A |
Wakayama, Japan |
44 | 17.6 |
C |
Braunschweig, Germany |
|
30 |
11.57 |
A |
Wakayama, Japan |
45 | 11.4 |
C |
Li?, Belgium |
|
31 |
11.10 |
A |
Wakayama, Japan |
46 | 16.61 |
B |
Delft, Netherlands |
|
32 |
9.65 |
A |
Wakayama, Japan |
47 | 19.82 |
B |
Cologne, Germany |
|
33 |
10.47 |
A |
Wakayama, Japan |
48 | 17.51 |
B |
Berlin-Dahlem, Germany |
|
34 |
11.58 |
A |
Wakayama, Japan |
49 | 17.83 |
B |
Basle, Switzerland |
|
35 |
11.24 |
A |
Wakayama, Japan |
50 | 18.3 |
C |
London, United Kingdom |
|
36 |
20.04 |
B |
Copenhagen, Denmark |
51 | 16.6 |
C |
Bremen, Germany |
|
37 |
17.6 |
C |
Copenhagen, Denmark |
52 | 17.73 |
B |
Amsterdam, Netherlands |
|
38 |
9.60 |
A |
Wakayama, Japan |
53 | 13.6 |
C |
Cologne, Germany |
|
39 |
17.1 |
C |
Frankfurt, Germany |
54 | 12.66 |
B |
Dublin, Eire |
|
40 |
16.98 |
B |
Seattle, United States of |
55 | 16.91 |
B |
Amsterdam, Netherlands |
|
America |
56 | 11.65 |
A |
Wakayama, Japan | |||
|
41 |
16.08 |
B |
Seattle, United States of |
57 | 11.43 |
B |
Japan, perhaps originally |
|
America |
|
|
|
from North China |
The content of "porphyroxine-meconidine" of opium from successive lancings of the same poppy capsules was determined.
The method used was based on the one described by L. Fuchs and W. Ullrich.10 Some modifications were introduced.
Experimental
About 0.15 g of opium was accurately weighed. In a small mortar, 0.1 g of calcium hydroxide and 2.5 cc of water, precisely measured, were added and the mixture rubbed to a smooth paste. After addition of 12.5 cc of water, precisely measured, the mixture was stirred for 20 minutes.
It was then filtered. 10 cc of the filtrate, precisely measured and brought into the extraction apparatus (fig. 5), was extracted continuously with peroxide-free ether for about 4 hours until a negative test for "porphyroxine-meconidine" was obtained with the return ether.
The ether was then evaporated on the steam-bath. To the residue, 0.5 cc of dilute HC1 (1 part by volume of conc. HC1 to 99 of water) was added, and the solution was heated in a boiling water-bath. After 5 minutes the colour was fully developed. The red solution was rinsed in to make exactly 40 cc or 20 cc in a volumetric flask.
The absorbance of the red-coloured solution was measured by a Beckman 13 spectrophotometer at 511 mµ, using a 1 cm cell.

The value for 10 cc of solution obtained from 0.1 g opium was calculated.
Opium
( b) From Papaver somniferum L.-" Ikkanshu" cultivated during 1954/55 at the Wakyama Experiment Station attached to the National Hygienic Laboratory.
By "Kiritori" method.-" Kiritori" is a method of collecting the latex from poppy capsules. Its meaning is cutting (Kiri) and taking from (tori), that is, to incise the unripe capsule and immediately take the latex away from the poppy capsule.
By" Oigaki" method.-" Oigaki" is another method. It means an additional (Oi) collection (gaki), that is, to collect the latex (" Asabuki ") which blows out (buki) early the following morning (Asa) from the same incision made in the "Kiritori" method.
( d) From P. somniferum cultivated during 1954/55 by a drug company (Nippon Shinyaku) in Kyoto.
White flowered variety, originally from Pakistan.
Violet-flowered variety, originally from Pakistan.
Opium samples distributed by the United Nations. UN No. 180 (first lancing), and UN No. 181 (second lancing); India, Rajasthan, variety Dhaturia.
|
Date of lancing | ||
|---|---|---|
|
|
c |
d |
|
I |
29 May | 2 June |
|
II |
31 May | 4 June |
|
III |
2 June |
|
|
IV |
4 June |
|
Results
Calculated values for the absorbance of the coloured solution of 10 cc, obtained from 0.1 g of opium, measured with Beckman B spectrophotometer at 511 mµ.
|
Lancing |
(a) |
(b) |
(c) |
(d) |
(e) |
|
I |
1. 524 |
2.175 | 2.129 | 1.157 | 0.755 |
|
II |
1.915 | 3.167 | 2.267 | 0.380 | 1.115 |
|
III |
2.487 | 2.407 | 2.145 |
|
|
|
IV |
2.228 | 2.376 | 2.973 |
|
|
Volume in which measurement carried out (cc.)
