Introduction
Metabolism and disposition
Disposition
Tissue storage of cannabis derivatives
Active metabolites.
Physical effects of cannabis intoxication
Clinical studies with smoked cannabis extracts or synthetic Δ-9-THC
Clinical studies with oral doses of cannabis or of Δ-9-THC (Hollister)
Relevance of laboratory experiments with cannabis and its actual usage
Interaction of cannabis with other drugs
Tolerance
Physical and psychological dependence: the marihuana habit
Summary
Bibliography
Author: Gabriel G. NAHAS
Pages: 9 to 39
Creation Date: 1973/01/01
All symptoms of cannabis intoxication have been known for centuries. The discovery of Δ-9-THC as the major psychoactive agent in cannabis, as well as the availability of techniques to measure its concentration, has given to the clinical pharmacologist the opportunity to study dose-response relationships of this drug.
* This article is based on a book published by the author.
Another important area which the clinical pharmacologist has investigated is the metabolism and distribution of Δ-9-THC, using tagged material. Analysis of these studies give a better understanding of the mode of action of the drug through the formation of active and inactive metabolites.
The first clinical study with synthetic material was made by Isbell et al. (1967) who showed that the physical and psychological effects of cannabis were related to the dose administered, and confirmed the older observations of Moreau (1845) about the hallucinogenic properties of cannabis. Isbell indeed concluded his study in a straight forward way: "The data in our experiments definitely indicate that the psychotomimetic effects of Δ-9-THC are dependent on dosage and that sufficiently high dosage (8 mg smoked, 30 mg ingested) can cause psychotic reactions in any individual. "Isbell therefore classified cannabis among the hallucinogens.
Two subsequent studies, by Weil (1969) and by Crancer et al. (1969), however, made with unextracted, aged material, containing a putative dose of Δ-9-THC (5 to 66 mg) only produced mild symptoms of intoxication which were not related to the amount of drug administered. The results of these studies indicated that cannabis was a "mild intoxicant" which produced effects not related to dosage, did not impair, and even improved performance of "chronic users". Following this work, a new pharmacological concept was introduced, that of "reverse tolerance".
From then on the great marihuana debate had started in the United States: Is cannabis an hallucinogen? Or is it a mild intoxicant when used in a dosage likely to be taken by habitual users in the population at large?
The difficulties of interpreting clinical studies with Δ-9-THC are due to the many factors which influence the development of cannabis intoxication in man (table I). These factors are the dosage of active material (mostly Δ-9-THC) in the preparation used, the route of intake (whether by inhalation or ingestion), the previous experience of the subject with the drug, the frequency of intake, and the development of tolerance to the drug; individual genetic characteristics pertaining to enzyme induction by Δ-9-THC and the formation of active metabolites are also very important; intake of other drugs which stimulate or inhibit enzyme induction will modify Δ-9-THC metabolism and its pharmacological activity; the mood and expectation of the subject, as well as the setting in which the drug is taken, will influence the psychological response. It is apparent that all of these variables cannot be controlled even in the best clinical experiments.
TABLE I
Factors influencing cannabis intoxication
Dose (amount of Δ-9-THC)
Route of intake:
Inhalation
Ingestion
Frequency of intake (development of tolerance)
Individual metabolism of Δ-9-THC and formation of active metabolites
Associated intake of drugs interacting with Δ-9-THC: alcohol, psychotropic drugs (barbiturates, amphetamines)
Mood of the subject (Expectation)
Setting:
Group (Interpersonal stimulation)
Solitary
When trying to determine the dosage administered to a subject the clinical pharmacologist is faced with a difficult task: He has presently no way to measure the amount of active ingredients actually absorbed in the blood because none of the available methods are sensitive enough. As all preparations are either ingested or smoked, the amounts absorbed by both routes will vary considerably from one subject to the next. And no clinical pharmacologist has yet felt free to administer Δ-9-THC intravenously to man, except in tracer amounts, because of its insolubility in water. But available techniques should allow investigators to measure accurately the amount of active ingredients present in the preparation which they use. With this measurement, imperfect as it is, dose-response relationship may be established and the range of dosage required to produce euphoria, dysphoria and hallucinations established. This range has now been bracketed in a number of studies which will be reviewed: it is close to that determined by Isbell.
Besides actual dosage delivered, the second factor to be considered is the personality of the subject and his previous knowledge or experience with the drug.
Previous cannabis usage by a person will modify the subjective effects produced by the drug either in laboratory or social setting. But many investigators do not or cannot define what they mean by "use ", "habitual use ", "chronic use ", or "experienced use" of the drug. Other investigators define the dosage of cannabis used in an experiment as one which will produce a "typical ", socially acceptable "high ", or a commonly experienced high, or a socially relevant dose. Such definitions are not quantitative enough to be scientifically acceptable. Many authors also speak of high, very high, moderate and low dosages without specifying the actual amount administered.
In Isbell's studies former narcotic addicts or prisonners were used as experimental subjects. They had a thorough previous knowledge of the subjective and intoxicating effects of drugs. Interpretation of the results observed in these subjects was criticized and it was pointed out that such data might not apply to a more representative sample of the population at large. Similar criticisms should also be applied to other studies performed on young paid volunteers recruited from the student community, many of them enthusiastic marihuana smokers who are convinced that cannabis is innocuous and are expecting pleasant effects from its use.
Jones (1971) performed uniques studies on a selected group of marihuana users who smoke relatively mild preparations (0.9 % THC). He was able to, distinguish the effects of the psycho-social factors associated with marihuana smoking from the pharmacologic effects of the drug, or the "effects of the mind on marihuana ".
The problem of tolerance to a drug which might be used habitually is of major importance. Can cannabis, which rapidly creates tolerance in animals, be used frequently by man without having to increase dosage in order to produce the desired effect? Is chronic use of cannabis associated with "reverse tolerance" and if so, by what mechanism? What about physical and psychological dependence? Investigators in clinical pharmacology have to attempt to answer all of these questions, which will sometime require critical comparison of present measurements with past observations.
Most significant studies were performed with tagged Δ-9-THC administered intravenously (Lemberger 1970, 1971). Results obtained confirmed those in animals: Δ-9-THC gives rise to nonpolar metabolites which are slowly eliminated from the body (fig. 1).
FIGURE 1
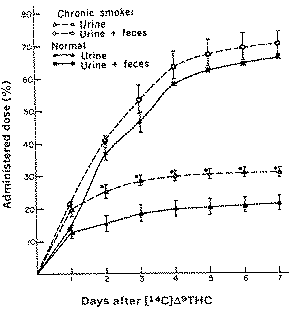
Comparison of the cumulative excretion of radioactivity in three chronic marihuana users and three non-users after intravenous injection of 14C-r 9-THC (Lemberger, 1971).
After intravenous administration of tracer amounts of C 14-Δ-9-THC to subjects who had never previously used cannabis extracts, the amount of this compound in plasma declines rapidly during the first hour, with a half-life of about 30 minutes. After one hour, the amount of Δ-9-THC falls much more slowly (with a half-life of 56 hours). The decline of total radioactivity and of the more polar ether extractable metabolites in plasma is similar to that of Δ-9-THC. A rapid initial decline precedes a much slower phase of disappearance from the plasma. Polar metabolites are formed rapidly and are present in plasma at higher concentrations than Δ-9-THC. The initially rapid decrease of C 14-Δ-9-THC in plasma represents redistribution of Δ-9-THC. from the intravascular compartment into tissues (including brain) and metabolism. These findings are consistent with the reported clinical effects of a small dose of inhaled marihuana (containing approximately 5 mg or less of Δ-9-THC) which are maximum within 15 minutes, diminished between 30 minutes and one hour, and largely dissipated by three hours. The slower decline of Δ-9-THC in plasma ( t1/2 56 hours) and of total radioactivity ( t1/2 67 hours) presumably represents retention and slow release of the drug from the tissues. Since Δ-9-THC is a nonpolar compound, it may accumulate in fat or other tissues such as lung which have an affinity for the drug, and it has already been reported that after intravenous administration of Δ-9-THC to animals, much higher levels of radioactivity were present in the lung than in other tissues.
Over a period of more than a week about 30% of the administered radioactivity is excreted in the urine and 50% in the faeces. The finding that Δ-9-THC and its metabolites persist in man for long periods indicates that the drug and its metabolites will accumulate in tissues when administered repeatedly. In chronic users, the half life of Δ-9-THC in blood plasma is 28 hours (instead of 57 hours for non-users). Apparent volume of distribution is similar in both groups. Chronic users will eliminate significantly more polar metabolites in the urine and less in the faeces than non-users. The total amount of metabolites eliminated in both groups is the same and requires more than one week. The more rapid urinary elimination of Δ-9-THC metabolites in chronic users gives a biochemical basis to the development of tolerance, which accompanies cannabis usage.
