Second Chapter1 Biological Assays of Morphine
I. CATATONY OF THE MOUSE'S TAIL
II. ACTION OF CHOLINIC ESTERS ON THE DENERVATED MUSCLE OF THE LEECH UNDER THE INFLUENCE OF MORPHINE
A. STUDY OF THE SENSITIZING EFFECT OF MORPHINE ON THE CETYLCHOLINIC CONTRACTION OF THE DENERVATED MUSCLE OF THE LEECH
B. STUDY OF THE ANTAGONIST EFFECT OF MORPHINE ON THE CONTRACTION CAUSED BY VARIOUS CHOLINIC ESTERS IN THE DENERVATED MUSCLE OF THE LEECH
III. SIMULTANEOUS APPLICATION OF THE THREE TESTS
IV. CONCLUSIONS
Third Chapter - What Happens to Morphine in the Organism
PART ONE - DETECTION OF OXYDIMORPHINE
PART TWO - DISAPPEARANCE, CONJUGATION AND ACCUMULATION OF MORPHINE in vitro AND in vivo IN NORMAL AND HABITUATED RATS
PART THREE - ELIMINATION OF MORPHINE BY THE HABITUATED ORGANISM
DETERMINATION OF THE QUANTITIES OF MORPHINE EXCRETED IN URINE AND FAECES BY NORMAL AND HABITUATED RATS
SUMMARY
PART FOUR - COMPARISON BETWEEN QUANTITIES OF MORPHINE ACCUMULATED IN THE ORGANISM OR ELIMINATED BY NORMAL AND HABITUATED RATS
CONCLUSION
Author: Denise G. Fichtenberg
Pages: 16 to 52
Creation Date: 1951/01/01
The readers of the "Bulletin on Narcotics" will regret to hear that Madame Denise G. Fichtenberg died suddenly on 25 September 1951. All who were acquainted with her, either personally or through her work, are united in paying homage to her memory and in sympathizing with the grief of her family. Born on 30 July 1907, she gave up her studies on her marriage, but took them up again in 1932. In 1934, as a "licencié ès-sciences naturelles"; she joined the staff of the Institute of Clinical Biology (Cochin Hospital) in the service of Prof. Achard, under the direction of Jeanne Lévy; she was later promoted to the post of assistant in the National Laboratory(Ministry of Public Health, Population and Academy of Medicine). From the beginning of her scientific career, she devoted herself to problems of general pharmacodynamics. She made important contributions to our knowledge of the mechanism of habituation to morphine in her doctor's (ès-sciences naturelles) thesis, a brilliant and appreciated work. She took part in the last International Congress on Physiology and as a Member of the "Société de Chimie Biologique de France" and of the "Union thérapeutique", Madame Fichtenberg discovered a technique for studying the rat's experimental habituation to morphine and for determining, through pharmacological dosages of morphine and of oxydimorphine, the fate of morphine in the organism. Thus became known internationally this intelligent and conscientious worker, whose life was devoted to science.
The difficulties involved in the study of what happens to morphine in the body and of its elimination relate to the extraction, the identification and the assay properly so-called of small quantities of morphine.
Knowing the difficulties of chemical micro-assays of this drug, explained in detail in the paper of R. Cahen (1935)2, I tried to determine the conditions of a pharmacological assay of morphine.
Since many authors have considered oxydimorphine as the first step in the degradation of morphine in living beings, I thought it essential to envisage the establishment of assay tests. These should permit:
A method of assaying small quantities of morphine which does not call for too laborious preliminary processes of extraction and purification.
The assay of morphine in the presence of at least equal quantities of oxydimorphine.
The identification and even the assay of oxydimorphine.
I studied on the one hand the reaction of the catatony of the mouse's tail (Straub, 1911) and on the other hand varying reaction of cholinic esters on the dorsal muscle of the leech, subjected to the action of Morphine and oxydimorphine.
Previous work. Herrmann (1912) used the behaviour of white mice receiving morphine hydrochloride, taking into consideration the observations of Straub. A subcutaneous injection into the mouse's back of doses of morphine hydrochloride between 0.005 mg. and 15 mg. produced a curvature of the tail in the form of an S, two to fifteen minutes after the injection, depending on the dose. Finally, the tail curves back along the body of the animal, the tip touching the centre of the head. It is a catatonic state emphasized by muscular movements, likerunning, and is best to be observed by placing the mice on a wooden board under a glass cloche and frightening them with a glass rod.
1The first part of this article was published in the Bulletin on Narcotics, vol. III, no. 3.
2Since that time, Hofmann and Popovici (1935) have advocated the use of the reduction of silicomolybdic acid by morphine in an alkaline medium, the blue colour of which depends on the quantity of morphine present; Deckert (1936-1938) has worked out a nephelometric process making it possible to evaluate the turbidity of the morphine-molybdate-vanadate complex for the assay of morphine in the tissues. The latter process has been improved and used by Oberst (1939)
The duration of this phenomenon varies with the doses administered. The following table given by Herrmann represents the average of several observations on mice of the same stock weighing about 16-20 g.
|
Solutions (Percentage) |
||||||
|---|---|---|---|---|---|---|
|
Morphine content (HCl) in mg. |
Beginning of the reaction (min.) |
Beginning of the maximum (min.) |
Beginning of the maximum (hours) |
Duration of the reaction (hours) | ||
|
1.5 cm 3 |
1 | 15 | 4 | 5 |
- |
- |
|
1 " |
1 | 10 | 3 | 4 |
- |
- |
|
0.5 " |
1 | 5 | 2 | 5 |
4.5 |
more than 20 |
|
0.25 " |
1 | 2.5 |
2-3 |
6 |
4.5 |
" 7 |
|
0.25 " |
0.5 | 1.25 | 6 | 8 | 4 |
" 6 |
|
0.1 " |
1 | 1 | 5 | 7 |
3.5 |
" 5 |
|
0.25 " |
0.25 | 0.625 | 7 | 9 | 4 |
5-6 |
|
0.5 " |
0.1 | 0.5 | 10 | 12 | 3 | 4 |
|
0.25 " |
0.125 | 0.3 | 6 | 11 |
2 1/2 |
4 |
|
0.2 " |
0.1 | 0.2 | 4 | 10 |
2 1/2 |
3 1/2 |
|
0.25 " |
0.0625 | 0.16 | 5 | 15 |
1 1/2 |
4 |
|
1.1 " |
0.1 | 0.1 | 6 | 12 | 2 | 4 |
|
0.2 " |
0.03 | 0.08 | 8 | 12 | 2 |
3 1/2 |
|
0.5 " |
0.01 | 0.05 | 7 | 13 | 2 | 3 |
|
0.25 " |
0.16 | 0.04 | 4 | 10 |
1 1/2 |
2 |
|
0.2 " |
0.01 | 0.02 | 13 | 20 | 1 |
1 1/2 |
|
0.1 " |
0.01 | 0.01 | 12 | 25 |
30-40 min. |
1 to 2 |
|
0.1 " |
0.005 | 0.005 | 15 |
- |
- |
45 min. |
The maximum represents the time during which the tail remains in its maximum position or returns to that position following a slight stimulus. For lethal doses (15 and 10 mg.) the reaction lasts only a short time (until paralysis sets in). For 0.005 mg. the maximum reaction does not occur. Herrmann considers 0.01 mg. as the lowest limit for which the maximum reaction can be observed. He found a direct relationship between the duration of the reaction, the duration of the maximum condition and the dose of morphine injected. These times make it possible to calculate approximately the quantity of morphine injected, especially if about 0.01 mg. is used.
He used that reaction to detect morphine in biological liquids and the organs. Despite the lack of accuracy and the need for using large groups of mice, it has the advantage of avoiding losses resulting from successive extractions.
Many authors, To and Rin (1933), Munch (1934), To (1935), Terada and Honda (1935), Juul (1939), Toff (1946), used. the same test to determine the quantity of morphine in the organs, the urine and the saliva of the horse.
Study of the test of the catatony of the tail
I myself used the Straub test to identify morphine and oxydimorphine, first in pure solutions and then added to organic extracts.
Each experiment is made on a group of twenty mice of the same stock, age and sex, weighing from 17 to 23 g.
The same animals are used again only after a rest period of eight days. They are given subcutaneous injections in the back in proportion to their weight, with the concentration of the 'solutions such that the volume injected does not exceed one cc. The mice are immediately placed on a wooden board covered with a large glass cloche with its upper end open to allow normal ventilation. While Herrmann determined the quantity of morphine injected by the total duration of the reaction and the duration of the maximum condition, Keil and Kluge (1934) obtained more consistent results by observing the percentage of animals with the maximum reaction and the length of that reaction. I followed the same technique as those authors.
A. PURE SOLUTIONS
(1) Effect of morphine. Doses of morphine hydro-chloride from 0.003 to 0.006 mg. per g. are injected subcutaneously into groups of twenty mice. For each dose the percentage of animals having the maximum reaction (the tail curved parallel .to the animal's back with its tip touching the middle of the head), the time of latency and the duration of the reaction is noted. The phenomenon can more easily be observed if the sides of the cloche are struck each five to ten minutes with a glass rod since the frightened mice begin to run and the curvature of the tail is accentuated.
The table below gives the results obtained with twenty mice and a dose of morphine hydrochloride of 0.004 mg. per g. For that dose 55 per cent of the animals showeds17 the maximum reaction. The average duration of the reaction was sixty-four minutes, the average time of latency twenty-three minutes. Individual variations are large (fifteen to forty minutes for the period of latency, thirty to ninety minutes for the duration of the reaction), making it necessary to experiment on groups of at least twenty mice.
|
Weight of the animals (in g.) |
Doses injected in each animal (in mg.) |
Maximum reaction time of latency (min.) |
Duration (min.) |
|---|---|---|---|
|
19 |
0.076 | ||
| 20.5 | 0.082 | 15 | 90 |
|
16 |
0.064 | 20 | 45 |
| 21.5 | 0.086 | 15 | 90 |
| 17 | 0.068 | 30 | 45 |
| 19.5 | 0.078 | ||
| 21 | 0.084 | 15 | 45 |
| 19 | 0.076 | ||
| 17 | 0.068 | 15 | 30 |
| 17.5 | 0.070 | ||
| 19 | 0.076 | ||
| 17.5 | 0.070 | 30 | 90 |
| 18 | 0.072 | 25 | 75 |
| 20 | 0.080 | ||
| 18.5 | 0.074 | 40 | 60 |
| 19 | 0.076 | ||
| 19 | 0.076 | 25 | 60 |
| 22.5 | 0.090 | ||
| 23 | 0.092 | ||
| 18.5 | 0.074 | 25 | 60 |
|
Average |
23 | 64 |

With that number of animals we found that the results were identical, as regards the percentage of animals reacting and the average duration of the reaction, for a dose of morphine hydrochloride.
|
Doses (δ per g.)* |
Percentage of animals showing catatony |
Average duration of the reaction (min.) |
|---|---|---|
| 5 | 75 | 94 |
| 5 | 70 | 90 |
In the course of this article δ stands for microgram.
Experiments made with increasing doses of morphine, hydrochloride are given below for groups of twenty mice.
|
Maximum reaction | ||||
|---|---|---|---|---|
|
Doses injected (δ per gr.) |
Concentration of solutions |
Percentage of animals with catatony |
Average latency period (min.) |
Average duration of reaction (min.) |
| 3 |
1/10,000 |
35 | 26 | 70 |
| 4 |
1/10,000 |
55 | 23 | 64 |
| 5 |
1/5,000 |
75 | 22 | 94 |
| 6 |
1/5,000 |
80 | 21 | 131 |
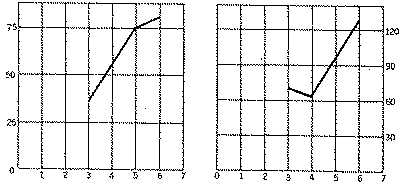
This table shows that the period of latency varies little, that the average period of reaction increases when the dose exceeds 0.004 mg. per g., and that the percentage of animals with catatony increases from 0.003 to 0.006 mg. per g. I have drawn graphs taking as abscissae the doses injected and as ordinates the percentages of animals reacting or the length of reaction.
With the help of these graphs it is possible to obtain the dose injected from the percentage of animals reacting and the length of reaction. Thus a solution injected at the rate of 1 cc. per 20 g., which I observed to have caused catatony in 35 per cent of the animals with an average length of reaction of seventy-six minutes, contained 0.003 mg. of morphine hydrochloride per cc.
(2) Action of oxydimorphine. Oxydimorphine hydrochloride injected subcutaneously into mice is about four times less effective than morphine hydrochloride (see table below). Thus a dose of 0.005 mg. per g. causes no maximum reaction in twenty mice. Twenty-five per cent of the animals show a maximum reaction to a dose of 0.01 mg. per g., the average duration being sixty-nine minutes. This dose corresponds to a morphine hydrochloride content of less than 0.003 mg. Furthermore, the reaction manifests itself much more slowly, the average period of latency being forty-six minutes as compared with twenty-six minutes in the case of 0.003 mg. of morphine hydrochloride.
Similarly, 0.015 mg. of oxydimorphine hydrochloride corresponds to less than 0.004 mg. of morphine hydrochloride.
|
Maximum reaction | ||||
|---|---|---|---|---|
|
Doses injected (δ per gr.) |
Concentration of solutions |
Percentage of animals with catatony |
Average latency period (min.) |
Average duration of reaction (min.) |
| 5 |
2/10,000 |
0 |
- |
- |
| 10 |
4/10,000 |
25 | 46 | 69 |
| 15 |
4/10.000 |
35 | 38 | 80 |
Active doses of oxydimorphine are four times larger than those of morphine, the reaction is less clear-cut and the period of latency is twice as long.
B. EFFECT OF MORPHINE ADDED TO ORGANIC EXTRACTS
Morphine hydrochloride added to the defibrinated blood of a dog or the homogenized liver of a rat causes a weaker reaction than a pure solution of morphine.
The extraction of morphine added to biological media is carried out by exhausting the blood (cold), and the tissues (under heat) with a volume of hydrochloric alcohol at 1/1000 equal to seven to ten times the weight of the liquid or of the organ concerned. The filtrate, evaporated to dryness under a blast of cold air, is dissolved with dilute hydrochloric acid (N/200). The solution is brought to a pH 6.3 with the help of N/100 diluted soda. The final concentration of the solution is such that the volume to be injected into the mice does not exceed 1 cc. per 20 g.
Injections made with these extracts contain about 20 per cent less morphine than the initial amounts. Thus when 0.025 cc. per g. of an extract containing 0.2 mg. of morphine per cc. is injected into mice, the proportion of animals reacting is 55 per cent and the average length of the reaction is sixty-five minutes, which according to graphs I and II corresponds to 0.004 mg. instead of 0.005 mg. In the case of a solution of another identical extract the proportion of animals reacting is 55 per cent and the average length of the reaction is 113 minutes. The number of animals reacting corresponds to 0.004 mg. instead of 0.005, a difference of 20 per cent; on the other hand the average length of reaction corresponds to 0.0055 mg. The lack of precision of the results does not depend on the method of extraction. As will be seen in a moment, the arraying of these extracts in the denervated muscle of the leech makes it possible to establish the quantity of morphine added. The error is due to the action of the extract itself.
(3) Advantages and drawbacks of this method. Since this method makes it possible to distinguish between morphine and oxydimorphine only from the quantitative standpoint, (oxydimorphine being four times less effective than morphine) and since the exact doses of morphine added to biological media cannot be determined by the extraction processes employed, it can be used only for the identification of these substances and not for an accurate assay.
Taken as a whole, the studies undertaken of the influence of morphine on the action of cholinic esters on the denervated muscle of the leech have shown that morphine sometimes produces an increase and sometimes a lessening of the contraction according to the esters employed.
Quastel, Tennenbaum and Wheatley (1936) and Quastel and Tennenbaum (1937) have demonstrated that morphine inhibits reversibly the contractions of the eserinated dorsal muscle of the leech under influence of acetylcholine. Kahane and Jeanne Lévy (1939) have established the influence of morphine on the contractions of the denervated muscle of the leech, caused by a whole series of ammonium quaternaries with ester functions; the addition of 10 to 200 δ of morphine hydrochloride to a Ringer bath of 20 cc. in which the denervated muscle of the leech is immersed has a different effect on the contraction, caused by the different esters. The following were observed: ( a) increased contraction in a first group of esters containing formyl-, acetyl-, propionyl- and isobutyrylcholine; ( b) reduced contraction, or even absence of contraction, in a second group containing acetyl- β-methylcholine, ethylic ester of betaine, and butyric, benzoic, hydrobromic, nitric and carbamic esters of choline.
Dodel, Dastugue and Bresson (1939) stressed the potentialization of acetylcholinic activity on the denervated non-eserinated dorsal muscle of the leech and showed that it is produced with many derivatives of morphine, heroin, dionin; codeine, dihydroxycodeinone, and with some alkaloids of opium (thebaine, papaverine), as well as with apomorphine.3
Kahane and Jeanne Levy (1939) interpreted morphine acetylcholine potentialization as due to two causes: the effect of potentialization (probably due to anticholinesterasic action) and that of antagonism (by direct action of morphine upon the muscle).
3It should also be mentioned that Mikhel'son (1941) uses the non-eserinated muscle of the leech to assay acetylcholine in the presence or absence of narcotics (morphine, codeine, diacetyl-morphine, ether and chloroform) and points out that morphine raises the sensitivity of acetylcholine through the inhibition of cholinesterasis. Torda and Wolff (1947) showed on the rectus abdominis of the frog that morphine in a highly concentrated form increases the effect of acetylcholine.
Morphine has an anticholinesterasic effect on pseudo-cholinesterasis (Kahane and Jeanne Lévy, 1936, Gautrelet and Scheiner, 1939), and on specific cholinesterasis of the brain (Bernheim and Bernheim, 1936,Annette Denys and Jeanne Lévy, 1947).
The antagonistic action on cholinic esters was demonstrated on the non-eserinated leech as regards esters insensitive to the action of cholinesterases (Kahane and Jeanne Lévy, 1936), and on the dorsal eserinated muscle of the leech as regards esters hydrolysed by cholinesterases by Quastel, Tennenbaum and Wheatley (1936) and by Kahane and Jeanne Lévy (1939).
We endeavoured to discover whether the phenomenon of morphine-acetylcholine potentialization on the denervated muscle of the leech has a quantitative character, in other words, whether, the dose of acetylcholine being constant, the potentialization depends on the dose of morphine acting upon the muscle.
We proceeded as follows:
The leech4 is affixed by the rear buccal and anal sucker s to a cork mat. The belly side is slit open all along the centre, beginning at the anal sucker. The nervous chain, situated to one side and recognizable by its ganglia and its dark colour, is carefully removed as are the genital organs.