| 40 | 40 | 40 | 20 | 20 |
Later experiments were made on opium front poppies grown from foreign seed obtained from botanical gardens with the following results:
|
Opium sample |
Absorbance lancing |
Country where seeds obtained | |
|
|
I |
II. |
|
|
f |
0.728 | 1.550 |
Rome, Italy |
|
g |
0.123 |
0. 589 |
Amsterdam, Netherlands |
|
h |
0.594 | 1.448 |
Braunschweig, Germany |
|
i |
0.702 | 1.152 |
Turin, Italy |
Calculated by the equivalent worked out by Farmilo and Kennett,11 the "Lovibond values" of the above readings should be the following (multiplication of the "1%" absorbance in a 1-cm cell by 30.2):
|
Lancings |
|||||
|---|---|---|---|---|---|
|
Opium sample |
Seed origin | ||||
|
|
I |
II |
III |
IV |
|
|
a |
46.0 | 57.8 | 75.1 | 67.3 |
Japan |
|
b |
65.7 | 95.2 | 72.7 | 71.7 |
Japan |
|
c |
64.3 | 68.5 | 64.8 | 89.8 |
Pakistan |
|
d |
34.9 | 11.5 |
|
|
Pakistan |
|
e |
22.8 | 33.7 |
|
|
India |
|
f |
22.0 | 46.8 |
|
|
Italy |
|
g |
3.7 | 17.8 |
|
|
Netherlands |
|
h |
17.9 | 43.7 |
|
|
Germany |
|
i |
21.2 | 34.8 |
|
|
Italy |
In Table V are given the results of comparative colorimetric determinations of "porphyroxine-meconidine" in Japanese opiums, in comparison with opiums of five other countries (China, India, Iran, Korea, Turkey). Twelve Japanese samples of recent production are included, with one sample of the former legal production, and two samples (Nos. 402 and 435) from poppies grown experimentally in Japan. Only in the case of one of the latter samples is the value as low as for the opiums of China, Iran, Korea, or Turkey (in the samples examined).
In our earliest determinations (in 1954) comparisons were made with a photoelectric colorimeter, using filter S.53. These have been converted to calculated" Lovibond values ", for ready, comparison with values given by the UN Secretariat, by the factor obtained by Fuchs & Ullrich,12 equivalent to multiplication of the "1%" absorbance by 39.5. In the second part of the table are determinations made with the Beckman B spectrophotometer. For these the "Lovibond values" have been calculated using the equivalent found by Farmilo and Kennett, already mentioned.
MAP II. Villages of Opium Poppy Cultivation
Black spot shows the village in which the opium poppy was actually cultivated under licence during 1954/55.
|
Nagano |
|
Wakayama |
|
|
1. |
Ooka, Sarashina |
1. |
Tasugawa, Arita |
|
2. |
Murakami, Sarashina |
2. |
Yuasa, Arita |
|
3. |
Sakagi, Hanishina |
3. |
Hiro, Arita |
|
4. |
Tanaka, Chiisagata |
4. |
Yura, Hidaka |
|
|
|
5. |
Ena, Hidaka |
|
Aichl |
|
Hiroshima |
|
|
|
|
1. |
Ono, Saeki |
|
1. |
Akabane, Atsumi |
2. |
Itsukaichi, Saeki |
|
1. |
Tawara, Atsumi |
3. |
Kurose, Kamo |
|
|
|
4. |
Suigano, Mitsugi |
|
Osaka |
|
5. |
Fuchu |
|
1. |
Toyokawa, Mishima |
Okayama |
|
|
2. |
Fukui, Mishima |
1. |
Sakuto, Aida |
|
3. |
Takatsuki |
|
|
|
4. |
Ibaragi |
Hyogo |
|
|
5. |
Suita |
1. |
Anji, Shiso |
Conclusions
The content of porphyroxine in Japanese opium is nearly always high, and sometimes extremely high. This is very characteristic.
The changes in the porphyroxime content with repeated lancings do not seem to be entirely regular, but in eight cases out of nine the content in opium of the second lancing was greater than in opium of the first lancing.