One of the deceptive aspects of cannabis derivatives is that long after their psychoactive effect has been dissipated, their polar metabolites are stored in body tissues for as long as a week.
Repeated administration of cannabis preparations at less than a week's interval will result in the accumulation of metabolites in tissues, including brain. Long-term effects of a chronic accumulation of non-polar metabolites will have to be appraised with special attention to memory, affectivity and performance of complex tasks. Possible relationship between this accumulation and the development of the "amotivational syndrome" should be investigated.
The possible adverse effects resulting from tissue storage of the metabolites of cannabis derivatives are now well recognized. As a result, the Food and Drug Administration (1971) has formulated the following regulations regarding the use of cannabis for clinical investigation in man:
Oral doses of the extract, Δ-8- or Δ-9-THC may be given daily for a period not to exceed one week. Parenteral doses or prolonged oral doses may not be administered to human subjects.
Inhalation studies involving the smoking of the standard extract of marihuana, Δ-8 or Δ-9-THC may be performed for short periods, i.e., 3 days, and repeated after a washout period of 3 days.
Studies utilizing the smoking of whole plant material may be performed for a period up to one month.
If such regulations will permit to rapidly assess the acute effect of known amounts of the active ingredient of cannabis, they preclude the performance of chronic studies in the foreseeable future.
While nonpsychoactive polar metabolites remain in tissues, and are slowly excreted in urine and faeces, there is evidence that psychoactive metabolites of Δ-9 and Δ-8-THC might also be formed (Truitt 1970, 1971; Nakazawa and Costa, 1971). They are the 11 hydroxy THC compounds.
The production of psychoactive metabolites of Δ-9-THC by enzyme induction in liver and lung could account for the elayed appearance in many subjects of their first recorded manifestations of cz\1annabis intoxication. Few effects are reported when the drug is taken or smoked for the first time. They only appear after a subsequent intake, and the same dosage as taken the first time is accompanied by greater effects the second or third time. However, this so-called "reverse tolerance" is of brief duration with continued intake of cannabis. Metabolic tolerance will continue to develop until enzyme induction has reached its maximum levels; in addition functional brain tissue tolerance to the active metabolites of Δ-9-THC will also develop. The resulting effects of these two processes will be that increases in drug intake will be required to obtain the same effects.

Plasma levels of 14C-r 9-THC, total radioactivity and ether-extractable radioactivity after the oral administration of 0.3 mg/kg of r 9-THC with 0.5 mg of 14C-r 9-THC to a chronic cannabis user. Blood samples were drawn at various times and plasma assayed for r 9-THC, total radioactivity and ether extractable radioactivity (Lemberger, 1971).
On the upper portion, the time course for the psychic effects of r 9-THC after its oral administration (0.3 mg/kg) is described (From Hollister et al., Clin. Pharmacol. Ther. 9:783, 1968).
The possible participation of active metabolites of Δ-9-THC in the development of the psychic effects of cannabis intoxication is supported by observations of Lemberger et al. (1971). They administered orally to a chronic user, tracer doses of C 14 tagged Δ-9-THC along with a pharmacological carrier dose (0.3 mg/kg). Very little Δ-9-THC were present in the plasma throughout the ten-hour observation period, while large amounts of polar metabolites including 11 hydroxy THC compounds were found. Their plasma concentration correlated well with the time course of psychic effects of orally administered Δ-9-THC as reported by Hollister (1968) (fig. 2).
The exact contribution of all of these metabolites to the multiple psychological and behavioural effects and after-effects of cannabis intoxication will be very difficult to assess. Nonspecific enzymes (oxidases) in the microsomal fraction of the cell are induced rapidly, in vivo and in vitro, to form these metabolites (Truitt, 1970). It is known that these same liver enzymes can be induced to higher rates of activity (initial methylation or hydroxylation steps) by repeated usage of many other drugs which include barbiturates, antidepressants, tranquillizers, analgesics, and anticoagulants. Chronic administration of the drugs produces a metabolic tolerance due to an induction of an increased activity of the enzymes. All of the drugs will interact with Δ-9-THC and might alter its bio-transformation and activity (fig. 3).

Schematic representation of the multiple effects of cannabis smoking on basic enzymatic and physiological mechanisms. These effects are mediated by r 9-THC and possibly by active metabolites, and lead to the development of functional and metabolic tolerance.
Moreau described the various dosage-related physical symptoms of cannabis intoxication and concluded that the physical disorders, which develop later than the mental disturbances, relate to the nervous system. These observations were confirmed one hundred years later when investigators were able to measure the physical changes produced by cannabis intoxication.
Such changes were studied systematically by experts appointed by Mayor La Guardia of New York City. Present day clinical recordings consistently report increased heart rate and dilation of conjunctival blood vessels; these changes are not accompanied by any specific biochemical alterations in body fluids identifiable by present techniques.
Cannabis derivatives acutely administered to Western smokers cause a significant increase in heart rate. The degree of tachycardia which is related to the dose of Δ-9-THC absorbed reaches a maximum 30 minutes after smoking and persists for more

Dose response increase in heart rate following increasing doses of smoked marihuana (Renault, 1971). than 40 minutes. In one study 65 % of the increase in heart rate observed could be associated with the concentration of Δ-9-THC in the cigarette (Johnson and Domino, 1971). Linear dose response curves were obtained on the heart rate of subjects smoking marihuana cigarettes containing 1 to 6.5 mg. of Δ-9-THC. Standardized Smoke Administration was delivered by means of a spirometer (fig. 4). A reproducible dose effect was observed in individual subjects, while variance between subjects in their heart rate response to marihuana inhalation was great. No difference was found between experienced and inexperienced smokers. Marihuana smoking suppressed the normal sinus arrythmus as well as the bradycardia associated with the Valsalva manoeuvre. With the highest Δ-9-THC concentration (6.5 mg.) maximum heart rates were in the range of 140 to 160/min. (Renault, 1971).
This increase in pulse parallels the intensity of the subjective effects. If the drug induces anxiety, a marked increase in heart rate occurs-while if it induces somnolence or sedation, a moderate rise is observed.
In the study by Johnson and Domino (1971) changes in electrocardiogram were minimal but premature ventricular contractions were observed in 2 of 15 subjects who smoked cigarettes containing 10 mg. or more of Δ-9-THC. Allentuck (1941) also reported in a few instances "a temperature sinus tachycardia or sinus bradycardia". By contrast, Isbell and Hollister did not report any arrythmias occurring in the subjects they studied. In view of the high incidence of acute cardiac pathology in the United States, a systematic evaluation of the effects of cannabis intoxication on the heart is required.
Changes in blood pressure have also been reported following cannabis intoxication. Reports are conflicting. Isbell et al. (1967), who studied prisoners, experienced marihuana users, given orally, 10 to 30 mg. of Δ-9-THC, reported no change or a decrease in blood pressure. So did Hollister (1971) who studied student volunteers given larger doses (30 to 70 mg. Δ-9-THC). With the higher dose, two of the subjects developed orthostatic hypotension. By contrast, Johnson and Domino report a significant rise in systolic and diastolic blood pressures when doses greater than 10 mg. Δ-9-THC were inhaled. Allentuck in the La Guardia Report also states that "the increase in pulse rate was usually accompanied by a rise in blood pressure", and Williams et al. (1946) in his study of 17 subjects smoking marihuana ad libitum observed a slightly increased blood pressure. The Significance of these conflicting reports is not clear.
Conjunctival blood vessel congestion is one of the most constant recognizable signs following marihuana smoking. This congestion is related to the dose of the drug; it lasts longer than the increase in heart rate and is still apparent 90 minutes after the end of smoking, but subsides in the following 24 hours. The mechanism of action is not known; it is not related to an irritation from smoke of the cigarette, but rather to a direct action of Δ-9-THC on the conjunctival vessels. An active congestion of the transverse ciliary vessels has been observed in India, among chronic users of cannabis (ganja). It is accompanied by a yellow discoloration of the conjunctiva due to deposition of a yellow pigment around the vessels, and is reported to still be present years after the drug was withheld (Chopra, 1969).