A 2 cm. long fragment is taken from the front part of the animal (below the buccal sucker) and a fine thread is affixed to each end. By one of the threads the leech is attached to the curved tip of a glass tube which lets in the air (three to four air bubbles per second). The other thread is attached to the recording needle which registers the contractions of the muscle on a black cylinder revolving at a speed of 0.06 mm. per second. The glass rod and the muscle, placed in a cylindrical tube,5 are immersed in a bath of 20 cc. of a Ringer solution composed as follows:
|
Pure sodium chloride R.P.6 |
6.48 g. |
|
Pure potassium chloride R.P. |
0.14 g. |
|
Pure calcium chloride R.P. |
0.12 g. |
|
Pure monosodium phosphate R.P. |
0.01 g. |
|
Pure bicarbonate of soda. |
0.20 g. |
|
Distilled water Q.S.P. |
1,000 cc. |
Before commencing the experiment, the muscle, placed in the tube, is washed and left to itself for one and a half to two hours. Under the effect of slight tension it dilates and the needle reaches a horizontal position.
4It is recommended that the leeches should be kept in very fresh water to ensure sensitiveness of the muscles and regular contractions.
5The glass tube is 100 mm. high and 23 mm. in diameter. It is tapered at the bottom and a rubber tube is attached to the narrow end to allow for emptying of the liquid.
to two hours. Under the effect of slight tension it dilates and the needle reaches a horizontal position.
Contractions of the denervated muscle of the leech caused by 50 to 500 δ of acetylcholine hydrochloride (SI) added to 20 cc. of Ringer bath are generally weak, and irregular. The muscle is washed three to four times in 20 cc. of Ringer between each dose of acetylcholine.
Example: Six successive doses of 500 δ of SI cause contractions of unequal extent (tracing No. 1).
For a constant dose of morphine hydrochloride (Mo) added to the Ringer bath one minute before SI, and without washing between Mo and SI, the contraction due to SI depends on the dose of SI.
Example: 50 δ of morphine hydrochloride potentialize the effect of 200 and 300 δ of acetylcholine hydrochloride. 50 δ of morphine hydrochloride do not potentialize the effect of 100 δ of acetylcholine hydrochloride (tracing No. 2).
Although the effect of SI-on the muscle is immediate and particularly effective in the first minute, it continues to register during the second minute. Contractions due to SI added to the bath one minute after Mo must be recorded within two minutes if comparable tracings are to be obtained.
Example: The effect of 200 δ of SI one minute after 100 δ of Mo is regular if recorded for two minutes and irregular if recorded for one minute (tracing No. 3).
Period of contact of morphine hydrochloride: different periods of contact were tried. If Mo is added to the Ringer bath one minute before SI, the effect is greater than if Mo is added half a minute or two minutes before SI.
Example: The effect of 100 δ of morphine hydrochloride added to the Ringer bath one minute before 500 δ of' acetylcholine hydrochloride is regular; added half a minute or two minutes before the acetylcholine the effect is weaker and irregular (tracing No. 4).
Intensity of fixation. The potentialization of the action of acetylcholine by morphine is transitory, since the initial sensitivity to acetylcholine reappears after the organ is washed.
A comparison of the action of morphine with that of eserine on tracings 5 and 6 shows that the latter is eliminated less rapidly than the former (Kahane and Jeanne Lévy, 1939).
Recovery of the leech muscle between two experiments. The time required for the muscle to recover its original tone averages five minutes. If the experiments are carried out (Mo, one minute, SI, two minutes, washing) every five or six minutes, equal doses produce identical contractions. If the time between two experiments is increased, the sensitivity of the muscle diminishes or varies.
Example: The action of 100 δ of Mo on 500 δ of SI is regular if the muscle Mo and SI is activated every five minutes; it is weaker after fifteen minutes (tracing No. 7).
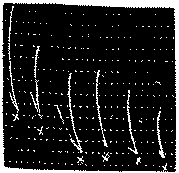
Study of the contractions caused by equal doses of acetylcholine hydrochloride on denervated leech muscle.
In x: 500δ of acetylcholine hydrochloride.

Study of the duration of registering the contraction of the leech muscle caused by acetylcholine hydrochloride added to Ringer bath 1 min. after a dose of morphine hydrochloride.
In x: 200δ of acetylcholine hydrochloride acting for 2 min., 2 min., 1 min., 1 min., 2 min.
In xx: 100δ of morphine hydrochloride.
Study of the potentialization of the action of acetylcholine hydrochloride on denervated leech muscle by a constant dose of morphine hydrochloride.
In x: successive actions of 100δ, 200δ, 300δ, 300δ, 300δ, 200δ, 200δ, 100δ, 100δ of acetylcholine hydrochloride.
In xx: Actions of 50δ of morphine hydrochloride.

Study of the time of contact of morphine hydrochloride.
In x: 500δ of acetylcholine hydrochloride.
In xx: 100δ of morphine hydrochloride acting 1 min., 1 min., 1 min., 1/2 min., 1/2 min., 1 min., 1 min., 2 min. before the acetylcholine hydrochloride.
In L: 4 washings of the organ in a Ringer solution.
Taking into account the results set forth above, I was able to establish a method for the assay of small quantities of morphine. The salts used are acetylcholine hydrochloride at 1/1,000 and morphine hydrochloride at 1/5,000. The dose of acetylcholine hydrochloride (100 to 500 δ), which causes a slight contraction of the muscle (0.5 to 1 cm.), recorded for two minutes, is determined, and the organ is washed with three times 20 cc. of Ringer after each dose of acetylcholine. After waiting for five minutes, 30 to 100 δ of morphine hydrochloride are added to the Ringer bath, sixty seconds before the acetylcholine, without washing; the contraction due to acetylcholine is recorded for two minutes, the organ is washed, and the experiment repeated every five minutes.
(a) For a given dose of acetylcholine, constant doses of morphine produce a constant sensitization, increasing and decreasing doses of morphine a larger or smaller sensitization.
Example: Action of 100, 75, 50, 40 and 30 δ of morphine hydrochloride on 200 δ of acetylcholine hydrochloride (tracing No. 8).
The following results are obtained from the tracing:
|
Morphine dose (in δ per 20 cc.) |
Acetylcholine dose (in δ per 20 cc.) |
Measurement of contraction (0.5 cm. height on the tracing) |
|---|---|---|
| 30 | 200 | 7 |
| 40 | 200 | 8 |
| 50 | 200 | 9 |
| 70 | 200 |
103/4 |
| 100 | 200 |
121/2 |
It is not possible to establish a standard curve for the action of morphine on acetylcholine because each leech has a particular sensitivity to morphine and to acetylcholine, and this may vary during the experiment. When assaying morphine, it is necessary constantly to check the action of the standard solution and to compare only doses producing identical contractions.
(b) Description of the assay of an unknown quantity of morphine hydrochloride in pure solution.
In the presence of a morphine solution of unknown titration, the action of any given dose of that solution is observed; an attempt is then made to find the dose causing the same action as an effective dose of the standard solution, and the action of a larger or smaller dose causing the same action as a larger or smaller dose of the standard solution.
Example: 0.2 cc. of solution A produces a weaker action than 40 δ of morphine hydrochloride; 0.25 cc. and 0.18 cc. produce the same contractions as 40 δ and 30 δ of morphine hydrochloride. The solution to be assayed must therefore contain 160 δ per cc., and that is actually its titration (tracing No. 9).
As in all biological assays, the standard and the sample to be assayed must be administered alternately, and an attempt must be made to find identical potentializations. In these circumstances, the experimental error generally does not exceed 15 per cent.
Oxydimorphine hydrochloride, in doses forty times stronger than the morphine hydrochloride, produces no action whatever on the leech muscle and does not increase the sensitivity of this muscle to acetylcholine.
Example: 4 mg. of oxydimorphine hydrochloride do not increase the action of 200 δ of acetylcholine hydrochloride, whereas 50 δ of morphine hydrochloride potentialize the action of 200 δ of acetylcholine hydrochloride. Moreover, 4 mg. of oxydimorphine do not modify the potentialization of the action of 50 δ of morphine in relation to 200 δ of acetylcholine hydrochloride (tracing No. 10).
The assay of morphine in pure solution, whether or not in the presence of oxydimorphine, may be used to detect morphine in biological liquids and tissues, after extracting the two alkaloids.
(a) Methods of extraction. The liquid of extraction, which should contain the whole of the alkaloids, should also, in their absence, produce no sensitizing effects whatever on the contraction due to acetylcholine hydrochloride.
Proteins must be eliminated from biological liquids and organs. Various experimental procedures have not satisfied me, since the extracts obtained produced an effect on the muscle itself, preventing or increasing the action of the acetylcholine. For these reasons I was obliged to abandon defaecation with ferric sulphate and calcium carbonate, defaecation with calcium hydroxide, extraction with ethyl acetate or benzene in a bicarbonate medium. The methods of extraction set forth below, on the other hand, enabled me to carry out quantitative extractions of morphine and oxydimorphine; they differ according to whether the assay applies to free morphine or to products of the conjugation of morphine produced in the system.
Free alkaloids
Blood, urine and faeces (homogenized with Fontainebleau sand) are treated with seven times their volume or weight of 1/1,000 hydrochloric alcohol, and then filtered. The filtrate is evaporated under a cold air current. The residue, dissolved with N/200 hydrochloric acid, is brought to pH 6.3 with N/100 soda. The dilution of these extracts should be such that the volumes added to the Ringer bath approximate as closely as possible to those of the standard solution.
The organs, homogenized with Fontainebleau sand, with the addition of seven times their weight of 1/1,000 hydrochloric alcohol, are exhausted by boiling for three hours in a reflux condenser, cooled and filtered.
The filtrate is treated as above. It is indispensable to treat the organs under heat, since otherwise the quantity of morphine recovered is 30 per cent less than what was added.
Total alkaloids
The blood and the organs (homogenized with sand) with the addition of seven times their volume or weight of absolute alcohol and half their volume or weight of concentrated hydrochloric acid are exhausted by boiling for three hours in a reflux condenser, cooled and filtered. The filtrate is evaporated under a current of cold air. The residue, dissolved with distilled water, is brought to pH 6.3 with concentrated and dilute soda.
The urine and faeces (pulverized), with the addition of a quarter of their volume or weight of concentrated hydrochloric acid, are placed in sealed ampoules and heated in an autoclave for thirty minutes at 120° C. Upon removal from the autoclave, seven times their weight of absolute alcohol is added to the contents of the ampoules, which are then filtered. The filtrate, evaporated cold, gives a residue which, after being dissolved with distilled water, is brought to pH 6.3 with the aid of concentrated and dilute soda.
Remark 1. I was unable to treat the blood and organs in an autoclave in the presence of concentrated hydrochloric acid because the extracts, in the absence of morphine, sensitized the action of acetylcholine on the muscle. If, however, the extracts are brought to boiling point in the presence of absolute alcohol and concentrated acid, they do not modify the effect of the acetylcholine.
The extracts of blood, organs, urine and faeces, treated under the above conditions, have no effect on the muscle itself and do not modify the contraction due to acetylcholine.
Two cc. of these extracts, prepared from 5 cc. of blood or urine or from 5 g. of organs or faeces may be added to the Ringer bath without producing any effect on the muscle.
Remark 2. I confirmed that the treatment of a solution of morphine hydrochloride with 1/1,000 hydrochloric alcohol (under heat or cold), with absolute alcohol and concentrated hydrochloric acid, or with concentrated hydrochloric acid in the autoclave, does not affect the morphine content.
|
Weight of Mo treated (in mg.) |
Treatment |
Recovered morphine (Weight in mg.) |
|---|---|---|
| 2 |
1 0/00 HCL alcohol - cold |
2 |
| 2 |
1 0/00 HCL alcohol - under heat |
2 |
| 1 |
Pure alcohol plus concentrated HCL - under heat |
1 |
| 1 |
Concentrated HCL, in autoclave |
1 |
(b) Assay of morphine added to a biological medium.
To blood, organs and pulverized faeces a solution of morphine hydrochloride is added; the morphine is extracted by one of the procedures described above, depending upon the case. The assay carried out on the muscle of the leech by using the morphine-acetylcholine potentialization makes it possible to assay the initial morphine with an error not exceeding 10 per cent.
The most concordant results are obtained when morphine is added to blood or urine; some divergencies are noted in connection with organs and faeces.
|
Medium | ||||
|---|---|---|---|---|
|
Mo added mg. |
Treatment |
Percentage morphine recovered | ||
|
Blood |
dog |
1 |
1 0/00 HCL alcohol - cold |
100 |
|
Liver |
rat |
2 |
1 0/00 HCL alcohol - under heat |
100 |
|
Muscle |
rat |
2 |
1 0/00 HCL alcohol - under heat |
90 |
|
Urine |
rat |
5 |
Concentrated HCL - in autoclave |
100 |
|
Faeces |
rat |
3 |
Concentrated HCL - in autoclave |
95 |
(c) Assay of morphine added to biological media in the presence of oxydimorphine hydrochloride. To the homogenized liver of the rat and to the urine of the rat, morphine hydrochloride and oxydimorphine hydrochloride are added.
They are treated under heat with seven times their weight or volume of 1/1,000 hydrochloric alcohol. After cooling and filtration, the filtrates are evaporated, the residues are dissolved with N/200 hydrochloric acid and the solutions are brought to pH 6.3.
The morphine is assayed on the muscle of the leech by morphine-acetylcholine potentialization. It will be seen from the following table that all the morphine which has been added is recovered.
The addition of oxydimorphine hydrochloride does not hinder the assay of morphine in biological media.
|
Medium |
Morphine added, mg. |
Oxydimorphine added mg. |
Percentage of morphine recovered |
|---|---|---|---|
|
7 g. liver |
2 | 2 | 100 |
|
6 cc. urine |
2 | 2 | 100 |
The biological assay of morphine by morphine-acetylcholine potentialization on the denervated muscle of the leech is characteristic of morphine in the presence of oxydimorphine.
If the leech muscles used are sensitive to increasing doses of morphine hydrochloride at close intervals (30, 35, 40, 45, 50 δ), experimental errors can be kept below 10 to 15 per cent by averaging the results obtained on two different muscles.

Leech muscle subjected to action of morphine hydrochloride. Return to initial sensitivity after washing. (After Kahane and Jeanne Lévy, 1939.)
In x: Action of 50δ of acetylcholine hydrochloride.
In xx: Action of 40δ of morphine hydrochloride.
In L: 4 washings of the organ in Ringer solution.
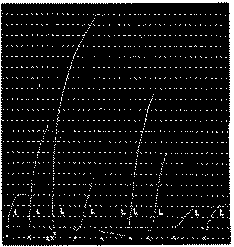
Leech muscle subjected to action of eserine sulphate. Return to initial sensitivity. (After Kahane and Jeanne Lévy, 1939.)
In x: Successive actions of 2δ, 5δ, 0.1 δ, 0.1 δ, 0.1 δ, 0.5 δ, 0.5 δ, 0.5 δ, 0.5 δ of acetylcholine hydrochloride.
In xx: Action of 50δ of eserine sulphate.
In L: 4 washings of the organ in Ringer solution.
If the muscular contractions are regular for identical quantities of morphine hydrochloride, the same muscle may easily be used to test the effect of twenty-five to thirty doses of morphine and acetylcholine, and three to four extracts can be assayed.
The disadvantages of this technique are due to the muscles, which are not always very regular or very sensitive, particularly in summer or in stormy weather. Frequently the sensitivity of the muscle to morphine varies only at doses of 50, 70 and 100 δ, the intermediate doses having no appreciable effects. In these conditions experimental errors may amount to 40 per cent, and the assay must then be repeated.
With a view to the biological assay of small quantities of morphine and oxydimorphine, I endeavoured, in collaboration with Jeanne Lévy (1939), to ascertain the amounts of morphine hydrochloride which would reduce by 20 to 30 per cent the contraction caused by various cholinic esters in the denervated muscle of the leech, the muscle being treated with eserine in the case of the acetic ester but not in the case of the hydrobromic, carbamic and nitric esters.
The results obtained are summarized in the following table:
|
Doses used (in δ) |
Doses of morphine hydrochloride causing reduction of the musuclar contraction by 20 to 30 per cent (in δ) | |
|---|---|---|
|
Hydrochloride of acetic ester |
0.15 - 2 |
50 - 60 |
|
Perchloride of hydrobromic ester |
75 - 175 |
10 - 30 |
|
Perchloride of carbamic ester |
5 - 20 |
40 - 60 |
|
Perchloride of nitric ester |
3 - 10 |
60 - 0 |
The Ringer bath in which the leech muscle is immersed is of 20 cc. capacity.
The contraction caused by the hydrobromic, carbamic and nitric esters is not potentialized by the presence of eserine (Kahane and Jeanne Lévy, 1939), and the effect of the morphine on the denervated muscle, whether or not treated with eserine, is identical. The hydrobromic ester of choline has a reasonably regular contractile effect on the leech muscle and is sensitive to the antagonist effect even of very small doses of morphine. It therefore appeared to be the most suitable substance for use in the systematic study of a method of assay.
The muscle is prepared as above and the Ringer solution used is identical with that described on page 20:
I began by ascertaining the optimum conditions for determining the quantitative extent of this antagonism. These are as follows: the contraction of the leech muscle caused by the hydrobromic ester is recorded for three minutes; the amounts of morphine used must not reduce the contraction of the leech muscle caused by the hydrobromic ester of choline by more than 40 per cent, since in this case the muscle rapidly becomes useless; and the period of contact between the morphine and the bromocholine is sixty seconds, each washing of the organ being followed by a rest period of seven minutes. With a view to accelerating the muscle's recovery of its initial sensitivity, an appreciable quantity of the hydrobromic ester, about two and a half times the normally active amount, should be added to the Ringer bath after each antagonism test.
After the addition of morphine, some muscles never regain their initial sensitivity, this being reduced in certain muscles and increased in others. Such muscles may nevertheless be used again if the initial quantity of the hydrobromic ester still causes neither too violent nor too weak a contraction; in these conditions the percentage reduction of the contractions may be calculated, provided that only reductions of the same order of magnitude are compared.
For a constant dose of hydrobromic ester, equal doses of morphine hydrochloride cause the same degree of antagonism.
Example: Equal doses of morphine (30 δ) used four times in a leech muscle which has decreasing sensitivity to the hydrobromic ester of choline cause more or less equal reductions in muscular contraction: 38, 36, 37, 35, 31 per cent (tracing No. 11).
For a constant dose of hydrobromic ester of choline, the reduction in the contraction of the leech muscle varies with the dose of morphine used.
Example: Increasing doses of morphine hydrochloride - 12.5, 17 and 25 δ - reduce the contractions caused by hydrobromic ester of choline, by 20, 33 and 40 per cent respectively (tracing No. 12).
I was able to work out a technique for assaying small quantities of morphine, the salts used being a 1/2,000 solution of perchloride of hydrobromic ester of choline and a 1/10,000 solution of morphine hydrochloride. The dose of perchloride of hydrobromic ester of choline (75 to 175 δ) causing a muscular contraction for three minutes, shown on the tracing by a curve of 4 to 6 cm. height, is determined. The organ is washed with three times 20 cc. of Ringer after each treatment with ester of choline. After a seven-minute interval, a quantity of morphine hydrochloride capable of causing a 20 to 40 per cent reduction in the initial muscular contraction is added to the bath. Sixty seconds after the morphine is added, the organ not being washed, the initial amount of hydrobromic ester causes a contraction, which is recorded for three minutes. The muscle is then thoroughly washed, and after a seven-minute interval treatment with two and a half times the active dose of hydrobromic ester, with a view to increasing the tonicity of the muscle, causes a contraction recorded for one minute to one minute and a half. It is then confirmed that the initial quantity of hydrobromic ester causes the same contraction as before the antagonism.