TABLE V
|
Lab. No. |
Absorbance (1%) |
Calculated Lovibond value |
Origin |
|
1 |
0.209 | 8.3 |
Turkey, Aydin, UN 1 D |
|
15 |
0.331 | 13.1 |
Turkey, export type, UN 15 |
|
18 |
0.192 | 7.6 |
Iran, Fars, UN 47 |
|
19 |
0.154 | 6.1 |
Iran, Khorassan, UN 51 |
|
24 |
0.835 | 33.0 |
India, Malwa, UN 35 |
|
35 |
0.220 | 8.7 |
China (Manchuria) |
|
36 |
0.240 | 9.5 |
China (Inner Mongolia) |
|
Lab. No. |
Absorbance (1%) |
Calculated Lovibond value |
Origin |
|
37 |
0.324 | 12.8 |
Korea (produced before World War II) |
|
38 |
0.128 | 5.1 |
China (Northern) |
|
39 |
1.917 | 75.7 |
Japan (former legal production) |
|
37 |
0.365 | 11.0 |
Korea, produced in Korea before World War II |
|
39 |
2.500 | 75.5 |
Japan, former legal production |
|
117 |
1.220 | 36.9 |
India, United Nations, Malwa (Excise) |
|
401 |
1.464 | 44.3 |
Japan, Aichi |
|
402 |
0.338 | 10.2 |
Japan, Kyoto, Takeda Herbal Garden |
|
403 |
2.008 | 60.7 |
Japan, Osaka |
|
404 |
1.376 | 41.5 |
Japan, Osaka |
|
405 |
2.276 | 68.8 |
Japan, Osaka |
|
408 |
2.778 | 84.0 |
Japan, Wakayama |
|
409 |
2.704 | 81.7 |
Japan, Wakayama |
|
435 |
1.000 | 30.2 |
Japan, Wakayama, Experiment Station of National Hygienic Laboratory |
|
436 |
3.344 | 101.0 |
Japan, Wakayama |
|
437 |
0.328 | 9.9 |
Iran, from Iranian Commissioner |
|
438 |
0.411 | 12.4 |
Turkey, import |
|
501 |
3.377 | 102.0 |
Japan, Wakayama |
|
502 |
2.674 | 80.8 |
Japan, Osaka |
|
503 |
0.971 | 29.3 |
Japan, Aichi |
|
504 |
1.583 | 47.8 |
Japan, Nagano |
|
505 |
0.820 | 24.8 |
Japan, Hiroshima |
TABLE VI
Morphine percentages in Japanese opium, crop season 1953/54
Method: All except 402, 435 by our K/15 method 13 402 and 435 by a modification of the Eder & Wäckerlin method 14
|
Lab. No. |
Morphine % |
Origin (in Japan) |
|
401 |
13.99 |
Kambe, Atsumi, Aichi |
|
402 |
10.75 |
Ichijoji, Sakyo, Kyoto |
|
403 |
10.75 |
Fukui, Mishima, Osaka |
|
404 |
14.24 |
Fukui, Mishima, Osaka |
|
405 |
10.29 |
Ibaragi, Osaka |
|
406 |
11.97 |
Ibaragi, Osaka |
|
407 |
11.53 |
Takatsuki, Osaka |
|
408 |
15.61 |
Minamihiro, Arita, Wakayama |
|
409 |
15.37 |
Minamihiro, Arita, Wakayama |
|
Lab. No. |
Morphine % |
Origin (in Japan) |
|
410 |
15.64 |
Minamihiro, Arita, Wakayama |
|
411 |
12.07 |
Tsugi, Arita, Wakayama |
|
412 |
15.73 |
Minamihiro, Arita, Wakayama |
|
413 |
11.80 |
Tsugi, Arita, Wakayama |
|
414 |
13.87 |
Minamihiro, Arita, Wakayama |
|
415 |
15.91 |
Minamihiro, Arita, Wakayama |
|
416 |
17.60 |
Minamihiro, Arita, Wakayama |
|
417 |
15.49 |
Tsugi, Arita, Wakayama |
|
418 |
15.95 |
Hiro, Arita, Wakayama |
|
419 |
12.55 |
Tsugi, Arita, Wakayama |
|
420 |
11.64 |
Tsugi, Arita, Wakayama |
|
421 |
15.23 |
Minamihiro, Arita, Wakayama |
|
422 |
13.66 |
Minamihiro, Arita, Wakayama |
|
423 |
14.08 |
Hiro, Arita, Wakayama |
|
424 |
14.60 |
Hiro, Arita, Wakayama |
|
425 |
13.30 |
Hiro, Arita, Wakayama |
|
426 |
13.51 |
Hiro, Arita, Wakayama |
|
427 |
14.25 |
Hiro, Arita, Wakayama |
|
428 |
14.55 |
Hiro, Arita, Wakayama |
|
429 |
15.18 |
Hiro, Arita, Wakayama |
|
430 |
15.29 |
Minamihiro, Arita, Wakayama |
|
431 |
15.08 |
Minamihiro, Arita, Wakayama |
|
432 |
13.94 |
Minamihiro, Arita, Wakayama |
|
433 |
10.62 |
Tsugi, Arita, Wakayama |
|
434 |
11.96 |
Tsugi, Arita, Wakayama |
|
435 |
14.12 |
Yada, Hidaka, Wakayama |
In Table VI the laboratory analyses are given for Japanese opiums of the crop season of 1953/54, and in Table VII analyses are Summarized for the crop season of 1954/55.