Unlike LSD which produces a hypereffectivity, cannabis intoxication does not alter deep tendon patellar or Achilles reflexes. In contradiction of earlier reports, pupil diameter is not changed following cannabis intoxication. There is a muscle weakness which can be objectively measured by the ergograph test (Hollister, 1969). Ptosis of the eyelids is also observed (Domino, 1971), as well as an impairment of body and hand steadiness. The ataxia is in general in all directions rather than predominant in any particular axis; it can be ascribed to both the central and peripheral nervous system effects of cannabis, which acts on the cerebellum and on the neuro-muscular function.
Nausea with vasomotor imbalance and vomiting is often reported by inexperienced subjects using cannabis extracts. Dryness of the mouth and naso-pharyngeal mucosa is usually reported by the cannabis smoker. This symptom might not be entirely due to the irritation of the smoking process and has been related to an atropine-like substance present in the smoke, which could cause a decrease in salivary flow (Gill et al. 1970).
Hepler and Frank (1971) reported that 9 of 11 subjects, after smoking 2 gm of marihuana containing 9% Δ-9-THC in a water pipe, presented a significant decrease in intraocular pressure.
These changes observed in occasional users smoking cigarettes containing a putative dose of 7.5-22.5 mg. Δ-9-THC are not very pronounced; they consist of an increase in per cent time of alpha band and an associated reduction in theta and beta bands. However, another chronic experiment performed by these same authors showed that marihuana smoked for 10 to 22 days by four previous heroin addicts produced dysphoria and EEG synchronization (Volavka, 1971).
Tart and Crawford (1970) report the effects of marihuana intoxication in sleep patterns in 150 experienced smokers. They report that moderate levels of marihuana intoxication have a sedative effect, but high level may overstimulate, ward off drowsiness and make sleep poorer.
Many subjects after cannabis usage have reported an increased appetite especially for sweets. This subjective symptom is not related to changes in blood sugar levels which remain constant at times when the cardiovascular and psychological effects of the drug are maximal. However, an abnormal glucose tolerance test has been reported in "chronic" marihuana smokers.
In studies where a single dose of 15-70 mg. of Δ-9-THC was ingested there were no changes in plasma cortisol levels, platelet serotonin, blood chemistry and hemotology, or urinary catecholamines excretion. Creatinine and phosphorous clearance were temporarily decreased, a phenomenon which has been observed with LSD (Hollister 1968).
Smoking is a common method of including cannabis intoxication, and it is the method chosen by most marihuana users because a swift effect can be obtained with a small amount of the substance.
The amount of active material absorbed in the blood stream will be, in part, a function of the method of smoking. Therefore, the experience of the smoker, and his motivation to obtain the desired effect will influence the amount of inhaled smoke which will be stored in the lungs. The amount of inhaled smoke may vary considerably: from 15 to 100 ml per inhalation. In order to control this variable, it is important to measure tidal volume as well as amount of smoke inhaled and breath-holding time. Even so, different subjects will not have the same efficiency in delivering the active ingredients contained in their cigarette to their pulmonary vessels. It is therefore difficult to gauge quantitatively the physiological and psychological effects of the same amount of cannabis smoked by different subjects.
The relationship between the active constituents of the cannabis cigarette and the chemical by-products which may be produced in the burning process of smoking (pyrolysis) is not elucidated. The experiments performed by Manno et al. (1970) and by Truitt (1971), indicate that after insuring complete smoking of the cigarette end approximately 50% of the Δ-9-THC originally present in the cigarette is delivered unchanged in the smoke (fig. 5). Assuming that mainstream smoke (which is inhaled) accounts for 90 % of the smoke produced by the burning cigarette, the maximum efficiency of delivery of Δ-9-THC by an experienced smoker would be 45%, providing the entire cigarette is burned.
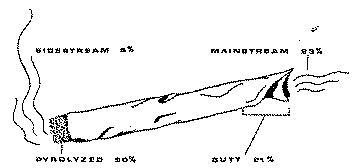
If the cigarette is not entirely consumed, as much as 50% of THC may accumulate in the unburned end or "roach". In this case, the efficiency of transfer of Δ-9-THC will be further decreased to 22%. Since half of the Δ-9-THC may remain in the butt of a cannabis cigarette, it is important in clinical experiments that all smokers consume their cigarettes to the same length. Otherwise the remaining butts should be weighed and the Δ-9-THC measured and substracted from the original amount present in the cigarette.
Some investigators have compared, in double-blind experiments, the effects of smoking cannabis extracts of different potency in experienced and novice subjects with the effects produced by a placebo. Chronic cannabis smokers claim they are able to identify the cigarette containing psychoactive ingredients. It has been proved that this is not the case, especially when the cigarettes contain small doses of toxic material (1% Δ-9-THC or less) (Jones, 1971). The characteristic smell of burning cannabis is not related to its active cannabinoids, and cannot be an index of potency.
It is generally agreed that Δ-9-THC is three to four times more potent when smoked than when taken orally. Smoking produces, within a few minutes, effects which will last from 1 to 4 hours. Oral dosage will only be felt after 1 hour and last for 6 to 8 hours. The reasons for the greater potency of cannabis when smoked as compared to a preparation orally administered are not clear.
That Δ-9 THC may possibly induce active metabolites in the lung might be a reason for the greater activity of smoked marihuana. The by-products of ingested cannabis first go through the liver where some may be inactivated. The exact mechanisms of action of ingested and smoked cannabis will only be clarified after completion of tracer studies.
The amount of active ingredients transferred through smoking cannabis will therefore depend upon many factors which include the method of smoking, the expertise of the smoker, the amount of mainstream smoke which can be trapped in the lung, the amount of side stream smoke, and the amount of psychoactive substance trapped in the unburned cigarette end. It is very difficult to keep all of these factors uniform from one subject to the next. A significant number of clinical studies did not control these important variables. Consequently the reported results are misleading (table II).
The first laboratory study made by Well and associates on smokers using marihuana cigarettes containing putative dosage of 4.5 to 18 mg. Δ-9-THC, does illustrate the uncertainties inherent in clinical investigations performed with cannabis of uncertain potency. In this study non-users experienced few subjective effects, demonstrated impaired performance on simple intellectual and psychomotor tests, moderate acceleration of heart rate (not dose related) and injection of the conjunctivae. "Experienced users" presented increases in heart rate higher than those observed in non-users and not dose related, reported a subjective" high" and a" slight improvement of their performance" (pursuit rotor test and digit-symbol substitution test). On the basis of these observations Well et al. concluded that "marihuana is a relatively mild intoxicant ", a view which can only be shared by all those who read their report. And readers were many since Well's paper was published in Science (1968), extensively quoted in Grinspoon's article on Marihuana published in Scientific American (1969) and the subject of a feature article on the front page of the New York Times. This paper certainly did contribute to the widely held belief in the United States that marihuana is a mild intoxicant with little untoward effects.
However, it is now apparent that the dose of psychoactive material absorbed in this study must have been quite low. All of the subsequent studies in which Δ-9-THC was actually measured, indicate that dosages similar to those used by Weil produced much more significant impairment in psychomotor performance and much more important dose related increases in heart rate. On the basis of the experience of another investigator who used unextracted natural material of a similar age, the reported dose of Δ-9-THC utilized by Weil might have been seven times lower than actually assumed. However, in the discussions of his results, Weil et al. do not mention at any time that the dosage used might have been lower than assumed in spite of the fact that they did not observe a dose response increase in heart rate. It is clear from Weil's study that a scientist should not interpret as a reliable index of cannabis intoxication the subjective feeling of a person who claims he is experiencing a "normal social marihuana high".
|
Heart rate |
Psychomotor performance |
|||||||
|---|---|---|---|---|---|---|---|---|
|
Authors |
Subjects |
Dose Δ-9-THC (MG.) in cigarettes |
Average increase (beat/min) |
Time |
Duration |
Dose dependence |
Change |
Dose dependence |
|
Weil
et al.
|
"Naïve"
|
4.5 (alleged)
|
16 |
15'
|
>90'
|
No
|
impaired
[
a]
|
Yes
|
| (1968) |
18.0 "
|
16 |
15'
|
<90'
|
impaired
[
a]
|
|||
|
"Chronic"
|
4.5 "
|
32 |
15'
|
>90'
|
No
|
unchanged
[
a]
|
No
|
|
|
18.0 "
|
-
|
-
|
-
|
unchanged
[
a]
|
||||
|
Crancer
|
"Naïve"
|
22.0 (alleged)
|
?
|
?
|
?
|
?