For this biological assay it is recommended that the standard substance and the sample under test be administered alternately. The test should be repeated on two or three different muscles and the average results may be subject to errors of not more than 20 to 25 per cent.
Example: 0.30 cc. and 0.35 cc. of the solution to be assayed caused reductions of 23 and 42 per cent respectively in the muscular contraction. Fifty δ of morphine hydrochloride caused a reduction of 27 per cent. The solution to be assayed should contain 161 δ per cc. Its titration was actually 160 δ (tracing No. 13).
The quantitative antagonism manifested by morphine and hydrobromic ester of choline in the denervated muscle of a leech also holds good for oxydimorphine, which, in doses of 10 to 40 δ, exercises like morphine itself an inhibitory effect on the contraction caused by hydrobromic ester of choline in the leech muscle.
Example: Increasing doses of oxydimorphine hydrochloride administered in conjunction with the same active quantity of bromocholine perchloride cause smaller and smaller contractions, and the percentage reductions are identical with those observed with morphine hydrochloride in the same muscles.
|
Oxydimorphine |
Morphine | |||
|---|---|---|---|---|
|
Doses of hydrobromic ester of choline (in δ) |
Doses of oxydi-morphine (in δ) |
Percentage reduction in the muscular contraction |
Dose of morphine (in δ) |
Percentage reduction in the muscular contraction |
| 100 | 25 | 30 |
- |
- |
| 100 | 30 | 36 | 30 | 35 |
| 100 | 40 | 50 | 40 | 48 |
Either morphine or oxydimorphine solutions can therefore be assayed with reference to a standard solution of either of the two alkaloids; but they cannot be differentiated by this method.
This method of assay may be used for determining the morphine and oxydimorphine in biological liquids and tissues, after extraction of the alkaloids, the methods of extraction being identical with those used in the case of the morphine-acetylcholine assay on the muscle of the leech.
Notes:
The maximum quantity of extract which can be added to the Ringer bath is 1 cc. in the case of extracts of blood or urine and 0.50 to 0.75 cc. in the case of extracts of organs or faeces, prepared from 1 to 5 cc. of blood or urine and 1 to 5 g. of organs or faeces. Owing to the absence of alkaloids, larger quantities of extracts would cause a reduction of 10 to 25 per cent in the muscular contractions caused by the hydrobromic ester and this would falsify the assay.
Assay of the two alkaloids after various treatments: Morphine hydrochloride or oxydimorphine, or a mixture of both, treated with absolute alcohol and concentrated hydrochloric acid or with hydrochloric acid in an autoclave, and assayed on the basis of the morphine-hydrobromic ester antagonism test on the muscle of the leech are recovered to the extent of 77 to 100 per cent, as shown by the examples summarized in the following table:
|
Morphine mg. |
Oxydi-morphine mg. |
Treatment |
Percentage recovered |
|---|---|---|---|
| 1 |
|
Alcohol - HCL cold |
90 to 100 |
|
|
2 |
Alcohol - HCL hot |
80 to 100 |
| 1 |
|
Alcohol - HCL hot |
100 |
| 7.5 | 2.5 |
Alcohol - HCL hot |
77 |
|
|
2 |
Alcohol - HCL in autoclave |
100 |
The differences found in the two assays are due to errors attributable to the method of assay.
(a) Assay of morphine and oxydimorphine added to biological media. Homogenized blood, urine, organs and faeces are added to a solution of morphine hydrochloride or oxydimorphine or a mixture of both. The method of extraction may be any of those described above. The alkaloids can be recovered to the extent of 80 to 100 per cent by assay using the morphine-hydrobromic ester antagonism test, as shown in the following table:
|
Medium |
Morphine added mg. |
Oxydi-morphine added mg. |
Treatment |
Percentage recovered |
|---|---|---|---|---|
|
Blood |
10 |
|
Alcohol - HCL cold |
100 |
|
Blood |
5 |
|
Alcohol - HCL hot |
100 |
|
Blood |
|
2 |
Alcohol - HCL cold |
85 |
|
Liver |
2 | 2 |
Alcohol - HCL hot |
100 |
|
Faeces |
4 | 2 |
Concentrated HCL in autoclave |
90 |
|
Urine |
4 |
|
Concentrated HCL in autoclave |
80 |
(b) Assay of morphine and oxydimorphine in biological media of animals injected with morphine.
Example: The liver, homogenized with Fontainebleau sand, of a rat previously injected with morphine hydrochloride is boiled for three hours in seven times its weight of 1/1,000 hydrochloric alcohol. After cooling and filtration the filtrate is evaporated in a current of cold air, the residue dissolved in 5 cc. of N/200 hydrochloric acid, the solution filtered, the filtrate brought to pH 6.3 and the volume of the liquid to 10 cc. The assay by morphine-hydrobromic ester antagonism on the denervated muscle of the leech is performed by using 0.50 cc. of extract, which causes the same reduction of effect as 30 δ of morphine hydrochloride, or 0.25 cc. of the extract and 15 δ of morphine hydrochloride, which also causes the same reduction as 30 δ of morphine (tracing No. 14). It may therefore be concluded that the extract contains 60 δ of morphine hydrochloride or oxydimorphine per cc.

Return of leech muscle to normal between two experiments.
In x: Action of 500δ of acetylcholine hydrochloride.
In xx: Action of 100δ of morphine hydrochloride.

Interval between 3 experiments: 5 min., 5 min., 15 min., 5 min.
Influence of different doses of morphine hydrochloride on the acetylcholinic contraction.
In x: Action of 200δ of acetylcholine hydrochloride.

In xx: Successive actions of 100δ, 100δ, 50δ, 75δ, 50δ, 50δ, 30δ, 50δ, 40δ of morphine hydrochloride.
(Reduced by ?) - Assay of a pure solution of morphine hydrochloride of unknown titration.
In x: Action of 250δ of aeetylcholine hydrochloride.
In xx M: Action of 40δ, 40δ, 40δ, 30δ, 40δ of morphine hydrochloride.
In xx A: Action of 0.2 cc., 0.25 cc., 0.18 cc. of the solution to be assayed.
The extract as a whole contains 600 δ of morphine hydrochloride or oxydimorphine.
(c) Advantages and disadvantages of the technique. The method of assay based on the morphine-hydrobromic ester of choline antagonism makes it possible to assay quantities of either morphine or oxydimorphine of the order of 20 to 50 δ per cc. of solution. If the muscles are sensitive to increasing doses, of 20, 30, 40 and 50 δ, and concordant results are obtained on two different muscles, the experimental errors do not exceed 15 to 25 per cent. Errors of this magnitude are low enough to be admissible in biological assays, particularly in view of the small amount of manipulation necessary for extraction.
Nevertheless, this method of assay is difficult and lengthy. The sensitivity of the denervated leech muscles to hydrobromic ester of choline is frequently irregular at the initial stage of experimentation, and the antagonism test cannot be repeated on the same muscle more than seven or eight times.
The main advantage of this test is that it enables the morphine and oxydimorphine content of an extract to be determined simultaneously.
By simultaneous application of the three biological tests described, I was able in my first experiments to analyse biological extracts both qualitatively and quantitatively in respect of their morphine and oxydimorphine content.
As the table below shows, it is possible by using the three tests to distinguish immediately extracts containing only morphine or oxydimorphine from those containing a mixture of the two alkaloids, if a solution of morphine hydrochloride is taken as standard.
|
Alkaloids contained in the extracts |
Sensitization |
Straub and antagonism test |
|---|---|---|
|
Morphine |
+ |
Quantitative results identical |
|
Oxydimorphine |
- |
Quantitative results different |
|
Morphine + oxydimorphine |
+ |
Quantitative results different |
Having worked out the sensitization test, which had become quantitative instead of qualitative, I abandoned the Straub test. I obtained the following results from these two tests on the muscle of the leech:
|
Alkaloids |
Sensitization and antagonism |
|---|---|
|
Morphine |
Quantitative results identical |
|
Oxydimorphine |
Quantitative results different |
|
Morphine + oxydimorphine |
Quantitative results different |
The extracts containing only oxydimorphine are assayed by the antagonism method, since oxydimorphine in tization test, which is more rapid and more exact. Hereafter I shall call this test B.
The extracts containing only oxydimorphine are assayed by the antagonism method, since oxydimorphine in doses forty times larger than morphine does not sensitize the acetylcholinic contraction of the leech muscle. The antagonism test is called test C in the third chapter. Finally, the extracts containing both alkaloids give different quantitative results in the two tests. In this case, the oxydimorphine content of the extract is the difference between the total alkaloid content obtained by the antagonism method and the morphine content obtained by the sensitization method.
Example: 6 cc. of rat urine, to which 2 mg. of oxydimorphine hydrochloride and 2 mg. of morphine hydrochloride are added, are treated with 1/1,000 hydrochloric alcohol. After filtration, the filtrate is evaporated; the residue is dissolved in 5 cc. of N/200 hydrochloric acid and brought to pH 6.3 and 10 cc.
The assay on the muscle of the leech is carried out by tests B and C. The results are shown in the following table:
|
Morphine and oxydimorphine added | ||||
|---|---|---|---|---|
|
Medium |
Morphine added in mg. |
Oxydi-morphine added in mg. |
Test B (in mg.) |
Test C (in mg.) |
|
6 cc. urine |
2 | 2 | 2 | 4 |
The total alkaloid content is obtained by test C, the morphine content by test B. The solution contains 2 mg. of morphine hydrochloride and 2 mg. of oxydimorphine hydrochloride.
The biological methods employed are, of course, less exact, particularly the antagonism method, than chemical methods; but they entail few losses at the time of the extraction of the alkaloids and are more rapid and more characteristic than the techniques of chemical assay; the latter require numerous manipulations in order to obtain the basic elements in a pure state.
As I have indicated in the introduction to this work, one of the oldest hypotheses attempting to explain the phenomenon of habituation assumes that morphine, in- jected into habituated individuals, does not reach the sensitive cell. The problem thus arises whether the morphine administered can be found in toto in the organs and excretions of normal and habituated rats or whether it undergoes a transformation or an identical or different destruction. In the latter case it is necessary to determine the free and conjugated morphine contents and, if possible, the manner in which the morphine is transformed.
To solve this problem:
I investigated whether morphine changes into oxydimorphine:
In vitro, in the blood of rats, after the addition of morphine,
In vivo, in the blood and tissues of rats injected with morphine;
I ascertained the free and conjugated morphine contents:
After the action, in vitro, of certain tissues (hepatic, cerebral, muscular) on morphine;
In the tissues of habituated and normal rats, after the injection of equal doses of morphine.
I studied the elimination of free and conjugated morphine in the urine and faeces of normal and habituated rats.
It is known that the oxidation of morphine, if carried out under certain conditions, leads to oxydimorphine; Leulier and Drevon (1931) have shown, moreover, that, in vitro, blood can transform morphine hydrochloride into oxydimorphine.
As long ago as 1883 Narme assumed that in habituation there occurs an acceleration of the process which permits the morphine to be partially eliminated in the form of its simplest oxidation product, oxydimorphine.
This hypothesis was adopted by many authors (for bibliography see Jeanne Lévy, 1934), some of whom found oxydimorphine in the tissues of habituated animals, while others were never able to detect it.
Confronted with the divergence of previous results regarding the presence in the organism of oxydimorphine, which is less toxic than morphine administered subcutaneously, I investigated whether, in the habituated animal, morphine is transformed, at least in part, into that alkaloid. The biological assay which I established for oxydimorphine enabled me to answer that question in the negative, and I shall indicate below, by way of example, a certain number of results obtained in respect of:
The action of blood on morphine in vitro.
The morphine and oxydimorphine contents of the blood and organs of normal and habituated rats.
Rat blood, plasma or serum is left in contact with morphine hydrochloride for twenty-four hours at a temperature of 37° C. The alkaloid is extracted, cold, with 1/1,000 hydrochloric alcohol (as described in the preceding chapter, page 23).
The assay is applied to the denervated leech muscle, on the basis of the sensitizing action produced by morphine in connexion with acetylcholine (test B) and of the antagonism produced by morphine and oxydimorphine in connexion with bromocholine (test C).
Under these conditions 25 to 50 per cent of the morphine added is not recovered, whatever assay is used.7
The following are some of the figures obtained:
|
Percentage of morphine recovered | |||||
|---|---|---|---|---|---|
|
Quantity of blood (in cc.) |
Quantity of plasma (in cc.) |
Quantity of serum (in cc.) |
Morphine added (mg.) |
Test B |
Test C |
| 1 |
|
|
1 | 70 | 60 |
| 2 |
|
|
4 | 50 | 50 |
| 4 |
|
|
1 | 70 | 75 |
|
|
2 |
|
2 | 50 | 50 |
|
|
|
1 | 2 | 50 | 50 |
If blood, plasma or serum, is treated with 1/1,000 hydrochloric alcohol immediately after the addition of morphine hydrochloride, the whole of the morphine added is recovered. On the other hand, 25 to 50 per cent of the morphine is lost if the blood, plasma or serum, to which morphine hydrochloride has been added, is left in contact for twenty-four hours at various temperatures: 0°, 20°, 37°, 42° and 60° C. Some of the figures obtained follow:
|
Percentage of morphine recovered | |||||
|---|---|---|---|---|---|
|
Quantity of blood (in cc.) |
Quantity of plasma (in cc.) |
Morphine added (mg.) |
Temperature |
Test B |
Test C |
| 2 |
|
2 | 0 | 60 | 55 |
| 4 |
|
2 | 20 | 70 | 60 |
| 2 |
|
4 | 37 | 75 | 70 |
|
|
2 | 2 | 42 | 50 | 60 |
|
|
2 | 2 | 60 | 65 | 60 |
Judging from the experiments, as much as 50 per cent of the original quantity of morphine may be lost, regardless of the temperature at which the blood and morphine are left. This loss is assuredly not caused by an enzymatic phenomenon, since it does not depend upon the temperature and also occurs at 60° C. It may be assumed that there occurs either a conjugation phenomenon or an adsorption of morphine on the proteins.
7The blood, serum or plasma of man, dog, rabbit, guinea-pig, mouse, has no effect on morphine since the latter is entirely recovered by means of the two tests. This work has not been published.

Influence of oxydimorphine hydrochloride on the acetylcholinic contraction.
In x: Action of 200δ of acetylcholine hydrochloride.
In xx: Action of 50δ of morphine hydrochloride.
In xxx: Action of 4 mg. of oxydimorphine hydrochloride.
In x: Action of 4 mg. of oxydimorphine hydrochloride+50δ of morphine hydrochloride.

(Reduced by ?) - Influence of equal doses of morphine hydrochloride on the contracting action of hydrobromic ester of choline on a leech muscle of decreasing sensitivity.
In x: Addition of 175δ of hydrobromic ester of choline in a 20 cc. Ringer bath.
In xx: Addition of 30δ of morphine hydrochloride.

(Reduced by ?) - Quantitative antagonism of hydrobromic ester of choline and of morphine on denervated leech muscle.
In x: Addition of 150δ of perchlorate of hydrobromic ester of choline in 20 cc. Ringer bath.
In xx: Addition of 350δ of perchloride of hydrobromic ester of choline.
In xxx: Addition of 12.5δ, 17δ, 25δ of morphine hydrochloride.
(α) Conjugation phenomenon
To eliminate or confirm the possibility of a conjugation phenomenon, blood and morphine, after a twenty-four hour contact, in an oven at 37° C, are treated with seven times their volume of absolute alcohol and half their volume of concentrated hydrochloric acid. The mixture is boiled for three hours, cooled and filtered; the filtrate is evaporated, dissolved with 5 cc. of distilled water and brought to pH 6.3. The assay is carried out by means of test B. The content of morphine recovered should, if a conjugation phenomenon occurs, be greater than in the cold extraction with hydrochloric alcohol. As will be seen later, the total morphine is assayed in the first case and the free morphine in the second case.
The following table shows that the percentages of morphine recovered are identical, whatever method of extraction is used. There is therefore no conjugation phenomenon.
|
Percentage of morphine recovered | |||
|---|---|---|---|
|
Quantities of blood (in cc.) |
Morphine added (mg.) |
Cold extraction |
Hot extraction |
| 1 | 2 | 67 | 67 |
| 2 | 2 | 67 | 75 |
| 4 | 2 | 50 | 50 |
| 2 | 4 | 70 | 75 |
(β) Adsorption phenomenon
One or two cc. of rat blood or serum are boiled for ten minutes in a water bath. To the coagulate, consisting of minute particles, are added 2 mg. of morphine hydrochloride and it is then heated for twenty-four hours in an oven at 37° C. The morphine is extracted cold with 1/1,000 hydrochloric alcohol and the assay is made by means of test B. Under those conditions, the losses of morphine are identical with those observed when the blood is maintained at different temperatures.
|
Quantity of Blood (cc.) |
Quantity of Serum (cc.) |
Morphine added mg. |
Percentage of morphine recovered |
|---|---|---|---|
| 1 |
|
2 | 63 |
| 2 |
|
2 | 55 |
|
|
2 | 2 | 65 |
There must thus occur an adsorption of morphine on to the proteins of rat blood.
Habituated rats
I wished to determine whether the same phenomenon of adsorption occurred with the blood of habituated animals.
(α) Morphine hydrochloride is added to the blood of rats habituated for seven weeks (group III) ; after twenty-four hours contact in an oven at 37° C., the same percentage of morphine is recovered as in the experiments made with the blood of normal animals.
The following are some of the results obtained:
|
Percentage of morphine recovered | |||
|---|---|---|---|
|
Quantities of blood (in cc.) |
Morphine added (in mg.) |
Test B |
Test C |
| 1 | 1 | 60 | 60 |
| 2 | 1 | 50 | 50 |
| 4 | 1 | 60 | 55 |
(β) The blood or serum of habituated rats is placed in a boiling water bath for ten minutes; the coagulate consisting of fine particles with an admixture of 2 mg. of morphine hydrochloride is maintained for twenty-four hours at 37° C. The morphine is extracted with cold hydrochloric alcohol. The percentages of morphine recovered are similar to those obtained by adding morphine to the coagulate of the blood or serum of normal rats. In the case of the blood of an habituated animal, adsorption of morphine on to the protein must occur.
Summary: Experiments showed that part of the morphine is adsorbed on to the proteins in the rat's blood; hence, it is not possible to recover all the morphine in the blood of that animal, whether normal or morphinized. With regard to the fraction of alkaloids not adsorbed on to the proteins, it may be taken that the blood of a normal or habituated rat does not convert morphine into oxydimorphine, since the results of the two tests are similar with both groups of animals.