As may be seen approximately (or calculated) from the latter table, the average content is only between 11% and 12 %, although opium of a much higher morphine percentage can be obtained by collecting the first lancings separately.
These determinations were made by the method of the Japanese Pharmacopoeia VI.
TABLE VII
Classification of Japanese opium samples according to morphine percentages, crop season 1954/55
|
Morphine percentage |
Province and number of samples | |||||||
|---|---|---|---|---|---|---|---|---|
|
|
Nagano |
Aichi |
Osaka |
Wakayama |
Hirochima |
Okayama |
Hyogo |
Total |
|
Below 4 |
|
1 |
|
|
|
|
|
1 |
|
4-5 |
|
|
|
1 |
|
|
|
1 |
|
5-6 |
|
2 |
|
2 |
|
|
|
4 |
|
6-7 |
|
|
|
1 | 3 |
|
1 | 5 |
|
7-8 |
|
|
2 | 3 | 6 | 2 | 5 | 18 |
|
8-9 |
3 | 13 | 12 | 16 | 9 | 3 | 4 | 60 |
|
9-10 |
6 | 27 | 50 | 36 | 6 | 5 | 8 | 138 |
|
10-11 |
10 | 59 | 76 | 161 | 7 | 8 | 10 | 331 |
|
11-12 |
11 | 64 | 59 | 243 | 10 | 8 | 3 | 398 |
|
12-13 |
17 | 48 | 34 | 184 | 6 | 1 | 1 | 291 |
|
13-14 |
5 | 23 | 7 | 127 | 3 | 1 | 2 | 168 |
|
14-15 |
4 | 19 | 4 | 41 |
|
1 |
|
69 |
|
15-16 |
|
9 |
|
11 |
|
|
|
20 |
|
16-17 |
1 | 6 | 1 | 3 | 1 |
|
|
12 |
|
17-18 |
|
4 |
|
|
1 | 1 |
|
6 |
|
Above 18 |
|
1 |
|
|
|
|
|
1 |
|
TOTAL.... |
57 | 276 | 245 | 829 | 52 | 30 | 34 | 1,523 |
C. G. Farmilo, H. L. J. Rhodes, H. R.. L. Hart & H. Taylor. "Detection of morphine in Papaver setigerum DC. ", Bulletin on Narcotics, Vol. V, No. 1, 26, 1953.
2Haruyo Asahina & Masako Ono, "Detection of morphine in poppy plants by paper chromatography ", United Nations document ST/SOA/ SER.K/39, 30 June 1955.
3Haruyo Asahina and Masako Ono, " Determination of morphine in opium tincture by paper chromatography ", United Nations document · ST/SOA/SER.K/40, 25 August 1955.
4Haruyo Asahina and Masako Ono, "Quantitative determination of morphine in opium by paper chromatography and spectrophotometry ", Bulletin on Narcotics, Vol. VIII, No. 4, 1956.
5An outline of this procedure is to be published by the United Nations Narcotics Division in the series ST/SOA/SER.K/...
6M. A. Vesselovskaya. "The Poppy - Its classification and its importance as an oleiferous crop " - Supplement 56 to the Bulletin of Applied Botany, of Genetics and Plant Breeding. The Lenin Academy of Agricultural Sciences in the USSR, Leningrad; 1933 (in Russian and English).
7An outline of this procedure is to be published by the United Nations Narcotics Division in the series ST/SOA/SER.K/...
8See footnote 2, p. 21.
9See footnote 4, p. 21.
10L. Fuchs & W. Ullrich, "The Colorimetric Determination of' Porphyroxine-Meconidine', 2nd communication. Experiments on the Determination of ‘ Porphyroxine-Meconidine' in Small Quantities of Opium". United Nations document ST/SOA/SER.K/19, 1 September 1953.
11Charles G. Farmilo & Patricia M. L. Kennett, "The Colorimetric Determination of ' Porphyroxine-Meconidine ', The Relation between Tintometer and Spectro-photometric Measurements obtained in the Determination of 'Porphyroxine-Meconidine' in Opiums ", United Nations document ST/SOA/SER.K/14, 26 January 1953.
12L. Fuchs & W. Ullrich, "The Colorimetric Determination of 'Porphyroxine-Meconidine'" United Nations document ST/SOA/ SER.K/10, 25 September 1952.
13Haruyo Asahina, "An assay procedure for opium based on that of the Japanese Pharmacopoeia, and morphine percentages obtained on samples from various countries ", United Nations document ST/SOA/SER. K/15, 11 March 1953. This procedure was adopted as official for the Japanese Pharmacopoeia as from 15 March 1955.
14R. Eder and E. Wackerlin, "The determination of morphine in opium ", Quarterly Journal of Pharmacy and Pharmacology, 10, 680 - 730, 1937.