|
not changed
[
b]
|
-
|
| (1969) |
"Chronic"
|
22.0 "
|
?
|
?
|
?
|
No
|
not changed
[
b]
|
No
|
|
66.0 "
|
?
|
?
|
?
|
not changed
[
b]
|
||||
|
Isbell
|
"Experienced"
|
5.0 | 40 |
60'
|
>4 hrs.
|
Yes
|
impaired
[
c]
|
Yes
|
| (1969) | 15.0 | 65 |
60'
|
>4 hrs.
|
impaired
[
c]
|
|||
|
Manno
et al.
|
"Experienced + Naive"
|
5.0 | 16 |
20'
|
>60'
|
Yes
|
impaired
[
d]
|
Yes
|
| (1970) | 10.0 | 36 |
20'
|
>60'
|
impaired
[
d]
|
|||
|
Johnson and
|
"Experienced"
|
1.5- 5.0
|
10-20
|
5'
|
>90'
|
Yes
|
not changed
[
e]
|
-
|
|
Domino
|
10.0-30.0
|
40-60
|
20'
|
>120'
|
not performed
|
|||
| (1971) | ||||||||
|
Dornbush
et al
|
"Experienced"
|
7.5 |
3-5
|
immediate
|
20'
|
Yes
|
not changed
[
f]
|
Yes
|
| (1971) | 22.5 |
18-26
|
immediate
|
60'
|
impaired
[
f]
|
|||
|
Renault
et al
|
"Inexperienced +
|
|||||||
| (1971) |
Experienced"
|
1.87 |
7
*
|
10-20'
|
>60'
|
|||
| 3.75 |
15
*
|
10-20
|
>60'
|
Yes
|
not performed
|
-
|
||
| 6.50 |
22
*
|
10-20'
|
>60'
|
|||||
|
Jones
|
"Frequent"
|
9.0 | 17.3 |
30'
|
-
|
-
|
not impaired
[
g]
|
-
|
| (1971) |
"Unfrequent"
|
9.0 | 32.0 |
30'
|
-
|
-
|
impaired
[
g]
|
-
|
Assumimg resting heart rate of 70/min.
a. Pursuit rotor performance.
b. Driving simulation.
c. Questionnaire.
d. Pursuit meter.
e. Auditory and visual threshold.
f. Reaction time, short-term memory
g. Digit Symbol substition, complex reaction time
A similar criticism can be made of the simulated driving study of Crancer, et al. (1969), also published in Science and partially quoted in Scientific American. Driving skills of subjects were tested with a driving simulator after they had consumed enough alcohol to approximate concentrations of 100 mg/100 ml plasma or smoked two marihuana cigarettes containing putative doses of 22 mg Δ-9-THC. Such a dose, in the careful studies of Isbell reported in 1967 two years earlier produced, when "smoked", hallucinations, depersonalization and derealization. Furthermore, all of the subjects in the study by Crancer were favorably disposed towards marihuana and some might have been tolerant to its effects while having a bias against alcohol. Under conditions of "marihuana intoxication", speedometer errors were increased (the subjects did not carefully watch the speedometer) but otherwise, driving was not impaired. By contrast, profound impairment was observed with the large doses of alcohol administered (equivalent to 1.5 to 2 ml of 95 % ethanol per kg). Crancer and associates also tested four inexperienced users who showed either no change or negligible improvement in their scores. They gave to four "habitual users" three times the dose used in their first experiment (66 mg Δ-9-THC) and these subjects did not show any significant driving impairment. Crancer concluded "that impairment in simulated driving performance is not a function of increased marihuana dosage or inexperience with the drug".
He does not discuss the discrepancy between his study and that of Isbell. However, he is careful not to state that the use of marihuana will not impair actual driving on the road or that it's use is safer than alcohol. But some of the readers of his paper were less cautious. (Grinspoon, 1969; Kaplan, 1971; Schofield, 1971.)
The 1971 N.I.M.H. Report to Congress on Marihuana and Health also reports uncritically the Crancer study and concludes "the legal level of alcohol intoxication (used in the study) is probably higher than typical levels of social use of alcohol. By contrast, the use of marihuana used in this research may have more closely approximated a typical level of social marihuana use". If the authors of the N.I.M.H. report are correct, marihuana used socially in the United States is more closely related to lawn grass than to the drug-type of cannabis sativa which has been used throughout the centuries for its intoxicating properties.
Such observations contrasted with those of Isbell's, who reported that smoking cigarettes containing 16 mg Δ-9-THC was accompanied by hallucinations and in one instance, by psychotic episodes. No such symptoms were reported by Weil who used a putative dose of 18 mg or by Crancer, who used an alleged dosage of 22 to 66 mg Δ-9-THC. It would appear that somebody must be wrong.
Isbell used Δ-9-THC which had been prepared and assayed by Korte from the Institute of Organic Chemistry of Bonn University in Germany. Forty healthy former opiate addicts serving prison sentences and abstinent from all drugs were studied. In addition to physical changes, (such as rectal temperature, pulse rate, blood pressure, pupillary diameter), subjective effects were evaluated by means of a questionnaire which permits assessment of personality and mood alterations typically considered as important determinants of drug effects. Thirty questions were selected from the "general drug", "marihuana" and LSD scales of the Addiction Research Inventory, while the remaining 33 questions dealt with alterations in body image, illusions, delusions, hallucinations and were designated as the psychotomimetic scale. A dose of 0.05 mg/kg Δ-9-THC smoked or 0.12 mg/kg ingested produced euphoria as well as time sense and perceptual changes. A dose of 0.2 to 0.25 mg/kg smoked or 0.3 to 0.48 mg/kg ingested was accompanied by marked distortion in visual and auditory perception, derealization, depersonalization and hallucinations. Isbell also established that for each subject physical changes (increase in pulse rate) and psychological changes were dose dependent. In a subsequent study he compared Δ-9-THC (15 to 225 mcg/kg smoked) and LSD (0.5 to 1.5 mcg intramuscular) in the same eight subjects. While the objective effects of both drugs were markedly different, the subjective effects could not be readily distinguished by using the special drug scales developed at the Addiction Research Center. Two patients out of 10 withdrew after experiencing psychotic reactions following THC. Both drugs were psychotomimetics-LSD was 160 times as potent as THC on the psychomimetic scale and 150 times as potent as THC on the general drug scale. But patients tolerant to LSD were not cross tolerant to THC, indicating that the mental effects of the two drugs are mediated by different mechanisms.
The pioneering observations of Isbell on the adverse effects of Δ-9-THC (10 mg smoked) on mental performance were substantiated by subsequent well-controlled studies performed by Forney's group. They compared the effect of placebo cigarettes with those containing marihuana extracts containing 10 mg Δ-9-THC, THC being assayed by the investigators themselves with the use of gas chromatography. These authors had demonstrated in previous studies that a cigarette containing 10 mg Δ-9-THC, smoked with maximum efficiency will deliver to the subject five mg. Cannabis impaired performance significantly on a pursuit meter as well as 5 of 9 performance tests done under conditions of delayed auditory feedback. Many more subjective symptoms were reported by those who smoked cannabis than those who smoked the placebo cigarettes and all were able to identify the active cigarettes without error. However, half of the group also reported that the placebo cigarettes were active, which confirms the studies of Jones and the unreliability of subjective identification of cannabis intoxication induced by smoking, because of the associated sensory perceptions which favour psychological conditioning. It is of interest to note that it was not possible to detect any cannabinols in the blood or urine of the subjects who smoked the cannabis.
A subsequent study made by the same group did cast still more uncertainty on the validity of the results reported by Weil et al. (1968) and Crancer et al. (1969). Twelve volunteers smoked placebo or marihuana cigarettes calibrated to deliver 2.5 or 5 mg Δ-9-THC which amounts to the same or to half the putative doses used by Weil, and one-quarter to one-sixth of the putative dose used by Crancer. In the course of the experiments the subjects were also given fruit juice or ethanol so as to produce concentration of 50 mg per 100 ml of plasma. All subjects who smoked cannabis presented a significant decrease in motor and mental performance which was equal to or greater than the dose produced by the alcohol. Alcohol produced an additive effect on performance impairment.
Hollister, a clinical pharmacologist specialized in the study of marihuana, commenting on the Crancer study, has this to say. "We simply asked our subjects when they were high (on marihuana) ' Do you think you could drive a car? ' Without exception the answer from those who really were high was ' no ' or ' you must be kidding '.