After injection of 1 mg. of morphine hydrochloride per g. into normal and habituated rats, the alkaloid contents of the blood and tissue extracts are comparable if the assay is carried out by means of tests B or C. On no occasion did I recover oxydimorphine. The following are some of the results obtained:
|
Condition of rats Biological media |
Alkaloid content expressed in cc. or g. | ||
|---|---|---|---|
|
Test B |
Test C | ||
|
Normal |
Blood |
120 | 100 |
|
|
Muscle |
50 | 55 |
|
Habituated |
Blood |
200 | 200 |
|
|
Liver |
125 | 125 |
|
|
Muscle |
300 | 280 |
Summary: Oxydimorphine was never detected in the blood and tissue of normal and habituated rats after injection of morphine. In the subsequent stages of this work, therefore, I used only test B for the assay of morphine, as being quicker and more accurate.
The experiments which have been made in this field and described in literature are definitely contradictory and we shall only deal with the most recent.[9]
With regard to the disappearance of morphine Ikeshima (1934) finds less morphine in the blood and tissues of habituated dogs than in those of normal dogs.
On the basis of his experiments on the monkey, Yokota (1935) does not agree that the ability of the tissues to destroy morphine increases during habituation. Kuwahara (1937) observes no difference in the quantity of morphine present in the blood and liver of rabbits, whether tolerant or not, one hour after the subcutaneous injection of 200 mg. of morphine sulphate per kg. Hinohara (1937) fails to demonstrate that morphine is destroyed when allowed to incubate, in vitro, with the muscle and blood of a rabbit, whether tolerant or normal. Zwikker and Steenhauer (1942) detected no morphine in the blood, stomachal and intestinal content of persons fatally poisoned with morphine administered subcutaneously. Ko (1937), after steeping the liver of both tolerant and non-tolerant rabbits, found in the resulting liquid from 70 to 80 per cent and from 53 to 67 per cent respectively of the initial quantity of morphine; from this he concluded that habituation increased the liver's capacity to destroy morphine.
With regard to conjugation, a very important piece of work carried out by Bernheim and Bernheim (1944, 1945) consisting in placing morphine in contact, in vitro, with thin sections of dog's, cat's, guinea-pig's and rat's liver, confirmed the experiments carried out by Simonnet (1937) and Inoue (1940). The latter recovered only 50 per cent of the morphine added to thin sections of liver. Bernheim and Bernheim state that the morphine is assayed in the free state and that the remainder is in conjugation: after treating the trichloracetic filtrate with normal hydrochloric acid in an autoclave for thirty minutes, they recovered all the morphine added. From this they conclude that a glucuronate is probably formed, since the conjugated derivative exists in the absence of sulphate ions (Bernheim and Bernheim, 1945). They also point out that this does not occur with brain sections. Unfortunately, Bernheim and Bernheim did not experiment with sections of the organs of tolerant animals.
Gross (1942) demonstrated in the dog that adulteration of the liver with carbon tetrachloride affects the conjugation of morphine. The total quantity of morphine found in the urine remains unchanged but the quantity of free morphine increases and the readily hydrolyzable fraction of the conjugated morphine is reduced in proportion.
Quite recently, Fawaz (1948) experimenting with sections of the liver of tolerant and normal rats found somewhat less morphine conjugated with the liver sections of tolerant rats. Moreover, by the total or partial hepatectomy of normal and tolerant rats, he demonstrated that both groups of rats survive the same 50 per cent lethal dose as animals not operated upon; from this he concludes, somewhat hastily perhaps, that tolerance is not dependent upon the metabolism of morphine in the liver, the spleen or the gastro-intestinal tract.
With regard to accumulation, experiments conducted by Oberst (1942) on morphine addicts showed the presence of free morphine in the liver and perspiration, of conjugated morphine in the bile, and of both free and conjugated morphine in the gastric content, whereas no morphine was found in the blood and saliva. He was himself surprised at this latter result, inasmuch as a large number of experimenters have detected morphine in the blood and saliva, and as the presence of morphine in the saliva is used as a test to detect the doping of race-horses. The chemical method employed is perhaps inadequate to assay the small quantities of morphine present in the blood and saliva of habituated individuals.
Teruuchi and Kay (1927) consider that, in the rabbit, morphine accumulates in the muscles and stomachal content in the case of acute morphinism, but is destroyed in the course of chronic morphinism; three hours after injecting a large quantity of morphine into a normal rabbit, they recovered 90 per cent of the drug and, three hours after the final injection, they recovered 16.36 per cent of the total quantity of morphine injected into a rabbit over a period of one month. In the first case, the muscles contained 33 per cent and the gastric content 18 per cent of the morphine injected, and in the second case, the muscles contained 3.6 per cent and the gastric content 0.60 per cent of the total quantity of morphine injected. On the other hand, after the last dose administered to a chronically morphinized rabbit, 50 per cent of the morphine was found in the muscles and 8 per cent in the stomach; hence, the normal animal differs only slightly from one habituated to the drug, since the morphine does not accumulate for long in the organism.
(1) Liver. The first experiments were made with homogenized rat's liver which was placedfor twenty-four hours in contact at a temperature of 37°C., with a solution of morphine hydrochloride in a physiological serum. When I failed to note any change in the morphine content of the homogenized tissue I proceeded, in the light of the experiments carried out by Bernheim and Bernheim, to add morphine to thin sections of liver and I noted results similar to those described by those authors.
(a) Homogenized hepatic tissue. 1, 2, 3 or 4 g. of rat's liver are homogenized with Fontainebleau sand and placed in contact for twenty-four hours, at a temperature of 37°C., with 1, 2, 3 or 10 mg. of 1 per cent morphine hydrochloride in a physiological serum. The tissue is then boiled for three hours in a reflux condenser with seven times its weight of 1/1,000 hydrochloric alcohol. The mixture is allowed to cool and filtered, the filtrate being evaporated in cold air. The residue is dissolved in N/200 hydrochloric acid and brought to pH 6.3. The morphine is then assayed by test B, 90 to 100 per cent of the initial quantity of morphine being recovered, by this method. The following table illustrates some of the results obtained with the livers of normal rats and of seven-week habituated rats (group III). The two groups of rats give identical results.
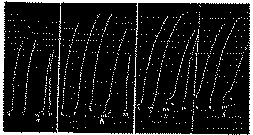
(Reduced by ?)-Determination of the morphine content of a solution containing a morphine salt.
In x: Addition of 150δ of perchloride of hydrobromic ester of choline to the 20 cc. Ringer bath.
In xx: Addition of 350δ of perchloride of hydrobromic ester of choline.
In xxx: Successive addition of 20δ of morphine hydrochloride, of 0.30 cc. or solution A, of 50δ of morphine hydrochloride and of 0.35 cc. of solution A.
0.35 cc. of solution A produces a slightly weaker inhibiting effect than 50δ of morphine hydrochloride; according to this assay, this solution therefore contains 161δ per cc. (its true titration is 160δ per cc.).
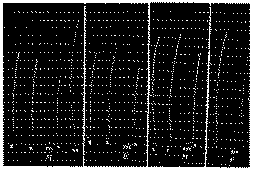
(2/3 reduction)-Determination of the aggregate, morphine and oxydimorphine content of a biological extract.
In x: Addition of 125δ of hydrobromic ester of choline in 20 cc. Ringer bath.
In xx: Addition of 400δ of hydrobromic ester of choline.
In xxx: Successive addition of 30δ of morphine hydrochloride, 0.50 cc. of rat's liver (E), of 30δ of morphine hydrochloride, of 0.25 cc. of extract (E), to which has been added 15δ of morphine hydrochloride. Extract E contains 60δ of morphine hydrochloride or oxydimorphine per cc.
|
Condition of rats |
Weight of liver (in mg.) |
Quantity of morphine added (in mg.) |
Percentage of morphine recovered |
|---|---|---|---|
|
Normal |
1 | 1 | 90 |
|
|
2 | 4 | 100 |
|
|
4 | 2 | 90 |
|
|
4.8 | 10 | 100 |
|
Habituated |
2 | 1 | 85 |
|
|
4 | 3 | 100 |
|
|
5 | 10 | 100 |
(b) Thin sections of liver. Thin sections of rat's liver are placed in an Erlenmeyer flask of 50 cc. capacity containing 4 cc. of Tyrode solution[10] without glucose. Morphine hydrochloride in a 1 per cent solution is then added to the mixture which is placed in a water-bath at a temperature of 37°C. and air is passed through the flask. After five hours the liver sections are removed, dried and weighed; the liquid is heated with either four times its volume of 1/1,000 hydrochloric alcohol or four times its volume of absolute alcohol and half its volume of concentrated hydrochloric acid. After the mixture has been cooled and filtered and the filtrate evaporated, the residue is dissolved in N/200 hydrochloric acid or distilled water and the liquid brought to pH 6.3. When sections of liver of normal rats or of group III habituated rats are placed in contact with morphine, the quantities of morphine recovered are respectively 50 to 63 per cent and 50 to 70 per cent of the initial quantity of the drug if the extraction is carried out with 1/1,000 hydrochloric alcohol, and 100 per cent for both groups of rats if the extraction is carried out with absolute alcohol and concentrated hydrochloric acid.
|
Percentage of morphine recovered | |||
|---|---|---|---|
|
State of animals |
Weight of sections (mg.) |
Alcohol HCl 1/1000 |
Alcohol+conc. HCl |
|
Normal |
92.5 | 62 | 100 |
|
|
234 | 50 | 100 |
|
|
318 | 60 | 100 |
|
Habituated |
250 | 70 | 100 |
|
|
250 | 60 | 100 |
Although I extracted the morphine differently from Bernheim and Bernheim,[11] my results were comparable to theirs. After extraction with 1/1,000 hydrochloric alcohol, the morphine measured represents the fraction of free morphine and possibly also a fraction of the conjugated morphine which is very easily hydrolyzed.
In extracting morphine with the aid of alcohol and concentrated acid, the total morphine is measured (free morphine + conjugated morphine): there is no need to autoclave to decompose the conjugated compound as Bernheim and Bernheim state, since the method of extraction utilized permits the recovery of 100 per cent of the original morphine.
Under aerobiosis, the thin sections of liver convert part of the morphine into a conjugated derivative, which I did not isolate.
(c) Perfusion of rat's liver by a solution of morphine hydrochloride. A fine syringe is inserted through the portal vein into the liver of a rat killed by decapitation with the abdomen and thorax exposed and Tyrode solution at 37° C. is introduced. The liver is carefully removed and placed in a flask kept at 37° C. by means of a double wall. The Tyrode solution, under pressure and at a temperature of 37° C. enters through the portal vein and flows out through the super-hepatic vein at a controlled rate of 100 drops a minute. The liver is perfused with the solution until the liquid discharged becomes colourless. The organ is then perfused with 30 cc. of Tyrode containing 1 per cent of morphine hydrochloride. The liquid discharged is collected and passed back into the liver. On each occasion the first 5 cc. are drawn off.
The morphine is assayed immediately by means of test B on the perfusion liquid, after verifying that, in the absence of morphine, the liquid has no action on the acetylcholinic contraction of the leech muscle.
The liver of a normal or habituated rat of group III is perfused six times with the same liquid. Each drawing off contains 1 mg. of morphine per cc. so that the perfusion of the liver of a normal or habituated rat does not result in any decrease in the morphine rate of the perfusion liquid. Simonnet (1937) obtained similar results in perfusing the liver of a normal dog or rabbit.
(2) Brain. Thin sections of the brain of normal and habituated rats are placed in an Erlenmeyer containing 4 cc. of Tyrode and 2 mg. of morphine hydrochloride. The flasks are kept in a water bath at 37° C. for five hours and the contents exposed to the air. The sections are then removed, dried and weighed. The liquid is treated, hot, for three hours, either with four times its volume of 1/1,000 hydrochloric alcohol or with four times its volume of absolute alcohol and half its volume of concentrated hydrochloric acid. After the filtrate has been cooled, filtered and evaporated, the residue is dissolved with N/200 hydrochloric acid or with distilled water and the liquid is brought to pH 6.3. The quantity, of morphine recovered is 72 to 82 per cent and 100 per cent respectively of the initial quantity, according to whether it is extracted with hydrochloric alcohol or with alcohol and concentrated acid. The results obtained are identical for brain sections of both normal and habituated rats.
|
Percentages of morphine recovered | |||
|---|---|---|---|
|
State of rate |
Weight of sections mg. |
Hydrochloric alcohol 1/1000 |
Alcohol plus conc. hydrochloric acid |
|
Normal |
155 | 80 | 100 |
|
|
138 | 72 | 100 |
|
Habituated |
154 | 70 | 100 |
|
|
94 | 72 | 100 |
These results do not agree with those of Bernheim and Bernheim, who did not observe any conjugation of morphine in brain sections under aerobiosis.
According to the experiments described above, the conjugation of morphine is less in the brain sections than in the liver sections.
(3) Muscle. Thin sections of the muscles of normal rats do not conjugate morphine under aerobiosis in the presence of morphine hydrochloride at 37°C., because the latter is entirely recovered in the Tyrode containing the tissue sections, after treatment with 1/1,000 hydrochloric alcohol. The muscle sections of habituated rats in groups II and III also have no effect on the morphine unless the conjugated derivative formed in the muscle is very readily hydrolyzable.
Oberst (1940), Gross and Thompson (1940) consider that injected morphine is excreted in three forms one free and two conjugated; one of these compounds is hydrolyzed at pH 1 in sixty minutes at 100° C, and the other, which is more stable, is only hydrolyzed with a strong acid in the autoclave. According to Oberst (1940), Gross and Thompson (1940), Bernheim and Bernheim (1945), morphine should conjugate with glycuronic acid. Although they did not succeed in producing the conjugated derivative or in synthesizing it, these authors described the products into which it decomposes. I confined myself to assaying the non-conjugated morphine and the total morphine after hydrolyzing the conjugated derivatives.
I made a point of comparing the free and conjugated morphine content of the blood and tissues of normal and habituated rats after the subcutaneous injection of 1 mg. of morphine hydrochloride per g. or 400 per cent of the 50 per cent lethal dose for normal rats and the total morphine content of the blood and tissues of normal and habituated rats after the subcutaneous injection of 0.200 mg. of morphine hydrochloride, or 80 per cent of the 50 per cent lethal dose for normal rats. More than 1 mg. per g. cannot be administered to normal rats, if they are to be kept alive for sixty minutes, or more than 0.200 mg. per g. if they are to survive for at least twenty-four hours.
The animals are given no food during the twenty-four hours before death. The normal rats are between three and four months old and weigh from 90 to 130 g. The habituated rats belong to group III and weigh from 80 to 120 g. They are given 0.500 mg. of morphine hydrochloride per g. the night before the experiment.
After the carotid has been cut, the blood is collected on sodium fluoride. The animal is decapitated; the liver, kidneys, brain, stomach, spleen, intestine and hind-leg muscles are removed and homogenized with Fontainebleau sand. The amount of blood collected is between 2 and 4 cc. The morphine is assayed in all the organs examined. The liver weighs 4 to 6 g., the kidneys 0.8-1.2 g., the brain 1.3 to 1.7 g., the stomach 1 to 5 g., the spleen 0.200 to 0.800 g., and the intestine 6-8 g. The two hind-leg muscles weigh from 3.4 to 4 g. after the removal of skin and bone.
The blood and tissues are treated differently, as described in the second chapter, according to whether it is desired to assay the total morphine or the free morphine.
The assays are made on a leech muscle by means of test B. The results are expressed in morphine hydrochloride.
(1) Free morphine content of the different tissues and biological liquids after subcutaneous injection of 1 mg. of morphine hydrochloride in rats. The strength of the solution of morphine hydrochloride used is 8 per cent. It is injected subcutaneously in one flank. In normal animals drowsiness appears fifteen minutes after the injection; these animals are killed fifteen, twenty, thirty, forty or sixty minutes after the injection.
The free morphine is extracted with 1/1,000 hydrochloric alcohol, cold from the blood and under heat from the tissues.
The averagel2free morphine content of the blood shows little variation between normal and habituated rats (Table I). It is greater in the tissues of habituated animals than of normal animals, but free morphine does not accumulate in the muscles in either case.
(2) Total morphine content of the various tissues and biological liquids of normal and habituated rats after subcutaneous injection of 1 mg. of morphine hydrochloride per g. The animals are killed twenty and sixty minutes after the injection. The total morphine in the blood and tissues is extracted by boiling for three hours with seven times their volume or weight of absolute alcohol and half their volume or weight of concentrated hydrochloric acid (D.I.19).
I used this method of hydrolysis in preference to autoclaving in the presence of concentrated hydrochloric acid. The results obtained after autoclaving potentialize the acetylcholinic contraction of the denervated leech muscle, even in the absence of morphine.
The morphine is undoubtedly released in its conjugated form, since:
(α) The total morphine content is identical in the urine and faeces[13] of the habituated rat both after this treatment and after autoclaving (see experiment below).
|
Morphine content in mg. per cc. or per g | ||
|---|---|---|
|
Alcohol (HCl) |
HCl autoclave | |
|
Urine |
0.50 | 0.50 |
|
Faeces |
0.70 | 0.65 |
(β) All the morphine is recovered in the Tyrode in which the liver and brain sections have been placed (when treated with pure alcohol and concentrated hydrochloric acid) and autoclaving is unnecessary (see page 34).
A comparison of the average[14] total, free and conjugated morphine content of the blood and tissues of normal and tolerant animals killed twenty and sixty minutes after subcutaneous injection of 1 mg. of morphine hydrochloride per g. (Table II) permits of the following conclusions:
The blood of the habituated rat contains more total and conjugated morphine twenty minutes after the injection of morphine than the blood of the normal rat. On the other hand, the total and conjugated morphine contents are identical sixty minutes after the injection.
The total and conjugated morphine contents of the liver and kidneys are identical for the two groups of rats. The brain of the normal rat contains more total and conjugated morphine sixty minutes after the injection than that of the habituated rat. The contrary phenomenon is observed in the muscles.
The spleen of the normal rat contains no morphine. In the habituated rat, however, the total morphine content is relatively high both in free and conjugated morphine.
The stomach and intestine of the normal rat contain no free morphine; in the habituated rat, on the other hand, the greater part of the morphine found in the organs is free morphine. The total morphine content of the stomach is similar in both groups of animals; it is higher in the case of the intestine of the habituated rat.
The morphine contents of the blood and tissues of the normal and habituated rat after the injection of 1 mg. of morphine hydrochloride per g. may be computed on' the basis of the following average figures for a rat of 100 g.: blood-8 cc., liver-5 g., kidney-1 g., brain-1.5 g., spleen-0.500 g., stomach-3 g., intestine-7 g., muscles-10 g. (muscles of the hind and forelegs and lumber muscles).
Twenty minutes after the injection of 1 mg. per g., the percentage of total morphine is greater in the habituated than in the normal rat, the difference being due to the conjugated morphine contents.
Sixty minutes after the injection, the percentages in total morphine are nearly identical for both groups of animals (Table III).
It should be emphasized that resorption is still incomplete sixty minutes after the injection: in the area where the injection was made, 20 to 30 mg. of morphine can be recovered in normal as well as in habituated rats.