In another study ten experienced marihuana smokers who smoked 2 to 3 cigarettes each containing an alleged dosage of 3.9 mg of Δ-9-THC had only minimal effects (jocularity, loosening of associations). When the material smoked was reanalysed, a tenfold decrease of its original potency was recorded.
A careful analysis of the studies performed on marihuana smokers illustrates the uncertainties inherent in smoking of the unextracted cannabis weed available in the United States. A great deal of this weed contains little Δ-9-THC or rapidly loses its potency if not properly stored. The deceptive aspect of many of the studies (table II) conducted with smoked marihuana is now quite apparent because the following factors were not ascertained: (1) the amount of active ingredients in the cigarette; (2) the fraction of the psychoactive material absorbed by the subject; (3) the extent of the placebo effect or psychological conditioning produced in " habitual smokers " by the smoking process. Only double-blind studies performed with carefully titrated extracted material undistinguishable from a placebo gave an opportunity to distinguish drug effect from psychological and emotional learned response. Such studies were performed by Jones, 1971.
Jones has attempted to distinguish the effects of the psychological and emotional conditioning associated with marihuana smoking from the pharmacologic effects of the drug. His studies were performed over several years on a large sample of young marihuana smokers from the San Francisco area.
One hundred paid student volunteers from the University of California who had smoked marihuana intermittently or daily for 3 years were given a 1 gram marihuana cigarette containing 0.9 % Δ-9-THC, or a placebo. The amount absorbed by each individual smoker could not be assessed, and was probably quite different from one to the other, but could not have been greater than 4 to 5 mg (50% of the amount present in the cigarette). The subjects were asked to rate the subjective estimation of their intoxication on a scale from 0 to 100. The mean rating of the group was 61 for marihuana and 34 for the placebo-but there was considerable overlap in individual ratings. Placebo was rated 0 to 90, cannabis from 0 to 95. Many subjects estimate that placebo and marihuana cigarettes were equal in potency. This was especially true for the frequent users of cannabis (more than one daily cigarette) who had developed a tolerance to the drug and rated placebo 52 and marihuana 48. For these " experienced, sophisticated users of cannabis ", the olfactory, oral and upper airway, sensory perceptions associated with smoking were sufficient to recapture the euphoric sensations they had felt in previous intoxications.
By contrast, infrequent smokers (less than 2 cigarettes a month), rated the marihuana significantly higher than the placebo. This well documented observation is at variance with the new cannabis folklore entertained by the " marihuana smokers " (Goode 1970), who claim that the experienced smoker is able to judge the intoxicating quality of the grass. " The greater the amount of experience with the drug, " says Goode, " the less likely it is that the subject has experienced either no reaction or nothing but a placebo reaction. In fact, the likelihood that a given person who has smoked marihuana more than a dozen times thinks that he has been high without actually experiencing what a truly experienced user would call a high, is practically nil. " This possibility is far from nil, as shown by the studies of Jones. The frequent users rated on the average the effects of placebo and marihuana cigarettes alike. As Jones says, " There may be a credibility gap in the marihuana culture. " The marihuana smoker makes the same misjudgement of psychotropic drug effects frequently made by consumers and professionals alike (Lennard et al. 1971).
The importance of psychological conditioning of frequent marihuana smokers was further illustrated by the fact that they gave a low rating to Δ-9-THC (25 mg) orally administered. When these frequent users of cannabis did not experience the familiar oral and nasal sensory perceptions associated with smoking, they were unable to re-capture a euphoric state of mind, and their physical or pharmacological tolerance to cannabis became apparent. Many of these students had developed physical tolerance to cannabis without knowing it which is a good exercise in self-deception. Instead of increased sensitivity to cannabis, the data of Jones suggest tolerance to the physiologic and psychological effects of marihuana in the frequent users. Pulse rate increase was significantly smaller, decrease in salivary flow was less marked in the frequent users than in infrequent users. Psychomotor performance as measured by complex reaction time and digit symbol substitution was significantly impaired solely in occasional smokers. Jones also demonstrated that marihuana smokers experienced greater euphoria and less dysphoria when they smoked in groups than when they smoked alone. Jones concludes, " These data suggest that marihuana, when smoked at ' socially relevant doses ' produces a level of intoxication that allows the attitude of the subject, his set and expectations, the setting and his past experience to interact in a complex way to determine how the subjective state will be labelled and reported. Many people have uncritically accepted the belief that the drug has specific effects on behaviour and experience, and that these can be readily identified. Although at high doses such a model may be valid, at the doses most youthful drug users are discussing, there is ample evidence that the effects of psychoactive drugs on behaviour and experience are often independent of the drugs' pharmacologic effects. "
The pattern of response of the smokers studied by Jones is consistent with the model where the smoker may obtain intermittent response from Δ-9-THC, but where much of the behaviour and subsequent response is maintained by conditioned reinforcement, " such as the ritual of ' lighting up ' in group, and the associated stimuli of smell, taste and visual perception. "
Jones criticizes the investigators who depend upon a subjective response to gauge cannabis intoxication. " They do so at the risk of studying behaviour in a non-specific psychological state rather than the pharmacological action of marihuana." Many physicians will agree on this point.
But Jones claims that the researcher must also attempt to " quantify the effect of interpersonal stimulation and the effects of subject expectation on the pharmacological action of cannabis ". By doing so, the researcher will be able to relate in "a meaningful way" the pharmacologic effect of cannabis to a given subjective state. Such complex investigations are of great interest from a scientific viewpoint, and will require formidable academic talent and financial outlay. But one fails to see how such studies will answer the basic question asked to the physician: Does cannabis represent a hazard to the health of man and especially of growing man?
The criterion used by Jones to define marihuana dosage is somewhat fragile, when he speaks of a " socially relevant dose ". How can such a dose be defined in terms of the present unstable student milieu? Jones criticizes the investigators who interpret experimental data on the basis of subjective response of the cannabis smoker, but he accepts the criterion of the "socially relevant dose" which is a still more uncertain yardstick.
Weil's studies which were so widely heralded by the lay and scientific press to indicate that cannabis was a mild intoxicant, were done with plant material which seemed to contain a subthreshold dosage of intoxicating substance. The dose of Δ-9-THC in the cannabis used in the studies reporting minimal psychological effects was either assayed in the laboratory by ultraviolet absorption spectrophotometry or by the National Institute of Mental Health (N.I.M.H.). It is now established that it is not possible to separate quantitatively various cannabinoids present in cannabis by ultraviolet absorption spectrophotometry. Furthermore, discrepancies have been reported between assays performed first at the N.I.M.H. with gas chromotography and subsequently at private laboratories (samples of marihuana allegedly containing 1.3% Δ-9-THC were found to have 0.2%). These discrepancies might be due to spontaneous inactivation in the crude material of Δ-9-THC during transport or storage.
In the light of the above one wonders why many investigators still fail to recognize the methodological shortcomings of the early studies of Weil and Crancer (Snyder, 1971).
All of the recent studies performed on volunteers given oral doses of cannabis material assayed for Δ-9-THC content, or known amounts of synthetic Δ-9-THC yield more uniform results than the studies performed with smoked material. For one thing, the psychological conditioning which affects marihuana smokers is not operative.
The findings of the first study performed by Isbell on former narcotic addicts serving prison sentences have already been mentioned. They have been in the main largely confirmed by other groups who used in addition to synthetic THC crude marihuana extracts, carefully assayed for THC content. Hollister (1969) used a group of student volunteers who ingested doses of 30 to 70 mg THC which corresponds to about twice the dosage used by Isbell. Perceptual and psychic changes reported by the subjects indicated more pronounced euphoria than experienced by the prisoners studied by Isbell in a correction institution. Sleepiness was more consistently observed, with deep sleep following the higher dose. Time sense was altered, hearing was less discriminate, and visual distortion abundant. Depersonalization, difficulty in concentrating and thinking were predominant. Many of the symptoms produced were like those elicited by the psychotomimetic LSD, mescaline, psilocytin, but Hollister observed fewer of such effects than Isbell did. Hollister also studied the effect of synhexyl, a synthetic Δ-8-THC homologue, which was extensively studied for possible clinical application. Similar effects were reported with this compound, but a three times greater dosage was required.
In a study performed by a group from the N.I.M.H. 20 mg of Δ-9-THC (putative dosage) was administered to 32 prisoners, paid volunteers who were ignorant of the nature of the drug under study, and were studied in a neutral setting (Waskow 1970). These subjects presented very mild physiological changes, with increases in heart rate much smaller than those reported by Isbell for similar or even lower dosage which raises the question of the real potency of the drug administered. They felt nevertheless, "considerable somatic discomfort, dizziness, weirdness, dream-like state, visual changes, alteration in time, sense and cognitive impairment." The feeling of euphoria, though present in some subjects, was not predominant.
In the study of Hollister, repetitive psychometric tests of arithmetic ability or free hand drawing were impaired in different ways; indicating slowing performance against time and loss of finer judgement. Subjects studied by the N.I.M.H. groups showed that only accuracy of serial addition was impaired but that other simple cognitive measurements were not altered (ability to say the alphabet or count backward).
Studies were also performed on volunteers given placebos or oral doses of crude marihuana extracts, asayed for Δ-9-THC content which ranged from 5 to 60 mg. In general the effects produced by these extracts were comparable to those produced by similar amounts of synthetic Δ-9-THC. Ingestion of extracts containing the equivalent amounts of 20, 40, 60 mg Δ-9-THC produced impairment of short-term memory; these impairments did not follow a smooth time function but were episodic, brief in duration and not always under volitional control; they were accompanied by intermittent lapses of memory. Furthermore, oral doses of marihuana extract containing 40 to 60 mg of Δ-9-THC, significantly impaired the social co-ordination of cognitive operations during a task that required sequential adjustments in reaching a goal. This disintegration of sequential thought is related to impaired immediate memory, and is associated with disorganized speech and thinking. Disturbance of this type has been called " temporal disintegration ". Cannabis does, therefore, interfere with the cerebral mechanism which controls the selection of information deriving from immediate memory storage. The influence of chronic cannabis intoxication on that mechanism, which is so important in the learning process during the formative school years, is not known, but one cannot exclude the possibility that it could be significantly impaired. The temporal in co-ordination of recent memory with a task to achieve may account in part for the speech pattern of the marihuana user who is not able to co-ordinate recent memory with temporal immediate goals. The intoxicated subject forgets what he is about to say the moment that immediately follows the end of a sentence, and he has a strong tendency to discuss matters that have nothing to do with the preceding sentence because the logical sequence of his thought escapes him. He acknowledges that he needs to exert a considerable effort in order to recall from one moment to the other the logical thread of what he is in the process of expressing.
Clark and Nakashima (1968), also used oral marihuana extracts on volunteer subjects never exposed before to cannabis and studied their discriminatory and retentive faculties. They observed the disruptive effects of cannabis in sequential thought which suggest impairment of rapid decision-making and of short-term memory. They noted important variations in the same subject and from one subject to another, as far as the dose required to impair individual performance. These authors conclude: "These results are consistent with those reached when studying the influence of drugs such as LSD on such a complex state as behaviour which is influenced by a multitude of non-pharmacological factors. It is impossible to predict the effect of marihuana on different individuals or on the same individual at different times and in different circumstances. This impossibility to predict the effects of cannabis still increases the dangers of using that drug."
In a subsequent experiment with a dose of cannabis equivalent to 66 mg/kg of body weight Δ-9-THC they observed significant impairment in complex reaction time, digit code memory, time estimation, hand steadiness and reading comprehension. The sporadic nature of the effect was noted with lapses in psychomotor response as attention waned.
These studies with ingested cannabis extract confirm the psychotomimetic properties of cannabis first described by Moreau. They indicate the greater danger of using this route of administration which requires larger dosage, is accompanied by much less psychological conditioning, and, therefore, a more rapid development of tolerance.
Some investigators question the relevance of experiments performed on volunteers in a laboratory setting with "large doses of strong, synthetic material" to the social use of marihuana smoked in an amount sufficient to produce a "normal, socially acceptable high."
Jones (1971) claims for instance: "To do socially relevant experiments with marihuana in the laboratory one must have some idea of what dose people are smoking in a ' typical, social situation'." His study contains exceedingly relevant social data which should outlast the "typical social situation" of the California student milieu of 1970, and endure in any social situation throughout the world: (1) That tolerance develops in daily marihuana users in spite of self denial; (2) That daily users "tend to have poorer work histories, school performance and social adjustments".
As shown by Hollister (1968) and Tart (1970), it is possible through the use of questionnaires to gather a basic group of symptoms which can be specifically tabulated for the different psychotropic drugs. It can now be reported that the clinical symptoms observed and described in laboratory studies of cannabis intoxication are quite similar to those described by those who use the drug socially. One such questionnaire shows that the most common symptoms reported are floating sensations, depersonalization, weakness, relaxation, perceptual changes in vision, hearing and touch, subjective slowing of time, loss of attention and immediate memory, difficulty of concentration, euphoria, sleepiness. Other answers indicate increased insight and enhanced sexual performance and enjoyment, claims which cannot be verified in the ordinary laboratory. Another questionnaire study of 42 randomly selected students who had used marihuana indicate the following results: 90% had experienced minor changes in perception (seeing colours or objects as more intense); about half had experienced major perceptual changes (hallucinating colours or designs); and 40% had experienced hallucinogenic reactions. These were as frequent among those who had not used mescaline or LSD, and it was not necessary to have used marihuana a great many times to present these reactions. It was concluded from these questionnaires that cannabis is an hallucinogen, a statement which concurs with that made by Moreau after he experienced the drug himself in 1840. All of these inquiries indicate that the respondents to the questionnaire corroborated experimental observations and had experienced with the same range of dosage used in the laboratory. Some of the dosages used were larger than required to produce a " normal, socially acceptable high" since hallucinogenic responses were frequently reported (Hollister 1971).
It has been demonstrated in the experimental animal that Δ-9-THC interacts with many commonly used psychotropic drugs: It acts synergistically with amphetamines and caffeine. It potentiates the depressant action of barbiturates and ethanol. In man, a similar potentiating effect between cannabis preparations and ethanol has been observed (Manno et al. 1971) and there is good reason to believe that interactions between cannabis preparations and barbiturates, caffeine and amphetamine are also present. This last interaction was already mentioned in the La Guardia Report. In addition to all of these drugs so commonly used in our society, one should mention the antihistamines, tranquillizers, phenothiazines, benzodiazepines, imipramines, butyrophenones. The resulting interactions of Δ-9-THC and other cannabinoids with these drugs which may stimulate or inhibit the same enzyme systems, must be carefully appraised. Indeed it has already been established that regular consumers of cannabis do not abstain from smoking cigarettes or drinking alcohol beverages. They are also prone to experiment and use other psychotropic drugs. The frequent daily smokers studied by Jones used more hallucinogen, alcohol and tobacco than occasional smokers. The marihuana smoker will also tend to be a heavy smoker of tobacco cigarettes. The mere act of smoking may enable him to recapture, through psychological conditioning associated with similar sensory perceptions, some of the pleasant effects of cannabis intoxication. It will be of interest to study the interaction of tobacco and cannabis on lung and heart function.
The usage of multiple drugs by daily chronic smokers of marihuana in the United States is so prevalent that it has led Mirin et al. (1971) to the following conclusions: "It is difficult to assess the effects of marihuana per se in many heavy users, since what is observed is a multiple drug abuse syndrome."
That tolerance to cannabis may develop in man, as it does in animals was first indicated by the following exerpt from the 1894 report of the Indian Hemp Drugs Commission: "Powerful and noxious drugs are occasionally introduced into the pipe; but this practice is confined to those excessive consumers on whom hemp alone has ceased to produce the desired effect of exhilaration or stupefaction." Evidence derived from observations made in India suggest that tolerance develops with chronic use of potent preparations. It would indeed appear doubtful that Indian smokers of ganja or charas could consume daily an estimated average of 70 mg of Δ-9-THC, as reported by Chopra and Chopra (1939) without having developed some tolerance. A similar dose in a novice would produce acute psychotomimetic effects. This tolerance would explain why chronic users of cannabis may use large amounts of potent preparations without suffering any apparent severe somatic toxicity. These older reports have been corroborated by more recent ones. Morrow, who performed psychomotor tests on non-users and habitual users of cannabis in the La Guardia Report states "Non-users generally seem to be more affected by the drug when it is ingested than are users". Williams and co-workers (1946) reported in 1946 that the repeated administration to volunteers of synhexyl (a synthetic derivative of cannabis which could be given in known amounts or, ad libitum, smoking of marihuana cigarettes) resulted in decreasing effects within 4 to 6 days. The subjects requested an increase in dose. During the experimental period which lasted 39 days (table II), the number of cigarettes smoked daily by the subject increased and the users experienced "euphoria for several days ", then "general lassitude and indifference ". These observations are indicative that chronic marihuana smokers may well develop tolerance to the psychological as well as physiological effects of cannabis intoxication.