(3) Total morphine content of various tissues and biological liquids following subcutaneous injection of 0.200 mg. of morphine hydrochloride per g. in normal and in habituated rats. The injection is made subcutaneously into one of the flanks, with a 4 per cent morphine hydrochloride solution.
The animals are killed twenty minutes and twenty-four hours after the injection.
The total morphine is extracted with absolute alcohol and concentrated hydrochloric acid, boiling for three hours.
The average[15] total morphine contents of the blood and tissues are similar for both groups of rats, except in the intestines of habituated rats which contain a considerable quantity (Table IV). The explanation lies in the quantity of morphine contained in the faeces, as will be seen in the part three of this chapter. ·
In operational conditions, resorption is complete after the injection in the area of insertion of the needle.
Twenty minutes after injection, the total morphine contents, calculated as above, are greater in habituated than in normal rats, but they are of the same order of magnitude after twenty-four hours (Table V).
|
Normal rats (in mg. per cc., resp. g.) |
Habituated rats (in mg. per cc., resp. g.) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
|
Time (min.) between injection and death |
Blood |
Liver |
Kidneys |
Brain |
Muscles |
Blood |
Liver |
Kidneys |
Brain |
Muscles |
| 15 | 0 | 0 | 0.06 | 0 | 0 | 0.10 | 0.10 | 0.30 | 0.20 | 0.03 |
| 20 | 0.13 | 0 | 0.10 | 0 | 0.03 | 0.10 | 0 | 0.45 | 0 | 0.06 |
| 30 | 0.11 | 0 | 0.25 | 0.12 | 0.05 | 0.10 | 0.03 | 0.20 | 0 | 0.15 |
| 40 | 0.10 | 0.05 | 0.35 | 0.19 | 0.07 | 0.25 | 0.02 | 0.25 | 0 | 0.30 |
| 60 | 0 | 0 | 0.25 | 0 | 0.10 | 0.10 | 0.03 | 0.15 | 0.15 | 0.15 |
|
Time (min.) between injection and death |
Blood mg./cm. |
Liver in mg. per g. |
Kidneys in mg. per g. |
Brain in mg. per g. |
Spleen in mg. per g. |
Stomach in mg. per g. |
Intestine in mg. per g. |
Muscles in mg. per g. | |
|---|---|---|---|---|---|---|---|---|---|
|
|
|
|
|
TOTAL |
MORPHINE |
|
|
|
|
|
Normal |
20 | 0.13 | 0.22 | 0.40 | 0.32 | 0 | 0.20 | 0.12 | 0.13 |
|
|
60 | 0.70 | 0.40 | 0.70 | 0.73 | 0 | 0.22 | 0.30 | 0.08 |
|
Habituated |
20 | 0.50 | 0.33 | 0.75 | 0.27 | 0.42 | 0.23 | 0.42 | 0.28 |
|
|
60 | 0.60 | 0.33 | 0.35 | 0.35 | 1.23 | 0.25 | 0.50 | 0.31 |
|
|
|
|
|
FREE |
MORPHINE |
|
|
|
|
|
Normal |
20 | 0.13 | 0 | 0.10 | 0 | 0 | 0 | 0 | 0.03 |
|
|
60 | 0 | 0 | 0.25 | 0 | 0 | 0 | 0 | 0.08 |
|
Habituated |
20 | 0.10 | 0 | 0.45 | 0 | 0.20 | 0.18 | 0.32 | 0.06 |
|
|
60 | 0.10 | 0.03 | 0.15 | 0.15 | 0.50 | 0.16 | 0.40 | 0.15 |
|
|
|
|
|
CONJUGATED |
MORPHINE |
|
|
|
|
|
Normal |
20 | 0 | 0.22 | 0.30 | 0.32 | 0 | 0.20 | 0.12 | 0.10 |
|
|
60 | 0.70 | 0.40 | 0.45 | 0.73 | 0 | 0.22 | 0.30 | 0 |
|
Habituated |
20 | 0.40 | 0.33 | 0.30 | 0.27 | 0.22 | 0.05 | 0.10 | 0.22 |
|
|
60 | 0.50 | 0.30 | 0.20 | 0.20 | 0.73 | 0.09 | 0.10 | 0.16 |
|
Normal rat |
Habituated rat | |||||
|---|---|---|---|---|---|---|
|
Killed twenty min after injection |
Free Mo. in mg. |
Conj. Mo. in mg. |
Total Mo. in mg. |
Free Mo. in mg. |
Conj. Mo. in mg. |
Total Mo. in mg. |
|
Blood |
2.2 | 0 | 2.2 | 0.80 | 3.20 | 4 |
|
Liver |
0 | 1.10 | 1.10 | 0 | 1.65 | 1.65 |
|
Kidneys |
0.10 | 0.30 | 0.40 | 0.45 | 0.30 | 0.75 |
|
Brain |
0 | 0.48 | 0.48 | 0.10 | 0.11 | 0.21 |
|
Spleen |
0 | 0 | 0 | 0.10 | 0.10 | 0.20 |
|
Stomach |
0 | 0.60 | 0.60 | 0.54 | 0.15 | 0.69 |
|
Intestine |
0 | 0.84 | 0.84 | 2.24 | 0.70 | 2.94 |
|
Muscles |
0.30 | 1 | 1.30 | 0.60 | 2.20 | 2.80 |
|
TOTALS |
2.60 | 4.32 | 6.92 | 4.83 | 8.41 | 13.24 |
|
Per cent |
2.6 | 4.3 | 6.9 | 4.8 | 8.4 | 13.2 |
|
Killed after sixty minutes |
|
|
|
|
|
|
|
Blood |
0 | 5.60 | 5.60 | 0.80 | 4 | 4.80 |
|
Liver |
0 | 2 | 2 | 0.15 | 1.50 | 1.65 |
|
Kidneys |
0.25 | 0.45 | 0.70 | 0.15 | 0.20 | 0.35 |
|
Brain |
0 | 1.10 | 1.10 | 0.22 | 0.30 | 0.52 |
|
Spleen |
0 | 0 | 0 | 0.25 | 0.35 | 0.60 |
|
Stomach |
0 | 0.66 | 0.66 | 0.48 | 0.27 | 0.75 |
|
Intestine |
0 | 2.10 | 2.10 | 2.80 | 0.70 | 3.50 |
|
Muscles |
0.80 | 0.80 | 0.80 | 1.50 | 1.60 | 3.10 |
|
TOTALS |
1.05 | 11.91 | 12.96 | 6.35 | 8.92 | 15.27 |
|
Per cent of morphine recovered |
1 | 11.9 | 12.9 | 6.3 | 8.9 | 15.2 |
|
Time (min.) between injection and death |
Blood mg. per cc. |
Liver in mg. per g. |
Kidneys in mg. per g. |
Brain in mg. per g. |
Spleen in mg. per g. |
Stomach in mg. per g. |
Intestine in mg. per g. |
Muscles in mg. per g. | |
|---|---|---|---|---|---|---|---|---|---|
|
Normal |
20 min. |
0.20 | 0.20 | 0.31 | 0.30 | 0 | 0 | 0 | 0.10 |
|
|
24 h. |
0 | 0.33 | 0.08 | 0.60 | 0 | 0 | 0.08 | 0.16 |
|
Habituated |
20 min. |
0.06 | 0.29 | 0.28 | 0.25 | 0 | 0 | 0.38 | 0.12 |
|
|
24 h. |
0 | 0.50 | 0.18 | 0.35 | 0 | 0 | 0.35 | 0 |
|
Normal Rat |
Habituated Rat | |||
|---|---|---|---|---|
|
After 20 min. Total Mo. in mg. |
After 24 h. Total Mo. in mg. |
After 20 min. Total Mo. in mg. |
After 24 h. Total Mo. in mg. | |
|
Blood |
1.60 | 0 | 0.48 | 0 |
|
Liver |
1.00 | 1.65 | 1.45 | 2.50 |
|
Kidneys |
0.31 | 0.08 | 0.28 | 0.18 |
|
Brain |
0.45 | 0.90 | 0.38 | 0.52 |
|
Spleen |
0 | 0 | 0 | 0 |
|
Stomach |
0 | 0 | 0 | 0 |
|
Intestine |
0 | 0.56 | 2.66 | 2.45 |
|
Muscles |
1 | 1.60 | 1.20 | 0 |
|
TOTAL |
4.36 | 4.79 | 6.45 | 5.65 |
|
Percentage of morphine recovered |
21.5 | 24 | 32 | 28 |
I found:
That liver sections of normal and habituated rats conjugate 37 to 50 per cent of the morphine added;
That brain sections of normal and habituated rats conjugate 20 to 30 per cent of the morphine added;
That muscle sections either do not conjugate morphine, or form a particular derivative which is very easily hydrolized;
That the conjugated morphine is recovered from the liver, kidneys, brain, stomach, intestine, muscles of normal and habituated rats and from the spleen of habituated rats after the injection of 1 mg. per g.;
That the percentages of total morphine in the blood and tissues examined after administration of 0.200 mg. and of 1 mg. of morphine hydrochloride per g. are higher, twenty minutes after injection, for the habituated rat (32 and 13.4 per cent respectively) than for the normal rat (21.5 and 6.9 per cent respectively) and are almost identical for both groups of rats when they are killed sixty minutes or twenty-four hours after the injection (24 per cent for habituated rats killed after twenty-four hours).[16]
That the free morphine content, after the administration of 1 mg. per g., is nearly identical for both groups of animals killed fifteen, twenty, thirty, forty, sixty minutes after injection, so that it follows that the differences are due to the conjugated morphine content.
In recent years, interesting studies have been made of the elimination of morphine, and precise information has been supplied on the subject.[17]
Earlier authors studied the elimination of morphine through the urine or faeces. Their findings were contradictory, even in normal animals. They recovered only a part of the morphine administered, and the majority observed no difference between habituated and normal animals so far as the quantity of morphine excreted and the rate of excretion were concerned.
The individual differences observed even made it impossible to classify the animal species accurately from the point of view of the quantity excreted, although dogs appeared to metabolize morphine more rapidly than rats.
As early as 1884, Stolnikow discovered that after administering a strong dose of morphine to a dog, the hydrolysis of urine produced a greater colour reaction than a non-hydrolyzed sample. Ends (1928) was the first to prove that morphine could be excreted in a free and combined form. He showed that the quantity of morphine found in the urine of a rabbit was greater after two hours hydrolysis using N/40 sulphuric acid, and that such urine contained a greater quantity of glycuronic acid:
Oberst (1940), after hydrolysis of the urine of a morphine addict (heating at 100°C. for three hours, with one-fifth concentrated hydrochloric acid) obtained a large percentage of combined morphine, amounting to as much as 25 per cent of the quantity injected as against 5 per cent of free morphine. The ratio of combined morphine to free morphine varies from 3 to 36, according to the dose injected. Oberst therefore confirms the existence of a combined form of morphine and suggests that the possibility of a conjugation mechanism should be accepted. When morphine is administered by mouth, the quantity excreted in a free form is less, and the quantity excreted in a combined form is greater, than when morphine is administered subcutaneously.
Gross and Thompson (1940) hydrolyzed canine urine in an autoclave for thirty minutes, in the presence of 10 per cent by volume of concentrated hydrochloric acid, and assayed a definitely greater quantity of morphine than that calculated before hydrolysis. They differentiate between free morphine (FM) and combined morphine (CM) which they deduce from the difference between total morphine (TM) and free morphine. The total quantity found in the urine and faeces of normal dogs was from 80 to 92 per cent. The greater part of the morphine excreted was combined morphine; in some cases it amounted to more than 70 percent. The proportion of morphine found in faeces was very small (2 per cent).
In the case of dogs habituated for a year, the proportion of morphine varied from 35 to 66 per cent, about 30 per cent being in combined form; the proportion of free morphine was practically the same as in normal dogs. The authors found very small quantities of morphine in the faeces of habituated dogs, a result which was confirmed by Oberst (1942) in morphine addicts; he chiefly found free morphine in the faeces,[18]
The increase in free glycuronic acid suggests that glycuronate is probably the principal form of conjugation in man (1941).
In 1941, Thompson and Gross showed that the percentage of free morphine excreted by dogs in urine is practically identical in both normal and tolerant animals. There are two kinds of combined morphine: one fraction is easily hydrolyzable (EH) and is revealed by acid hydrolysis at pH I, after being heated to a temperature of 100°C. for two or three hours; the other is difficult to hydrolyze (DH) and cannot be assayed until urine, acidified with 5 per cent of its volume of concentrated hydrochloric acid, has been passed through an autoclave for thirty minutes. The percentages of excretion of these two forms are not the same for habituated and normal dogs. Habituated dogs excrete a greater quantity of (EH) than normal dogs, while the reverse applies in the case of the (DH) fraction. The total quantity of morphine excreted in urine, in proportion to the dose injected, is 80 to 92 per cent in-the case of normal animals, 20 per cent of which is in form (FM), 8 per cent in form (EH) and 60 per cent in form (DH). In habituated animals, 35 to 66 per cent of the morphine administered is recovered, composed of 20 per cent (FM), 16 per cent (EH) and 29 per cent (DH). Approximately 8 to 20 per cent morphine is not recovered in the case of normal animals and 34 to 65 per cent in that of habituated animals. These new results as a whole seem to suggest a greater destruction and a slower rate of conjugation in the habituated animal than in the normal animal.
On the basis of the results of Oberst, Gross,and Thompson, I endeavoured to determine in the urine and faeces of normal and habituated rats the quantities of free and conjugated morphine excreted after an injection of morphine.
Normal and habituated rats are placed in metabolism cages enabling urine and faeces to be obtained separately. These are collected twenty-four, forty-eight and seventy-two hours after the morphine injection.
The normal rats are from three to four months old. The subcutaneous injection of morphine hydrochloride (0.200 mg. per g., or 80 per cent of a 50 per cent lethal dose) produces apathy for four to five hours, during which period the animals do not feed. That dose does not alter the volumes of urine excreted which vary from 2 to 10 cc. per day; on the other hand, the excretion of faeces ceases during the first twenty-four or forty-eight hours after the injection. When the faeces reappear, they have a very solid consistency.
The habituated animals are excited after the morphine injection and feed normally. The volumes of urine excreted are almost the same as those of the normal animals, varying from 1 to 6 cc. Faeces are excreted daily and are diarrhoeic.
It is unnecessary to collect the excretions after seventy-two hours; after that period the morphine content for both normal and habituated rats is practically nil.
Each day the urine and faeces are separated into two equal parts, one for the extraction of free morphine (FM) and the other for the extraction of total morphine (TM). Conjugated morphine (CM) is represented by the difference between TM and FM.
Free morphine is extracted by treating the urine or the pulverized faeces in seven to ten times their volume or weight of 1/1,000 hydrochloric alcohol. The filtrate is evaporated under a current of cold air.
After the residue has been dissolved in 5 cc. of N/200 hydrochloric acid, N/100 soda is added until pH 6.3 is . reached. The solution is brought to 10 cc. with distilled water.
Concentrated hydrochloric acid, equalling a quarter of the volume of the urine and half the weight of the faeces, is added to the urine and pulverized faeces in which total morphine is sought. They are placed in sealed ampoules and autoclaved for thirty minutes at 120°C. They are then treated with seven to ten times their volume or weight of absolute alcohol and filtered. The filtrate is evaporated. The residue is then dissolved in 5 cc. of distilled water and brought to pH 6.3 first with concentrated soda and then with N/10 soda.
The morphine assay is carried, out on the denervated muscle of the leech, through the sensitizing action of morphine on acetylcholine (test B). I did not use the assay by the morphine-bromocholine antagonist action (test-C) having found that it gives the same results as test B.
Rats weighing 115 to 167 g. are given 0.200 mg. of morphine hydrochloride per g. The tables covering normal rats mention the quantifies of urine and faeces excreted over a period of seventy-two hours, and the total, free and conjugated morphine content found in the urine and the faeces. The average content of total morphine excreted is 60 per cent of the dose injected (the percent age figures vary from 44 to 85), comprising 13.3 per cent of free (FM) and 46.7 per cent of conjugated morphine (CM).
The average urinary excretion is 48 per cent of, the morphine injected, 12 per cent of which is free morphine and 36 per cent conjugated morphine.
An average of 12 per cent of morphine is excreted in the faeces, 1.4 per cent of which is free and 10.6 per cent conjugated morphine (see Table VI -- normal rats).
Compared with the results obtained by Thompson and Gross (1940-1941) in normal dogs, the quantity of mor- phine eliminated in the urine is much leas in rats (43 per cent) than in dogs (80-92 per cent). The free morphine content is about the same in both species (12 per cent for rats, 20 per cent for dogs).
I studied successively the elimination of morphine administered by various methods to rats in groups I, II, III and IV.
Group I, three successive subcutaneous injections of 0.100 mg. of morphine hydrochloride per g. at twenty-four hour intervals.
Group II, subcutaneous injections daily for three weeks of rapidly increasing doses of morphine hydrochloride varying from 0.100 to 0.500 mg. per g.
Group III, subcutaneous injections daily for a period of seven weeks, of morphine hydrochloride varying from 0.100 to 0.500 mg. per g., increased daily by 0.010 mg. per g.
Group IV, subcutaneous injections daily for twelve weeks of morphine hydrochloride varying from 0.100 to 0.500 mg. per g. for seven weeks, and then a constant dose of 0.500 mg. per g. for five weeks.
(a) Rats in Group I. These rats are given a subcutaneous injection of 0.200 mg. of morphine hydrochloride per g. on the fourth 'day. The urine and faeces are collected for three days.
The total morphine content excreted varies from 54.3 to 63 per cent of the quantity of morphine injected, 20 per cent of which on an average is free morphine and 38.6 per cent conjugated. The average amount of total morphine eliminated in the urine is 55.2 per cent of the injected dose, 17.5 per cent of which is free and 37.7 per cent conjugated morphine. The quantity of total morphine excreted in the faeces is small -- 3.4 per cent, 2.5 per cent of which is free and 0.9 per cent conjugated morphine (Table VII, group I).
(b) Rats in Group II. The morphine content is determined after the injection of 0.500 mg. of morphine hydrochloride per g. The quantity of total morphine excreted after that dose, 200 per cent of the 50 per cent lethal dose for normal animals, varies from 50 to 100 per cent (average 81.2 per cent) of the quantity injected. The average content in free morphine is 15.5 per cent and in conjugated morphine 38.3 per cent. The average quantity of free morphine eliminated in the urine is 52.5 per cent, 14.2 per cent of which is free morphine and 38.3 per cent conjugated morphine.
In their faeces, rats eliminate on an average 28.7 per cent of total morphine, 1.2 per cent of which is free morphine and 27.5 per cent conjugated morphine (Table VIII, group II).
(c) Rats in Group III (α) The rats are given 0.200 mg. of morphine hydrochloride per g. subcutaneously. The amount of total morphine excreted after that dose varies from 27 to 50 per cent (average 43 per cent) of the quantity injected, the average free morphine content being 14 per cent, and that of conjugated morphine 29 per cent.
The content of total morphine found in the urine is 26.6 per cent, 10.3 per cent of which is free morphine and 16.3 per cent conjugated morphine.