Wilson and Linken report from England in 1965 that a few adolescent cannabis users tend to use increasing doses. These chronic smokers, who find great difficulty in breaking the habit and do not particularly enjoy their dependent needs, appear to suffer from some degree of psychic dependence coupled with a requirement for fairly high doses of the drug.
Miras reports in 1969 that hashish smokers he has known in Greece for 20 years "are able to smoke at least 10 times as much as other people. If a beginner smoked the same quantity, he would collapse."
The rapid development of tolerance to cannabis preparations is noted in a report describing the respiratory complications of 31 young American soldiers stationed in Germany. They smoked monthly, for 6 to 15 months, 100 grams or more hashish, probably smuggled from North Africa or the Middle East. "Every patient described the development of this hashish tolerance as one which simply occurred by consuming increasing amounts over a few weeks period. This amount corresponds to three to four cigarettes a day containing one gram of hashish, which would represent 20 to 70 mg of Δ-9-THC. Such a figure corresponds to the one given by Chopra for the dosage used by chronic smokers of ganja in India.
A candid report from Israel by Freedman and Peer (1968) is indicative of the development of tolerance to cannabis. Seven out of twenty-one pimps or prostitutes with little or no high school education, between 18-42 years of age, median age 28, studied in Israel admitted to having to increase the dose of hashish they were taking. "When your body gets used to it you want it stronger, the body needs more and more." "You start off with a small cigarette, then a big one, then a narguila" (water pipe). The remainder in this group (fourteen) claimed that they used the same amount of hashish. However, if one takes into account the age of the hashish user or the length of his habit, it can be observed that all of the members of the group (five) who had used hashish for more than 12 years also admitted using opium.
All of this data is in agreement with the marked development of tolerance to cannabis derivatives in animals. It does not support the concept of "reverse" tolerance suggested by Weil et al. (1968) and entertained uncritically by other authors and which has become part of the new folklore on cannabis. This condition has been described by habitual smokers of marihuana, who claim that following initial exposure to the drug, they will need a lesser amount to become intoxicated, and will not feel constrained to increase the dose in order to obtain the desired effect. This so-called reverse tolerance can be accounted for by the enzymatic induction which occurs after initial exposure to the drug, and also by the positive reinforcement which accompanies the smoking process. This phenomenon is short lived, as functional tolerance which rapidly develops to the active metabolites of cannabis predominates over psychological conditioning.
How long will a frequent marihuana smoker be able to develop physical tolerance to cannabis and still experience the same euphoric response to the same dosage of the drug? The scientific evidence presented by Jones answers this question, and confirms the older observations reported in the literature - "frequent users (daily) develop a tolerance to the physical as well as the psychologic effects of cannabis. The frequent users used the drug longer (5.2 years), had greater experience with alcohol and hallucinogens, and smoked more tobacco. They tended to have poorer work histories, school performance and social adjustment, to miss scheduled test sessions and to miss appointments."
Jones adds the following comment: "If tolerance to behavioural and physiologic effects developed without tolerance to the desirable subjective effects, cannabis would perhaps be a useful drug. This would be a situation similar to that seen in tobacco smoking. Unfortunately, it appears that tolerance to the sought-after subjective effects also occurs with marihuana."
It is well known that a few strong-willed, motivated persons, endowed with powerful detoxifying enzyme systems, can control for many years the amount of drugs (including opiates and cocaine) they can take in order to obtain the desired effect. Such persons like the French artist and author Jean Cocteau, or the mysterious M. X, interviewed by Grinspoon, fall out of the normal range of any random population sample; rapid tolerance to psychoactive drugs is most likely to develop among the average human being especially if he is an adolescent with a labile personality and an uncertain future, or if he belongs to the underprivileged groups which today in our society are striving to achieve a better life. It is very doubtful that the use of cannabis would help them achieve this goal. It is now well established that pharmacological tolerance develops to Δ-9-THC and cannabis. The tolerance to smoked cannabis can be controlled by casual users for some time, through psychological conditioning. However, all pharmacological and clinical evidence presently available indicates that frequent (daily) users of cannabis will develop tolerance to the physiologic as well as the psychologic effects of the drug. This tolerance to cannabis gives a physiological basis to the necessity for the frequent smoker to increase dosage, or to use more potent psychotropic drugs such as other hallucinogens or the opiates.
It is time to revise the popular belief that "there is little or no tendency to increase dosage of cannabis since there is little or no development of tolerance" (WHO Expert Committee, 1964). Such a belief is currently reinforced by all of the statements contained in the books on cannabis available in 1971 to the English reader (Kaplan 1971; Snyder 1971; Grinspoon 1971; Schofield 1971).
A misconception seems to have penetrated into the mind of many psychologists and physicians who believe that " addiction ", meaning physical dependence accompanied by withdrawal symptoms, is the main criterion by which the potential harm of a drug to the individual or to society should be gauged. One must be very careful about drawing too sharp a line between physical and psychological dependence. There is no complete dichotomy between mind and body. Psychological function does also have physiological and biochemical bases. The desire for instant gratification is a profound psychological reinforcer. Physical dependence does not develop with central nervous system stimulants such as cocaine which is known to create in an individual one of the most enslaving types of drug dependence.
Addiction to a drug is not a function of the ability of the drug to produce withdrawal symptoms. Drug dependence results basically from the reproducible interaction between an individual and a pleasure-inducing biologically active molecule. The common denominator of all drug dependence is the psychological reinforcement resulting from reward associated with the past individual drug interaction and the subsequent increasing desire for repeated reinforcement (Seevers, 1970). On this basis it is deceptive to categorize marihuana as a "soft" acceptable drug.
The example of Freud, who recommended cocaine to his friend, von Fleishl, to help him withdraw from morphine addiction, should be kept in mind. With this new drug, von Fleishl was able to withdraw from morphine; but he used cocaine instead, and ever increasing doses until he was in a state of constant intoxication. As a result of this sad experience, Freud, even when he suffered from cancer, did not take pain killing medications, except aspirin (Lennard, 1971). As Jones (1953), his biographer, says, "Freud, like all good doctors, was averse to taking drugs." The example of cocaine clearly demonstrates that a strong psychological reinforcement is the only necessary requirement to perpetuate a most compelling form of drug addiction.
While cannabis users will develop tolerance to the drug, they do not present any significant physical dependence identifiable by specific withdrawal symptoms, similar to those occurring with heroin or ethanol. The symptoms observed following discontinuation of heavy use are relatively mild. Loss of appetite, insomnia, irritability are well-tolerated, but it is well documented that cannabis may create a state of psychological dependence which is an important obstacle to discontinue usage.
In India, "chronic hemp habituation" says Chopra (1969), "is a self-inflicted disease. It is progressive and seldom abates by itself. The urge to smoke may become so great that the individual inhales until he loses consciousness. Repeated use of the drug leads to a craving and psychological dependence."
Soueif (1967) reports that in Egypt 65 % of the consumers of hashish declared they were unable to stop although they expressed a wish to discontinue their habituation. According to Soueif, "Among hashish users there is a definite pattern of oscillation of temperamental traits, swinging between two opposite poles, that of social ease, a desire to mix, acquiescence and elation (euphoria) when under immediate drug effects and that of ascendancy, seclusiveness, negativism, depression of the mood and pugnacity (which may be considered as main components of a psychic withdrawal syndrome) when the subjects are deprived of the drug. This pattern of oscillation of the subject's personality, between the drugged state and the state of deprivation from the drug, may be considered", according to Soueif, "as the behavioural core of a state of psychic dependence. One of the most salient characteristics of this state is a need to continue taking the drug not only to attain the feeling of well being but also to avoid feeling low."
Statements reported are: "It finishes you off and makes you lazy too. You have to either give up the drug or your future. It can affect your health sometimes, because it's a drug. A person is always like a drunk - high - doesn't know what is happening with him and it interferes with his future. It louses up your family life, kids, doesn't let you get ahead and make a living." Many of the young habitual cannabis smokers in the western culture strongly deny any ill effect on performance or interpersonal relationships. The observations of Yardley (1968) who was a proctor in 1965 at Oxford University are indicative of the self-deception experienced by students who fall into the habit of using cannabis regularly. "Every one of those who were regular takers seemed to be convinced that cannabis was not habit forming; that they had not developed any real habit of taking it; and that they would give up the drug at any time at will; that it was a cleaner practice than the taking of alcohol; and that it should be legalized. But most of those who had become accustomed to taking this drug regularly had to call on professional help to give it up. Furthermore, it was plain that those who did take it regularly tended gradually to increase their consumption and a certain number of them, small but perhaps significant, graduated to hard drugs."