The total amount of morphine found in the faeces averages 16.4 per cent, 3.5 per cent of which is free morphine and 12.9 per cent conjugated morphine (Table IX, group III, 0.200 mg. per g.).
(β) The urine and faeces of two rats were collected after a subcutaneous injection of 0.500 mg. of morphine hydrochloride per g. The quantities of total morphine excreted were 36.8 and 31 per cent (average 33.9 per cent) of the close injected of which 23.6 and 19.9 per cent were free morphine, and 18.2 and 12 per cent conjugated morphine. The average percentage of total morphine eliminated in the urine is 19.8 per cent, of which 15.7 per cent is free morphine and 18.2 and 12 per cent conjugated morphine. The average percentage of total morphine eliminated in the urine is 19.8 per cent, 15.7 per cent of which is free morphine and 4.1 per cent conju- gated morphine. The average content of total morphine in the faeces is 14.1 per cent, 5.7 per cent of which is free morphine and 8.4 per cent conjugated morphine (Table X, group III, 0.500 mg. per g.).
The percentage of morphine excreted in the urine is less after the administration of 0.500 mg. per g. than after the administration of 0.200 mg. per g.
>(d) Rats in Group IV. As I stated in the chapter on the habituation of rats, these animals are intoxicated with morphine. Excretion is examined after a subcutaneous injection of 0.200 mg. of morphine hydrochloride per g. They excrete from 108 to 244 per cent of the quantity injected, or an average of 140.6 per cent -- more morphine than they received in their last injection.
The greater part of this morphine is excreted in conjugated form (average 126.1 per cent), the average percentage of free morphine being 14.5 per cent or the same as that of normal animals. The average content of total morphine in the urine varied slightly from that of normal rats. The increase in the morphine excreted was due to the conjugated morphine content in the faeces, (average 81.3 per cent), the free morphine content of the faeces being 2.1 per cent on an average (Table XI, group IV).
|
Urine after 24 hours |
Urine after 48 hours |
Urine after 72 hours |
Morphine in urine | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Weight of rats (in g.) |
Morph. injec. (mg.) |
Urine emitted (cc.) |
MoF (in γ)* |
MoC (in γ) |
MoT (in γ) |
Urine emitted (cc.) |
MoF (in γ) |
MoC (in γ) |
MoT (in γ) |
Urine emitted (cc.) |
MoF (in γ) |
MoC (in γ) |
MoT (in γ) |
MoF (in γ) |
MoC (in γ) |
MoT (in γ) |
| 115 | 23 | 10.8 | 4,0 00 | 6,000 | 10,000 | 6.2 | 0 | 3,320 | 3,320 | 6.8 | 0 | 2,200 | 2,200 | 4,000 | 11,520 | 15,520 |
| 120 | 24 | 9.2 | 1,000 | 5,660 | 6,660 | 5.2 | 0 | 3,320 | 3,320 | 4 | 0 | 1,000 | 1,000 | 1,000 | 9,980 | 10,980 |
| 148 | 29.6 | 4 | 2,000 | 5,500 | 7,500 | 0.4 |
|
|
|
3.6 | 0 | 4,000 | 4,000 | 2,000 | 9,500 | 11,500 |
| 145 | 29 | 4.5 | 2,000 | 10,250 | 12,250 | 1.3 |
|
|
|
4.2 | 0 | 4,000 | 4,000 | 2,000 | 14,250 | 16,250 |
| 150 | 30 | 1 | 2,000 | 2,000 | 4,000 | 4 | 5,000 | 1,000 | 6,000 | 1.2 | 0 | 7,000 | 3,000 | 10,000 | ||
| 167 | 33.4 | 4 | 3,320 | 10,000 | 13,320 | 2 | 1,200 | 2,120 | 3,320 | 4.4 | 0 | 1,700 | 1,700 | 4,520 | 13,820 | 18,340 |
|
Faeces after 24 hours |
Faeces after 48 hours |
Faeces after 72 hours |
Morphine in faeces | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Weight of rats (in g.) |
Morph. injec. (mg.) |
Faeces excre. (in g.) |
MoF (in γ) |
MoC (in γ) |
MoT (in γ) |
Faeces excre. (in g.) |
MoF (in γ) |
MoC (in γ) |
MoT (in γ) |
Faeces excre. (in g.) |
MoF (in γ) |
MoC (in γ) |
MoT (in γ) |
MoF (in γ) |
MoC (in γ) |
MoT (in γ) |
| 115 | 23 | 0.750 |
|
|
|
0.250 | 0 | 3,000 | 3,000 | 0.8 | 0 | 1,330 | 1,330 | 0 | 4,330 | 4,330 |
| 120 | 24 | 0.050 | 1.050 |
|
|
|
0 | 1,200 | 1,200 | 2.4 | 0 | 1,000 | 1,000 | 0 | 2,200 | 2,200 |
| 148 | 29.6 | 0 |
|
|
|
0 |
|
|
|
1.6 | 500 | 1,040 | 1,540 | 500 | 1,040 | 1,540 |
| 145 | 29 | 0 |
|
|
|
0 |
|
|
|
1.4 | 500 | 500 | 1,000 | 500 | 500 | 1,000 |
| 150 | 30 | 0 |
|
|
|
0 |
|
|
|
1.6 | 800 | 5,860 | 6,660 | 800 | 5,860 | 6,660 |
| 167 | 33.4 | 0 |
|
|
|
0 |
|
|
|
1.1 | 600 | 2,720 | 3,320 | 600 | 2,720 | 3,320 |
|
Morphine excreted |
Percentages of morphine recovered | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Morph. injec. (mg.) |
MoF (in γ) |
MoC (in γ) |
MoT (in γ) |
MoF |
MoC |
MoT |
MoF |
MoC |
MoT |
MoF |
MoC |
MoT |
| 23 | 4,000 | 15,850 | 19,850 | 17.3 | 68.7 | 86 | 17.3 | 50.1 | 67.4 | 0 | 18.6 | 18.6 |
| 24 | 1,000 | 12,180 | 13,180 | 4.5 | 49.5 | 54 | 4.5 | 40.4 | 44.9 | 0 | 9.1 | 9.1 |
| 29.6 | 2,500 | 10,540 | 13,040 | 8.4 | 35.6 | 44 | 6.8 | 32 | 38.8 | 1.6 | 3.6 | 5.2 |
| 29 | 2,500 | 14,750 | 17,250 | 8.6 | 51.4 | 60 | 6.9 | 49.7 | 56.6 | 1.7 | 1.7 | 3.4 |
| 30 | 7,800 | 8,860 | 16,660 | 26 | 29 | 55 | 23 | 10 | 33 | 3 | 19 | 22 |
| 33.4 | 5,120 | 16,540 | 21,660 | 15.3 | 49.7 | 65 | 13.5 | 41.5 | 55 | 1.8 | 8.2 | 10 |
|
|
|
|
Averages |
13.3 | 46.7 | 60 | 12 | 36 | 48 | 1.4 | 10.6 | 12 |
In these tables the sign γ stands for microgram.
|
Urine after 24 hours |
Urine after 48 hours |
Urine after 72 hours |
Morphine in urine | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Weight of rats (in g.) |
Morph. injec. (mg.) |
Urine emitted (cc.) |
MoF (in γ)* |
MoC (in γ) |
MoT (in γ) |
Urine emitted (cc.) |
MoF (in γ) |
MoC (in γ) |
MoT (in γ) |
Urine emitted (cc.) |
MoF (in γ) |
MoC (in γ) |
MoT (in γ) |
MoF (in γ) |
MoC (in γ) |
MoT (in γ) |
| 100 | 20 | 6.8 | 3,000 | 7,000 | 10,000 | 6 | 0 | 1,000 | 1,000 | 4.2 | 0 | 600 | 600 | 3,000 | 8,600 | 11,600 |
| 100 | 20 | 8.2 | 4,000 | 6,000 | 10,000 | 4 | 0 | 500 | 500 | 0.8 | 0 | 0 | 0 | 4,000 | 6,500 | 10,500 |
|
Faeces after 24 hours |
Faeces after 48 hours |
Faeces after 72 hours |
Morphine in faeces | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Weight of rats (in g.) |
Morph. injec. (mg.) |
Faeces excre. (in g.) |
MoF (in γ) |
MoC (in γ) |
MoT (in γ) |
Faeces excre. (in g.) |
MoF (in γ) |
MoC (in γ) |
MoT (in γ) |
Faeces excre. (in g.) |
MoF (in γ) |
MoC (in γ) |
MoT (in γ) |
MoF (in γ) |
MoC (in γ) |
MoT (in γ) |
| 100 | 20 | 0.750 |
|
|
|
1.55 | 1,000 | 0 | 1,000 | 0.450 | 0 | 0 | 0 | 1,000 | 0 | 1,000 |
| 1,00 | 20 | 0.900 |
|
|
|
0.40 | 0 | 240 | 240 | 0.900 | 0 | 120 | 120 | 0 | 360 | 360 |
|
Morphine excreted |
Percentages of morphine recovered | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Morph. injec. (mg.) |
MoF (in γ) |
MoC (in γ) |
MoT (in γ) |
MoF |
MoC |
MoT |
MoF |
MoC |
MoT |
MoF |
MoC |
MoT |
| 20 | 4,000 | 8,600 | 12,600 | 20 | 43 | 63 | 15 | 43 | 58 | 5 | 0 | 5 |
| 20 | 4,000 | 6,860 | 10,860 | 20 | 34.3 | 54.3 | 20 | 32.5 | 52.5 | 0 | 1.8 | 1.8 |
|
|
|
|
Averages |
20 | 38.6 | 58.6 | 17.5 | 37.7 | 55.2 | 2.5 | 0.9 | 3.4 |
|
Urine after 24 hours |
Urine after 48 hours |
Urine after 72 hours |
Morphine in urine | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Weight of rats (in g.) |
Morph. injec. (mg.) |
Urine emitted (cc.) |
MoF (in γ)* |
MoC (in γ) |
MoT (in γ) |
Urine emitted (cc.) |
MoF (in γ) |
MoC (in γ) |
MoT (in γ) |
Urine emitted (cc.) |
MoF (in γ) |
MoC (in γ) |
MoT (in γ) |
MoF (in γ) |
MoC (in γ) |
MoT (in γ) |
| 108 | 54 | 13 | 4,000 | 0 | 4,000 | 0 | 0 |
|
0 | 3.8 | 0 | 1,000 | 1,000 | 4,000 | 1,000 | 5,000 |
| 94 | 47 | 6 | 10,000 | 20,000 | 30,000 | 3 | 0 |
|
0 | 3.8 | 0 |
|
0 | 10,000 | 20,000 | 30,000 |
| 108 | 54 | 3.6 | 6,000 | 20,000 | 26,000 | 8.5 | 300 | 4,700 | 5,000 | 3.5 | 0 | 1,000 | 1,000 | 6,300 | 25,700 | 32,000 |
| 94 | 47 | 2.4 | 8,000 | 24,000 | 32,000 | 8.6 | 0 | 4,000 | 4,000 | 4.2 | 0 | 1,000 | 1,000 | 8,000 | 29,000 | 37,000 |
|
Faeces after 24 hours |
Faeces after 48 hours |
Faeces after 72 hours |
Morphine in faeces | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Weight of rats (in g.) |
Morph. injec. (mg.) |
Faeces excre. (in g.) |
MoF (in γ) |
MoC (in γ) |
MoT (in γ) |
Faeces excre. (in g.) |
MoF (in γ) |
MoC (in γ) |
MoT (in γ) |
Faeces excre. (in g.) |
MoF (in γ) |
MoC (in γ) |
MoT (in γ) |
MoF (in γ) |
MoC (in γ) |
MoT (in γ) |
| 108 | 54 | 0 |
|
|
|
6.4 | 0 | 12,000 | 12,000 | 1.8 | 0 | 10,000 | 10,000 | 0 | 22,000 | 22,000 |
| 94 | 47 | 1.3 | 0 | 5,000 | 5,000 | 1.25 |
|
|
0 | 1.3 | 0 | 3,000 | 3,000 | 0 | 8,000 | 8,000 |
| 108 | 54 | 0 |
|
|
|
3 | 1,500 | 18,500 | 20,000 | 2.7 | 0 | 2,000 | 2,000 | 1,500 | 20,500 | 22,000 |
| 94 | 47 | 0 |
|
|
|
0.8 | 1,500 | 0 | 1,000 | 1.8 | 0 | 6,600 | 6,600 | 1,000 | 6,600 | 7,600 |
|
Morphine excreted |
Percentages of morphine recovered | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Morph. injec. (mg.) |
MoF (in γ) |
MoC (in γ) |
MoT (in γ) |
MoF |
MoC |
MoT |
MoF |
MoC |
MoT |
MoF |
MoC |
MoT |
| 54 | 4,000 | 23,000 | 27,000 | 7.4 | 42.6 | 50 | 7.4 | 1.8 | 9.2 | 0 | 40 8 | 40.8 |
| 47 | 10,000 | 28,000 | 38,000 | 21.3 | 58.7 | 60 | 21 | 42 | 63 | 0 | 17 | 17 |
| 54 | 7,800 | 46,200 | 54,000 | 14.4 | 85.6 | 100 | 11.6 | 47.6 | 59.2 | 2.7 | 38.1 | 40.8 |
| 47 | 9,000 | 35,600 | 44,600 | 19 | 76 | 95 | 17 | 61.7 | 78.7 | 2.1 | 14.2 | 16.3 |
|
|
|
|
Averages |
15.5 | 65.7 | 81.2 | 14.2 | 38.3 | 52.5 | 1.2 | 27.5 | 28.7 |
|
Urine after 24 hours |
Urine after 48 hours |
Urine after 72 hours |
Morphine in urine |
|||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Weight of rats (in g.) |
Morph. injec. (mg.) |
Urine emitted (cc.) |
MoF (in γ)* |
MoC (in γ) |
MoT (in γ) |
Urine emitted (cc.) |
MoF (in γ) |
MoC (in γ) |
MoT (in γ) |
Urine emitted (cc.) |
MoF (in γ) |
MoC (in γ) |
MoT (in γ) |
MoF (in γ) |
MoC (in γ) |
MoT (in γ) |
| 100 | 20 | 3.8 | 1,480 | 3,520 | 5,000 | 0 |
|
|
|
3.4 | 500 | 1,500 | 2,000 | 1,980 | 5,020 | 7,000 |
| 100 | 20 | 1.8 | 2,000 | 3,000 | 5,000 | 0 |
|
|
|
5.2 | 0 | 1,600 | 1,600 | 2,000 | 4,600 | 6,600 |
| 102 | 20.4 | 1.5 | 670 | 3,330 | 4,000 | 0 |
|
|
|
2.2 | 200 | 50 | 250 | 870 | 3,380 | 4,250 |
| 90 | 18 | 2.3 | 2,500 | 0 | 2,500 | 3.4 | 0 | 1,000 | 1,000 | 2.2 | 0 | 500 | 500 | 2,500 | 1,500 | 4,000 |
| 91 | 18.2 | 1.4 | 2,500 | 0 | 2,500 | 6.3 | 0 |
l,000 |
1,000 | 3.5 | 0 | 500 | 500 | 2,500 | 1,500 | 4,000 |
|
Faeces after 24 hours |
Faeces after 48 hours |
Faeces after 72 hours |
Morphine in faeces | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Weight of rats (in g.) |
Morph. injec. (mg.) |
Faeces excre. (in g.) |
MoF (in γ) |
MoC (in γ) |
MoT (in γ) |
Faeces excre. (in g.) |
MoF (in γ) |
MoC (in γ) |
MoT (in γ) |
Faeces excre. (in g.) |
MoF (in γ) |
MoC (in γ) |
MoT (in γ) |
MoF (in γ) |
MoC (in γ) |
MoT (in γ) |
| 100 | 20 | 1 |
|
|
|
0.5 | 1,000 | 1,000 | 2,000 | 0 |
|
|
|
1,000 | 1,000 | 1,000 |
| 100 | 20 | 0.5 |
|
|
|
2.1 | 1,000 | 1,000 | 2,000 | 0 |
|
|
|
1,000 | 1,000 | 1,000 |
| 102 | 20.4 | 4.1 | 500 | 500 | 1,000 | 2 | 150 | 150 | 3.7 | 125 | 0 | 125 | 775 | 500 | 1,275 | |
| 90 | 18 | 1.2 | 0 | 1,140 | 1,140 | 1.5 | 0 | 2,660 | 2,660 | 1.4 | 0 | 1,320 | 1,320 |
|
5,120 | 5,120 |
| 92 | 18.2 | 1 | 740 | 920 | 1,660 | 1.8 | 0 | 2,000 | 2,000 | 0.5 | 0 | 1,250 | 1,250 | 740 | 4,170 | 4,910 |
|
Morphine excreted |
Percentages of morphine recovered | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Morph. injec. (mg.) |
MoF (in γ) |
MoC (in γ) |
MoT (in γ) |
MoF |
MoC |
MoT |
MoF |
MoC |
MoT |
MoF |
MoC |
MoT |
| 20 | 2,980 | 6,020 | 9,000 | 14.9 | 30.1 | 45 | 9.9 | 25.1 | 35 | 5 | 5 | 10 |
| 20 | 3,000 | 5,600 | 8,600 | 15 | 28 | 43 | 10 | 23 | 33 | 5 | 5 | 10 |
| 20.4 | 1,645 | 3,880 | 5,525 | 8 | 19 | 27 | 4.2 | 16.6 | 20.8 | 3.4 | 2.8 | 6.2 |
| 18 | 2,500 | 6,620 | 9,120 | 13.8 | 36.8 | 50.6 | 13.8 | 8.4 | 22.2 | 0 | 28.4 | 28 4 |
| 18.2 | 3,240 | 5,670 | 8,910 | 17.7 | 31.2 | 48.9 | 13.8 | 8.2 | 22 | 4 | 22.9 | 26.9 |
|
|
|
|
Averages |
14 | 29 | 43 | 10.3 | 16.3 | 26.6 | 3.5 | 12.9 | 16.4 |
|
Urine after 24 hours |
Urine after 48 hours |
Urine after 72 hours |
Morphine in urine | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Weight of rats (in g.) |
Morph. injec. (mg.) |
Urine emitted (cc.) |
MoF (in γ)* |
MoC (in γ) |
MoT (in γ) |
Urine emitted (cc.) |
MoF (in γ) |
MoC (in γ) |
MoT (in γ) |
Urine emitted (cc.) |
MoF (in γ) |
MoC (in γ) |
MoT (in γ) |
MoF (in γ) |
MoC (in γ) |
MoT (in γ) |
| 82 | 41 | 3.6 | 8,000 | 0 | 8,000 | 6.8 | 500 | 2,300 | 2,800 | 0 |
|
|
|
8,500 | 2,300 | 10,800 |
| 99 | 49.5 | 4.2 | 5,000 | 0 | 5,000 | 6.4 | 300 | 900 | 1,200 | 6.3 | 0 | 400 | 400 | 5,300 | 1,300 | 6,600 |
|
Faeces after 24 hours |
Faeces after 48 hours |
Faeces after 72 hours |
Morphine in faeces | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Weight of rats (in g.) |
Morph. injec. (mg.) |
Faeces excre. (in g.) |
MoF (in γ) |
MoC (in γ) |
MoT (in γ) |
Faeces excre. (in g.) |
MoF (in γ) |
MoC (in γ) |
MoT (in γ) |
Faeces excre. (in g.) |
MoF (in γ) |
MoC (in γ) |
MoT (in γ) |
MoF (in γ) |
MoC (in γ) |
MoT (in γ) |
| 82 | 41 | 1.35 | 1,000 | 0 | 1,000 | 1.7 | 200 | 800 | 1,000 | 1 | 0 | 2,320 | 2,320 | 1,200 | 3,120 | 4,320 |
| 99 | 49.5 | 2.05 | 4,000 | 0 | 4,000 | 2 | 200 | 200 | 400 | 2 | 0 | 4,400 | 4,400 | 4,200 | 4,600 | 8,800 |
|
Morphine excreted |
Percentages of morphine recovered | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Morph. injec. (mg.) |
MoF (in γ) |
MoC (in γ) |
MoT (in γ) |
MoF |
MoC |
MoT |
MoF |
MoC |
MoT |
MoF |
MoC |
MoT |
| 41 | 9,700 | 5,420 | 15,120 | 23.6 | 13.2 | 36 8 | 20.7 | 5 6 | 26 3 | 2.9 | 7.6 | 10 5 |
| 49.5 | 9,500 | 5,900 | 15,400 | 19 | 12 | 31 | 10 7 | 2.6 | 13.3 | 8.5 | 9.2 | 17.7 |
|
|
|
|
Averages |
21.3 | 12 6 | 33.9 | 15.7 | 4.1 | 19.8 | 5.7 | 8.4 | 14.1 |
|
Urine after 24 hours |
Urine after 48 hours |
Urine after 72 hours |
Morphine in urine | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Weight of rats (in g.) |
Morph. injec. (mg.) |
Urine emitted (cc.) |
MoF (in γ)* |
MoC (in γ) |
MoT (in γ) |
Urine emitted (cc.) |
MoF (in γ) |
MoC (in γ) |
MoT (in γ) |
Urine emitted (cc.) |
MoF (in γ) |
MoC (in γ) |
MoT (in γ) |
MoF (in γ) |
MoC (in γ) |
MoT (in γ) |
| 114 | 22.8 | 2.4 | 2,000 | 4,660 | 6,660 | 8 | 0 | 4,000 | 4,000 | 10 | 0 | 6,660 | 6,660 | 2,000 | 15,320 | 17,320 |
| 90 | 18 | 1.8 | 3,320 | 6,680 | 10,000 | 4 | 0 | 2,000 | 2,000 | 0 |
|
|
|
3,320 | 8,680 | 12,000 |
| 95 | 19 | 6.8 | 2,400 | 13,600 | 16,000 | 0 | 0 |
|
0 | 0 |
|
|
|
2,400 | 13,600 | 16,000 |
| 88 | 17.6 | 2.8 | 1,000 | 4,000 | 5,000 | 3.8 | 0 | 1,000 | 1,000 | 1 | 0 | 0 | 0 | 1,000 | 5,000 | 6,000 |
| 100 | 20 | 2 6 | 3,200 | 1,800 | 5,000 | 6 | 0 |
|
0 | 2 | 0 | 0 | 0 | 3,200 | 1,800 | 5,000 |
|
Faeces after 24 hours |
Faeces after 48 hours |
Faeces after 72 hours |
Morphine in faeces | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Weight of rats (in g.) |
Morph. injec. (mg.) |
Faeces excre. (in g.) |
MoF (in γ) |
MoC (in γ) |
MoT (in γ) |
Faeces excre. (in g.) |
MoF (in γ) |
MoC (in γ) |
MoT (in γ) |
Faeces excre. (in g.) |
MoF (in γ) |
MoC (in γ) |
MoT (in γ) |
MoF (in γ) |
MoC (in γ) |
MoT (in γ) |
| 114 | 22.8 | 2.4 | 1,400 | 2,600 | 4,000 | 1.7 | 200 | 3,120 | 3,320 | 3.4 | 0 | 5,000 | 5,000 | 1,600 | 10,720 | 12,320 |
| 90 | 18 | 1.2 | 500 | 4,500 | 5,000 | 0 |
|
|
0 | 2.2 | 0 | 2,500 | 2,500 | 500 | 7,000 | 7,500 |
| 95 | 19 | 2.8 | 200 | 2,600 | 2,800 | 1.2 | 0 | 500 | 500 | 1.8 | 0 | 1,400 | 1,400 | 200 | 4,500 | 4,700 |
| 88 | 17.6 | 2.4 | 0 | 28,000 | 28,000 | 1.55 | 0 | 4,000 | 4,000 | 3.25 | 0 | 5,000 | 5,000 | 0 | 37,000 | 37,000 |
| 100 | 20 | 3.3 | 0 | 14,000 | 14,000 | 2 | 0 | 3,320 | 3,320 | 1.6 | 0 | 0 | 0 | 0 | 17,320 | 17,320 |
|
Faeces after 24 hours |
Faeces after 48 hours |
Faeces after 72 hours | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Weight of rats (in g.) |
Morph. injec. (mg.) |
Faeces excre. (in g.) |
MoF (in γ) |
MoC (in γ) |
MoT (in γ) |
Faeces excre. (in g.) |
MoF (in γ) |
MoC (in γ) |
MoT (in γ) |
Faeces excre. (in g.) |
MoF (in γ) |
MoC (in γ) |
MoT (in γ) |
| 22.8 | 3,600 | 26,040 | 29,540 | 15.8 | 114.2 | 130 | 8.8 | 67.2 | 76 | 7 | 47 | 54 | |
| 18 | 3,820 | 15,680 | 19,500 | 21.2 | 87.1 | 108.3 | 18.6 |
48. |
66.6 | 2.8 | 38.9 | 41.7 | |
| 19 | 2,600 | 18,100 | 20,700 | 13.7 | 95.2 | 108.9 | 12.6 | 71.6 | 84.2 | 1 | 23.7 | 24.7 | |
| 17.6 | 1,000 | 42,000 | 43,000 | 5.8 | 238.5 | 244.3 | 5.6 | 28.4 | 34 | 0 | 210 3 | 210.3 | |
| 20 | 3,200 | 19,120 | 22,320 | 16 | 95.6 | 111.6 | 16 | 9 | 25 | 0 | 86.6 | 86.6 | |
|
|
|
|
Averages |
14.5 | 126.1 | 140.6 | 12.3 | 44.8 | 57.1 | 2.1 | 81.3 | 83.4 | |
|
Percentages of morphine recovered | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
|
Condition of rats |
Duration of habituation |
Morphine injected (in mg. per g.) |
MoF |
MoC |
MoT |
MoF |
MoC |
MoT |
MoF |
MoC |
MoT |
|
Normal |
|
0.200 | 13.3 | 46.7 | 60 | 12 | 36 | 48 | 1.4 | 10.6 | 12 |
|
Group I |
3 days | 0.200 | 20 | 38.6 | 58.6 | 17.5 | 37.7 | 55.2 | 2.5 | 0.9 | 3.4 |
|
Group II |
3 weeks | 0.500 | 15.5 | 65.7 | 81.2 | 14.2 | 38.3 | 52.5 | 1.2 | 27.5 | 28.7 |
|
Group III |
7 weeks | 0.200 | 14 | 29 | 43 | 10.3 | 16.3 | 26.6 | 3.5 | 12.9 | 16.4 |
|
Group III |
7 weeks | 0.500 | 21.3 | 12.6 | 33.9 | 15.7 | 4.1 | 19.8 | 5.7 | 8.4 | 14.1 |
|
Group IV |
12 weeks | 0.200 | 14.5 | 126.1 | 140.6 | 12.3 | 44.8 | 57.1 | 2.1 | 81.3 | 83.4 |
(a) Excretion of morphine after injection of 0.200 mg. per g.
A comparison between the excretion of morphine by normal rats and by the various groups of habituated rats after the subcutaneous injection of 0.200 mg. of morphine hydrochloride per g. shows:
(α) Total morphine: Normal rats and the rats in group I eliminate identical quantities of morphine, particularly in their urine. The rats in group III excrete less, the difference being due to the morphine content of the urine. On the other hand, the rats in group IV excrete more morphine than normal rats, the increase being due to the quantity excreted in the faeces (3.4 per cent as against 12 per cent for normal rats).
( β) Free morphine: The percentages for free morphine excreted in urine and faeces are almost identical for normal rats and rats in groups I, III and IV.
(γ) Conjugated morphine: The differences in excretion observed are due to the conjugated morphine eliminated in the urine, except for the rats in group IV, for which the conjugated morphine content in the faeces is much larger than for the other groups.
Normal rats and those in group I excrete equivalent quantities of conjugated morphine in the urine.
The rats in group III excrete less conjugated morphine than normal rats, the difference being due to the conjugated morphine content of the urine.
With dogs, Thompson and Gross recovered less morphine excreted by habituated animals than by normal animals, the percentage of free morphine in the urine being substantially the same. Similarly, habituated rats excrete less morphine than normal rats, the percentages of free morphine in the urine being equal; the percentage of conjugated morphine in the urine is less with habituated rats than with normal rats (16 and 36 per cent).
(b) Elimination of morphine after injection of 0.500 mg. per g.
The habituated rats in groups II and III, the excretions of which are gathered after the injection of a dose of morphine hydrochloride equivalent to 0.500 mg. per g., differ in their behaviour.
(α) Total morphine: The rats in group III, after injection of 0.500 mg. per g., excrete somewhat less accumulated morphine than those in the same group after the injection of 0.200 mg. per g., a feature identical with that observed in morphine addicts by Oberst (1940), who noted that a slight decrease in the percentage excreted when the quantities administered are increased. The rats in group II excrete more morphine in the urine and faeces than the rats in group III. The percentage of total morphine in the urine is quantitatively the same as that for normal rats. The percentage of total morphine in the faeces is higher than that in normal rats.
( β) Free morphine: The percentages of free morphine in the urine and faeces are the same for the rats in groups II and III and for normal rats.
(γ) Conjugated morphine: The rats in group III, the excretions of which are examined after the administration of 0.500 mg. per g., eliminate less conjugated morphine than the rats in the same group after the administration of 0.200 mg. per g. They excrete in the urine 12.6 and 29 per cent of conjugated morphine and in the faeces 8.4 and 12.9 per cent, respectively.
The rats in group II excrete on an average more conjugated morphine than normal rats (65.7 and 46.7 per cent), the difference being due to the excretion of conjugated morphine in the faeces (27.5 and 20.6 per cent).
The rats injected for three days (group I) do not behave very differently from normal rats, but it should be recalled that though they tolerate toxic doses of morphine they are still sensitive to its analgesic action. The rats injected for seven weeks (group III), which tolerate toxic doses of morphine and upon which it no longer has any analgesic action, excrete less morphine than normal rats. The rats injected for twelve weeks (group IV), which have received very large doses of morphine and are in a weak state, excrete more morphine than they receive.
The rats injected for three weeks (group II), which have been habituated very rapidly and have undergone a less progressive intoxication, behave similarly to those in group IV, which may be explained by their method of habituation.
(1) The quantities of total morphine found in the organs of normal and habituated rats of 100 g., having received 0.200 and 1 mg. of morphine hydrochloride per g., and examined twenty-four hours, twenty minutes and sixty minutes respectively after the injection[19] are compared in the following table:
The organism of a normal rat receiving 1 mg. of morphine hydrochloride per g. contains, after twenty minutes, 6.9 mg. of total morphine. That of a habituated rat treated in the same manner contains 13.2 mg., whereas that of a habituated rat examined twenty-four hours after the injection of 0.200 mg. per g. contains 5.6 mg. Hence the organism of a habituated rat, twenty minutes after the administration of 1 mg. per g., contains a quantity very similar to that found in a normal rat, plus the morphine found in the habituated rat which has not received any morphine for twenty-four hours (6.9 + 5.6= 12.5).
|
Normal rats (Morphine expressed in mg.) |
Habituated rats (Morphine expressed in mg.) | |||||
|---|---|---|---|---|---|---|
|
24 hours after 0.200 mg./g. |
20 min. after 1 mg. per g. |
60 min. after 1 mg. per g. |
24 hours after 0.200 mg./g. |
20 min. after 1 mg. per g. |
60 min. after 1 mg. per g. | |
|
Blood and various tissues |
4.23 | 5.46 | 10.2 | 3.20 | 9.61 | 11.02 |
|
Stomach |
0 | 0.60 | 0.66 | 0 | 0.69 | 0.75 |
|
Intestines |
0.56 | 0.84 | 2.10 | 2.45 | 2.94 | 3.5 |
|
|
4.79 | 6.90 | 12.96 | 5.65 | 13.24 | 15.27 |
The organism of a habituated rat killed sixty minutes after the injection of 1 mg. of morphine per g. contains less morphine (15.2) than the sum of the morphine contents of the normal rat (12.9) plus that found in the habituated rat (5.6) which has received no morphine for twenty-four hours (12.9 + 5.6= 18.5). Hence the habituated rat destroys the morphine more quickly than the normal rat; the difference is already appreciable in one hour: 18.5-15.2 =3.3.
The figures for the total morphine contents of the biological liquids, the tissues examined and the excretions, twenty-four hours after the administration of 0.200 mg.per g., show that the habituated rat[20] destroys or transforms more morphine than the normal rat[21] (see table below):
|
Normal rat (in mg.) |
Habituated rat (in mg.) | |
|---|---|---|
|
Blood and various tissues |
4.23 | 3.20 |
|
Intestine |
0.56 | 2.45 |
|
Urine |
9 | 3.8 |
|
Faeces |
0 | 0.76 |
|
|
13.79 | 10.21 |
|
Percentage of morphine |
69 | 51 |
The morphine content found in the normal rat (13.8 mg.) is greater than in the habituated rat (10.2 mg.); but it should be remembered that the latter received first a progressively increasing dose of morphine over a period of six weeks and then, during the seventh week, a daily dose 250 per cent of that injected into the normal rat; there can be no doubt, in view of the above results, that at the time of the last injection its organism contained at least as much morphine as that found after the administration of 0.200 mg. per g. (5.6 mg.). Moreover, a great deal of the morphine in the organism of the habituated rat is located in the intestine[22] (2.45 mg. as against 0.56 mg. for a normal rat).
Hence a comparison of the percentages of morphine found, which are higher in the normal rat than in the habituated rat (69 and 51 per cent respectively), seem to me to indicate clearly that the latter transforms or destroys more than the former.
(3) No accumulation of morphine takes place, either in the muscles or in any of the organs. There is no morphine in the muscles of the habituated rat twenty-four hours after the injection, and in that of the normal rat only 8 per cent of the morphine administered is found there. In the habituated rat, a large part of the morphine (12 per cent) is found in the intestine and will later be excreted in the faeces.
>(4) The elimination of conjugated morphine is much greater in the habituated animals than in the normal ones.
On the other hand, if they are habituated too quickly or intoxicated by administration of large doses (group IV) the quantity of conjugated morphine eliminated by the faeces rises rapidly and may even exceed the amount of the last dose of morphine injected. This result seems to me surprising; a priori I should have expected in such a case to find more free morphine, not conjugated morphine. Hence the large elimination of conjugated morphine proves that in certain conditions, if the organism is no longer capable of transforming or destroying the morphine,[23] it can still effect a conjugation, which does not prevent it from succumbing rapidly to the toxic effect of the drug.
Among the numerous hypotheses which have attempted to explain habituation to morphine, that of Loofs (1922) suggesting that morphine may take the place of a cellular constituent, choline, in lecithin, is untenable, first, because there is no accumulation of morphine in the various organs or tissues, and secondly, because, using the technique of Oucet and Kahane (1946) for assaying the choline, I found no difference in the total choline content of the organisms of normal and habituated rats (0.711 to 0.829 mg. per g. and 0.797 to 0.823 mg. per g. respectively).
I attempted to achieve the state of habituation experimentally:
In mice, which do not become habituated to morphine;
In rats, which quickly become habituated to toxic doses and can tolerate by subcutaneous injection, 400 per cent of the 50 per cent lethal dose for a normal animal. The analgesic action decreases after the third day of treatment for an effective dose of morphine hydrochloride, and disappears with the same dose after fifteen days of daily administration.
The rat rapidly (in five days) ceases to tolerate toxic doses and in twenty-five days again becomes sensitive to the analgesic action of the initial effective dose. It does not manifest any cross habituation to hypnotics (evipan, soneryl).
The rat becomes accustomed to repeated injections of heroin in increasing dose and tolerates by subcutaneous injection, ten times the 50 per cent lethal dose for a normal animal. The analgesic action of an effective dose of heroin diminishes and disappears almost entirely after fourteen days of daily administration. There is no sign of cross habituation to evipan.
The rat becomes habituated less easily to small constant doses of heroin but can nevertheless, in these conditions, tolerate 200 per cent of the 50 per cent lethal dose for a normal animal, subcutaneously administered, and the analgesic action weakens in fourteen days.
I worked out two techniques of biological assay on the denervated muscle of the leech, one specific for morphine in the presence or absence of oxydimorphine, the other permitting the simultaneous assay of both alkaloids, and I evolved a technique for the extraction of morphine, total and free, which has the advantage of being relatively simple.
The technique for the assay of morphine alone, in the presence or absence of oxydimorphine, makes use of the increase in the acetylcholinic contraction of the muscle of the leech, under the influence of 30 to 200 δ of morphine hydrochloride.
The technique for the simultaneous assay of morphine and oxydimorphine consists in reducing, by the addition of 15 to 50 δ of morphine or oxydimorphine hydrochloride, the contraction caused by the hydrobromic ester of choline on the muscle of the leech.
I studied the fixation of morphine in the organism and its elimination:
Oxydimorphine is not found in the tissues of either the normal or the habituated rat in contact with morphine, or in the organism of rats after the injection of morphine;
The determination of the free and conjugated morphine contents of the different tissues and biological liquids proves that there is no accumulation of morphine in the rat's organism;
The total morphine contents of the blood and tissues were examined comparatively for the normal and the habituated rat after administration of 0.200 mg. and 1 mg. of morphine hydrochloride per g. The animals were killed twenty minutes, sixty minutes and twenty-four hours after the injection. In those conditions, the morphine contents, which are distinctly higher in the habituated rat twenty minutes after the injection, become equal after sixty minutes. In one hour the habituated rat is capable of transforming or destroying part of the morphine administered;
The elimination of total morphine (free plus conjugated) through the urine and faeces which is less in habituated rats that have received regularly increasing doses for seven weeks than in normal rats, is due to a difference in the conjugated morphine.
A comparison of the amounts of morphine found in the organs, biological liquids and excretions indicates that the morphine disappears or is transformed more rapidly in the organism of habituated rats than in normal rats.
I can therefore answer the two questions which I raised at the beginning of this work in the following way:
During the habituation of the rat, it is not possible to identify oxydimorphine in the organs and excretions;
The organisms of habituated rats transform or destroy more morphine than that of normal rats.
It has not been possible to ascertain the precise nature of the transformations undergone by the morphine, and different enzymatic systems must be responsible for them. Research would have to be undertaken to observe concurrently, in vitro and in vivo, the action of the morphine on the enzymatic systems of normal and habituated rats.
6R.P. = Rhone-Poulenc.
8I ascertained that 2 or 4 mg. of morphine hydrochloride in solution in physiological serum, can be recovered after having been kept in the oven at 37° C. for twenty-four hours.
9For the period prior to 1934, see Jeanne Lévy.
l0Composition of the Tyrode solution: pure sodium chloride, R.P, 9 g.; pure potassium chloride, R.P., 0.42 g.; pure dry calcium chloride, R P., 0.12 g.; pure magnesium chloride, R.P., 0 005 g.; pure sodium bicarbonate, 0.5 g.; distilled water Q.S.P., 1000 cc.
11Bernheim and Bernheim (1944, 1945) hydrolize in the presence of hydrochloric acid N (free morphine) and also autoclave in the presence of normal hydrochloric acid (total morphine).
12Four rats are used for each experiment.
13The autoclaving of the urine and faeces has been described in the second chapter.
14Four animals are used, for each experiment
15The experiments are carried out with groups of four rats.
16These different morphine contents will be discussed in part four of this chapter.
17Sumwalt in a very comprehensive review studied the resorption and eventual destination of morphine.
18According to Oberst (1941), codeine, which does not produce habituation phenomena, is also excreted in the two forms L and C.
19It should be recalled that resorption is incomplete one hour after the injection in both groups of rats, the quantities found in the injected region being similar in the normal and the habituated rat.
20Rat habituated for seven weeks.
21It should be remembered that in these conditions the resorption of morphine is total after twenty-four hours.
22No food is given to the rats for twenty-four hours before they are killed. In addition the intestine of the normal rat contains no faecal matter twenty-four hours after the administration of morphine, whereas that of the habituated rat does, and when it is fed normally the latter does not excrete faeces until forty-eight or seventy-two hours after the morphine injection.
23A number of writers have begun to study the action of morphine on the intermediary metabolism of the brain, the striated muscle and the enzymatic systems (Gross and Pierce, 1933; Seevers and Shideman, 1941; Shideman and Seevers, 1941 1942; Shideman, 1946; Watts, 1949). It would be premature to attempt to draw conclusions concerning the mechanism of habituation.
ABE (K.) - Tohoku.J.exp. Med.1936, 29, 299-306.
AMSLER (C.) - Arch exp. Path.Pharmakol.1931, 161, 233-46; Wien.Khn.Wochr.1935, I, 815; Arch exp.Path.Pharmak.1937, 185, 263-6.
ANTON (G.) and BIRK (E) - Arch.exp.Path Pharmak.1935, 177, 226-34.
BARBOUR (H. G.), HUNTER (L. G.) and RICKEY (C H.) - J.Pharmaeol.exp.Therap.1929, 36, 251-77.
BARBOUR (H. G.), PORTER (J. A.) and SEELYE (J. M.) - J. Pharmacol.exp.Therap.1939, 65, 332-42.
BERNHEIM, (F.) and BERNHEIM (M. L. C.) - J.Pharmacol. exp.Therap.1936, 4, 427-36; ibidem, 1944, 81, 374-7; ibidem, 1945, 83, 85.
BERTSCHICK (G.) - Arch.exp.Path.Pharmak.1934, 177, 56-59.
BIBERFELD (J.)-Biochem.Ztschr.1916, 77, 283-97.
BIGGAM (A. G.), ARAFA (M. A.) and RAGAB (M. A.) - Lancet, 1, 922. 1932.
BIRK (E.) - Giessen Disser.1933, 21.
BUCKEL (L.) and LEVY (Jeanne)-Anesthesie-Analgesie,1949, 1-18; Communication mars 1948.
CAHEN (R.) - C.R.Soc.Biol.1934, 115, 817-8; ibidem, 1934, 115, 819-20; Th.doct.es sciences, Paris, 1935; C.R.Soc.Biol.1935, 119, 124-6. ibidem, 126-8; ibidem, 1936, 123, 488-90.
CHENG, (C. L.) and MA (W. C.) - Assoc.Trop.Med.1934, 1, 659-73.
CHEU (K. K.) and ROBBINS (E. B.) - J.Am.Pharmacol. Assoc.1944, 33, 80-2.
CLOETTA (M.) - Arch.exp.Path.Pharmak.1903, 50, 452-80.
DE BODO (R. C.) - J.Pharmacol.exp.Therap.1944, 82, 74-85.
DECKERT (W.) - Arch.exp.Path.Pharmak.1936, 180, 656-71; Z. Anal.Chem.1938, 112, 241-57.
DELAVILLE (M.) - C.R.Acad.Sci.1938, 207, 94-6.
DELAVILLE (M.) and RUSSELL (Mlle) - C.R.Soc.Biol.1938, 128, 941-3.
DENYS (Mme A.) and LEVY (Jeanne) - C.R.Soc.Biol.1947, 141, 653-6.
DETRICK (L. E.) and THIENES (C. H.) - Arch.intern. Pharmacody.1941, 66, 130-7.
DODEL (P.), DASTUGUE (C) and BRESSON (A) - CR. Soc.Biol.1939, 132, 267-8.
DONNINI (A.) - Atti acad.fisiocritici Siena,1937, 11, 5, 67-8; ibidem, 68-9. Boll.Soc.ital.biol.sper. 1937, 12, 376-7; idem, 91, 297-309; ibidem, 305-8.
DOWNS (A. W.) and EDDY (N. B.) - J.Lab.Clin.Med.1928, 13, 739-44.
DUCET (G.) and KAHANE (E.) - Bull Soc. Chim.Biol.1946, 28, 794-805.
DUMEZ-J.Am.M.Ass.1919, 72, 1069-72.
EDDY (N. B.) - J.Pharmacol.exp.Therap.1939, 6, 182-201; Sup-plement of Public Health Report. No. 165, p.687-758, 1940.
EMERSON (G. A.) and PHATAK (N. M.) - Un.Calif.Pub.-Pharmacology,1938, 1, 77-92.
ENDO (M.) - Hokkaido Igakusasschi, 1938, 16, 1166-70.
ERCOLI (N.) and LEWIS (M. W.) - J.Pharmacol.exp.Therap.-1945, 84, 301-17.
FAUST (E. S.) - Arch.exp.Path.Pharmak.1900, 44, 217-38.
FAVARGER (P.) and ROSSELET P.) - Arch,intern.Pharma-codynamie, 1944, 69, 382-7.
FAWAZ (G.) - Proc.Soc.Exp.Biol.Med.1948, 68, 262-3.
FICHTENBERG (D. G ) - C.R.Soc.Biol.1939, 130, 316-8; C.R.-Soc.Biol.1946, 140, 282-3; ibidem, 1948, 142, 54-5.
FICHTENBERG (D. G.) and LEVY (Jeanne) - C.R.Soc.Biol.-1939, 130, 312-5.
FITZHUGH (O. G.) - J.Pharmacol exp.Therap.1939, 67, 423-8.
FLEISCHMAN (P.) - Bioch. Ztschr.1931, 241, 233-55.
FROMMEL (I. E.), PIQUET (J.), GUENOD (C. L.), LOUTFI (C. L.) and ARON (J.) - Helv.Physiol.Pharmacol.Acta, 1945, 3, 83-90.
FULTON (C. C.) - Amer.J.Pharmacy, 1933, 105, 503-10.
FURU (F.) - Jikken. Yakubutsukugaku Zasshi, 1937, 14, 231-41.
GAUTRELET (J.) and SCHEINER (H.) - C.R.Soc.Biol.1939, 131, 738-40.
GIORDANO (C. B.) - Arch.intern Pharmacodynamie, 1933, 46, 410-16 and 446-9.
GIRNDT (O.) and LIPSCHITZ (W.) - Arch.exp.Path.Pharmak.-1931, 159, 249-64.
GOETZL (F. R.), BURRIL (D Y) and-IVY (A. C) - J.Pharmacol.exp.Therap.1944, 82, 110-119.
GOLD (H.) - J.Pharmacol.exp.Therap.1929, 35, 355-62
GOTTLIEB (R.) - Munch.med.Woch 1926, 73, 595-6.
GROSS (E. G.) - Proc.Soc.Exp.Biol.Med.1942, 51, 61-3.
GROSS (E. G.) and PIERCE (I. H.) - J.Pharmacol.exp.-Therap.1935, 53, 156-68.
GROSS (E. G.), PLANT (O. H.) and THOMPSON (V.) - J.Pharmacol.exp.Therap.1938, 63, 13.
GROSS (E. G.) and THOMPSON (V.) - J.Pharmacol.exp.-Therap.1940, 68, 413-18.
HAYASHI (A.) - J.of.orient.Med.1934, 20, 28-39; ibidem, 1934, 21, 27-62 and 63-78; ibidem, 1934, 21, 79-80, ibidem, 81-82; ibidem, 83-84.
HERMANN (O.) - Bioch.Ztschr.1912, 39, 216-31.
HILDEBRANDT (F.) - Arch.exp.Path.Pharmak 1922, 92, 68-95.
HIMMELSBACH (C. K.), GERLACH (C. H.) and STANTON (E. J.) - J. Pharmacol.exp.Therap.1935, 53, 179-188.
HINOHARA (S.) - Japan.J.Med.Sci.IV Pharmacol.1937, 10, 195-6.
HOFMANN (R.) and POPOVICI (H.) - Pharm.Zentrahhalle, 1935, 76, 346-8.
HOTTA (S.) - Tokyo Igk.Z.1931, 45, 986-97. Japan.J.Med.Sci.IV Pharmacol.1932, VI, 115-32.
IKESHIMA (S.) - Japan.J.Med.Sci.IV Pharmacol.1934, 8, 91-4.
INGERSOLL (E. H.) - Virgnia Med.Monthly, 1940, 67, 407-10.
INOUE (T.) - Japan J.Med.Sci.IV Pharmacol.1940, 12, 75-77.
ISSEKUTZ (Br.) VARADY (M.) - Arch.exp.Path.Pharmak.-1934, 176, 35-9.
JOEL (E.) and ETTINGER (A.) - Arch.exp.Path.Pharmak.-1926, 115, 334-50.
JUUL (A.) - Arch.intern.Pharmacodynamie, 1939, 62, 69-78.
KAHANE (E.) and LEVY (Jeanne)-C.R.Soc.Biol.1936, 121, 1596-8; ibidem, 1939, 130, 309-12.
KEESER (E.), OELKERS (H. A.) and RAITZ (W.) - Arch.-exp.Path.Pharmak.1933, 173, 622-32.
KEIL (W.) and KLUGE (A.) - Arch.exp.Path.Pharmak.1934, 174, 493-501.
KEIL (W.) and POHLS (F. H.) - Arch.exp.Path.Pharmak.1936, 181, 285 91.
KO (C.) - Japan J.Med.Sci.IV. Pharmacol.1935, 8, 46-9
KO (K.)-Ibidem, 1937, 10, 199 202.
KO (T. R.) - J. Orient.Med.1939, 30, 289; ibidem, 1939, 30, 259-60.
KOBAYASHI (A.) - Japan J.Med.Sci.IV, 1930, 189-201.
KOH (M.)-Transactions Soc. Path. Jap.1933, 23, 480.
KREITMAIR (H.) - Ann de Merk, 1928, 318 23
KUWAHARA (H.)-Japan.J Med.Sci.IV. Pharmacol.1937, 10, 196-9; ibidem, 1938, 11, 141-2.
LEULIER (A.) and DREVON (B.) - C.R.Ac.Sci.1931, 192, 1129-31.
LEULIER (A.) and POMME (B) - Bull.Ac.Med.1930, 104, 105-7.
LEVY (Jeanne)-Bull Soc.Ch.Biol.1934, 16, 631-709; ibidem, 1945, 27, 584-96.
LEVY (Jeanne) and CAHEN (R.) - C.R.Soc.Biol.1933, 112, 167-70.
LEWIS (J. R.)-J. Pharmacol.exp.Therap.1949, 96, 31-7.
LOOFS (F. A.) - Ztschr.neur.Psychiatrie, 1922, 79, 433-62.
MA (W. C.)-Chinese J.Physiol.1932, 6, 359-90; ibidem, 1933, 7, 287-342; ibidem, 1936, 10, 599-636; ibidem, 1937, 12, 427-84; ibidem, 1939, 14, 411-30.
MARIE (A.) - C.R.Soc.Biol.1907, 63, 380-2.
MARME (W.) - Deutsche Med.Woch.1883, 9, 197-8.
MATSCHULAN (G.) - Arch.exp.Path.Pharmak.1937, 187, 230-3.
MIKEL'SON (M. Y.) - Byull.Eksptl Biol Med. URSS, 1941, 11, 230-3.
MILLER (G. H ) and PLANT (O.) - J. Pharm-acol.exp.Therap.1926, 28, 241-9.
MUNCH (J. C.) - J.Am.Pharm.Assoc.1934, 23, 1185-7.
MYERS (H. B.) - J.Pharmacol.exp.Therap.1916, 8, 417-37
MYERS (G. N.) - J.Hyg 1939, 39, 375-404 and 512-28.
OBERST (F. W ) - J.Labor clin.Med.1939, 24, 318-29. J.Pharmacol.exp.Therap 1940, 69, 240-5; ibidem, 1941, 73, 401-4; ibidem, 1942, 74,37-41.
OKAWA (Z.) - Mit.Med.Akad.Kioto, 1940, 28, 1104-5.
PIERCE (I. H.) and PLANT (O. H.) - J.Pharmacol.exp.-Therap.1927, 31, 212.
PIERCE (I. H.) and PLANT (O. H.) - J.Pharmacol.exp.-Therap.1930, 39, 265-6; ibidem, 1932, 46, 201-28.
PLANT (O. H.) and PIERCE (I. H.)-Proc.Amer.J.Pharmacol.exp.-Therap.1927, 13, 210-11.
PLANT (O. H.) and PIERCE (I. R.) - J.Pharmacol.exp.-Therap.1928, 33, 329-57; ibidem, 1933, 48, 284-5; ibidem, 1933, 49, 432-49.
PLANT (O. H.) and SLAUGHTER (D.) - J.Pharmacol.exp.-Therap.1936, 58, 417-27; ibidem, 1938, 62, 106-10.
QUASTEL (J. H.) and TENNENBAUM (M.) - J.Pharmacol.exp.-Therap.1937, 60, 228-233.
QUASTEL (J. H.), TENNENBAUM (M.) and WHEATLEY (A. H. M.) - Biochem.Journ 1936, 30, 1668-81.
RA (H.) Folia Pharmacol Japan 1941, 32, 76-102; ibidem, 1941, 32, 103-58.
RENTZ (E. D.) and KESARBANI (D. N.) - Arch.exp.-Path.Pharmak.1941, 198, 107 113
RO (A.) - Japan J Med.Sci.IV Pharmacol 1934, 8, 99-100.
RUBSAMEN (W.) - Arch exp Path.1908, 59, 227-44.
SAN FILIPO (G) - Arch.ital.Sci Farmacol.1939, 8, 161-72.
SATO (S) - TchokuJ.exp.Med.1940, 38, 205-9.
SCHMITT (C. F.) and LIVINGSTON (A. E) - Amer. J.-Physiol.1929, 90, 506, J.Pharmacol.expTherap.1933, 47, 443-71.
SEEVERS (M. H ) - J Pharmacol.exp.Therap.1934, 51, 141-2.
SEEVERS (M. H ) and SHIDEMAN (F. E.) - J Pharmacol.-exp.Therap.1941, 71, 373 82.
SHEU (P. T.) - J Biochem.1935, 31, 173-95.
SHIDEMAN (F. E.) - J.Pharmacol.exp. Therap.1946, 86, 242-47.
SHIDEMAN (F. E.) and SEEVERS (M. H.) - J. Pharmacol.-exp.Therap.1941, 71, 383 93; ibidem, 1942, 74, 88-94.
SIMONNET (H.) - C.R.Acad.Sci.1937, 204, 1371 2.
SLAUGHTER (D.), TREADWELL (C. R.) and GALES (J. W.) - J Lab. Clin.Med.1943, 28, 1199-202.
STOLNIKOW - Ztschr.Physiol.Chem.1884, 8, 235-71.
STRAUB (W.) - Deutsch.med.Wochenschr.1911, 1462.
SUMWALT (M.)-Suppl.Public Health Reports 1940, 165, 759-811.
SUO (M.) - Folia Pharmacol.Japan 1932, 14, 260-75.
TAKAYANAKI (T.) - Arch.exp.Path.Pharmak.1924, 102, 167-75.
TATUM (A. L.), SEEVERS (M. H ) and COLLINS (K. H.) - J.Pharmacol.exp.Therap.1927, 31, 213-14; ibidem, 1929, 36, 447-75.
TEMPLETON (R. D.), GALAPEAUX (E. A.) and ADLER (H. F.) - Proc.Soc.Exp.Biol.Med.1940, 45, 98-104.
TERADA (B.) and HONDA (M.) - J Orient Med.1935, 22, 677-85.
TERUUCHI (Y.) and KAI (S.) - J Pharmacol.exp.Therap.1927, 31, 177-99.
THOMPSON (V) and GROSS (E. G.) - J Pharmacol.exp.-Therap.1941, 72, 138-145.
TIN (K.) - Folia Pharmacol.Japan.1940, 28, 212-42
TO (S.) - Japan J.Med.Sci.IV Pharmacol 1935, 8, 93-115.
TO (S.) and RIN (A) - Japan J Med IV Pharmacol 1933, 7, 11-15.
TOFT (H. T) - Acta Pharmacol Toxicol.1946, 2, 149-55
VAN DONGEN (K.) - Arch ges Physical 1915, 162, 54-66.
VAN EGMOND (A. J) - Arch.exp.Patch.Pharmak.1911,65, 197-213.
WADA (N.), TANAKA (H.), HIRANO (T.) and taneiti (Y.) - Tohoku J.exp.Med.1938, 34, 52-71.
WATTS (D. T.) - J.Pharmacol.exp Therap.1949, 95, 117-21.
WEGER (P.) and AMSLER (G.) - Arch.exp.Path.Pharmak.1936, 189, 9-12, ibidem, 1936, 181, 489-93.
WILKER (A) and FRANK (K.) - J.Pharmacol.exp.Therap. 1943, 94, 382-400.
WOLFF (N. A.), RINGEL (C.) and FRY (E. G.) - J.Pharma-col.exp.Therap.1933, 47, 391-409.
WRIGHT (C. I.) - Science New-York, 1940, 92, 244 5.
WRIGHT (C. I.) and SABINE (J. C) - J.Pharma-col.exp.Therap.1943, 78, 375-84.
YOKOTA (I. N.) - Japan J.Med Sci IV Pharmacol.1935, 8, 53-6.
YOSIKAWA (N) - Proc.Japan.Pharmacol.Soc.1940, 13, 74-5.
ZWIKKER (J. J. L.) and STEENHAUER (A. J.) - Pharm.-Weekblad, 1942, 79, 241-5.