Man is a creature of habit, and Americans are not immune from the marihuana habit. This habit has been reported among American smokers between 20 and 30 years of age who have usually a history of four or five years of marihuana use (Scher, 1970). They describe a decreasing interest in the use of marihuana, but seem completely unwilling or unable to discontinue usage of the drug. They develop diminished pleasure from the drug, requiring more (a stronger amount or a different variety) to produce the "high" - all signs of the same increased tolerance which now becomes so apparent in frequent users.
The Israeli users of hashish studied by Freedman and Peer resemble closely, in this respect, their Egyptian neighbours. Half of those questioned, acknowledged candidly that they could not give up hashish. Some of the answers of these Israeli cannabis smokers are revealing: "Maybe somebody with a strong character can quit. Because all you do is to smell it and you're back smoking." Eleven out of 21 said: "You can kick the habit, and you don't need the hospital - it's only hard the first month." However, when the members of the group were asked, "Do you know of cases where people who quit went back to smoking again?" Twenty out of 21 answered they knew such people, because "you always go back to it - you can't stop." This statement contrasts with the preceding one because, "It is easier to be frank when talking about others than when talking about one's self".
Clinical studies with measured amounts of Δ-9-THC, performed mostly on student volunteers in the United States, indicate the following:
The first widely publicized studies claimed that smoked marihuana containing 5 to 66 mg Δ-9-THC was a "mild intoxicant". It appears that these studies were performed with unextracted material containing, in reality, subthreshold amounts of Δ-9-THC.
The half life of Δ-9-THC in blood plasma is 28 hours for chronic marihuana smokers, as compared to 57 hours for non-users. Apparent volume of distribution is similar to both groups. Chronic users eliminate significantly more polar metabolites in the urine, and less in the faeces than non-users. The total amount of metabolites eliminated in both groups is the same and requires more than one week. Repeated administration of cannabinoids at less than a week interval will results in accumulation of metabolites in tissue including brain.
Δ-9-THC is three to four times more active when smoked than when ingested.
Tachycardia which is dose related and lasts during the whole period of intoxication is the most significant clinical feature observed after administration of Δ-9-THC or cannabis extracts. A dose related conjunctival injection is also observed.
Δ-9-THC in doses of 15 mg smoked and 40 mg ingested is psychotomimetic and will produce hallucinations. Such a dose is within the range which may be consumed by habitual cannabis users in all countries including the United States.
Cannabis extracts containing 5-10 mg of THC smoked or 15-25 mg ingested impair significantly motor and mental performance.
Frequent (daily) users of cannabis will develop physiologic and psychologic tolerance to the drug which will require greater dosage, and may lead them to more potent psychotropic drugs such as LSD or opiate derivatives.
Although cannabis users do not develop any physical dependence identifiable by a specific withdrawal symptom, they present a psychological dependence to the drug.
These studies in clinical pharmacology underline the potential health and social liabilities associated with cannabis intoxication. All of the major effects of cannabis derivatives justify their classification in Delay's functional nomenclature of psychotropic drugs among the "psychodysleptic" compounds which "disintegrate mental function and produce a distortion of judgement and memory with impairment in evaluating the reality situation ".
_____, (1971), A Review of the Biomedical Effects of Marihuana on Man in the Military Environment, Federation of American Society for Experimental Biology, Bethesda, Md.
Abel, E. (1970), "Marijuana and memory ", Nature, 227:1151-1152.
Abramson, H. A., M. E. Jarvik, M. R. Kaufman, C. Kornetsky, A. Levine and M. Wagner (1955), "Lysergic acid diethylamide (LSD-25). Physiological and perceptual responses", J. Psychol. 39:3-60.
Allentuck, S. (1941), "Medical aspects of the marijuana problem in the City of New York", in: The Marihuana Papers, edited by D. A. Soloman, pp. 269-284. Bobbs-Merrill Co., Inc., New York.
Allentuck, S. and K. M. Bowman (1942), "The psychiatric aspects of marihuana intoxication ", Amer. J. Psychiat., 99:248-251.
Baker, A. A. and E. G. Lucas (1969), "Some hospital admissions associated with cannabis ", Lancet 1:148.
Baker-Bates, E. T. (1935), "A case of Cannabis indica intoxication ", Lancet, 1:811.
Beringer, K., et al. (1932), "Zur Klinik des Hashischrausches ", Der Nervenarzt, 5:337-350.
Brill, N. O., E. Crumpton, I. M. Frank, J. S. Hochman, P. Lomas, W. H. McGlothlin and L. J. West (1970), "The marijuana problem ", Ann. Intern. Med., 73:449-465.
Bromberg, W. (1934), Marihuana intoxication," A clinical study of Cannabis sativa intoxication ", Amer. J. Psychiat., 91:303-330.
Burns, J. J. (1967), "Symposium on Drug interactions ", The Pharmacologist, 9-77:81.
Caldwell, D. V., S. A. Myers and E. F. Domino (1970), Effect of marihuana smoking on sensory thresholds in man ", In: Psychotomimetic Drugs, edited by E. H. Efron, pp. 299-321. Raven Press, New York.
Caldwell, D. F., S. A. Myers, E. F. Domino and D. E. Merriam (1969), "Auditory and visual threshold effects of marihuana on man ", Percept. Motor Skills, 29:755-759.
Chopra, G. S. (1969), "Man and marijuana ", Intern. J. Addict., 4:215-247.
Chopra, R. N. and G. S. Chopra (1939), "The present positions of hemp drug addiction in India", Indian J. Med. Res. Mem.. 31:1-119.
Christiansen, J. and O. J. Rafaelsen (1969), "Cannabis metabolites in urine after oral administration ", Psychopharmacologia, 15:60-63.
Clark, L. D., R. Hughes and E. N. Nakashima (1970), "Behavioral effects of marihuana: Experimental studies ", Arch. Gen. Psychiat. (Chicago), 23:193-198.
Clark, L. D. and E. N. Nakashima (1968), "Experimental studies of marihuana ", Amer. J. Psychiat. 125:135-140.
Colbach, E. (1971), "Marijuana use by GIs in Viet Nam ", Amer. J. Psychiat., 128:96-99.
Council on Mental Health and Committee on Alcoholism and Drug Dependence (1967)," Dependence on Cannabis (marihuana)", J. Amer. Med. Ass., 201:368-371.
Crancer, A., Jr., J. M. Dille, J. C. Delay, J. E. Wallace and M. D. Haykin (1969)," Comparison of the effects of marihuana and alcohol on simulated driving performance ", Science, 164:851-854.
Delay, J. (1965), "Psychotropic drugs and experimental psychiatry ", Int. J. Neuropsychiat., 1:104-117.
Dinnerstein, A. J. (1968), "Marihuana and perceptual style. A theoretical note", Percept. Motor Skills, 26:1016-1018.
Dornbush, R. L., M. Fink and A. M. Freedman (1971)," Marijuana, memory and perception ", Amer. J. Psychiat., 128:2.
Eddy, N. B., H. Halbach, H. Isbell and M. Seevers (1965), "Drug dependence: Its significance and characteristics ", Bull. W. H. O., 32:721-733.
Edwards, G. (1968), "The problem of Cannabis dependence ", Practitioner, 200:226-233.
FDA Announcement (1971), "Clinical studies with marihuana and tetrahydrocannabinol currently being allowed by the Food and Drug Administration ", Clin. Pharmacol.
Fisher, S. (1970), "Nonspecific factors as determinants of behavior response to drugs ", in: Clinical Handbook of Psychopharmacology, edited by A. DiMascio and R. I. Shader, pp. 17-39. Science House, New York.
Foltz, R. L., G. W. Kinzer, R. I. Mitchell and E. B. Truitt, Jr. (1971), "The fate of cannabinoid components of marihuana during smoking" (in press).
Forney, R. B. (1971), "Toxicology of marihuana ", Pharmacol. Rev., 23:279-284.
Frank, I. M., R. S. Hepler, S. Stier and W. Rickles (1971), "Marijuana tobacco and functions affecting driving ", Paper presented at the American Psychiatric Association Annual Meeting, Washington, D. C., May (in press).
Fraser, J. D. (1949), "Withdrawal symptoms in Cannabis indica addicts ", Lancet, 2:747.
Freedman, H. and N. Rockmore (1946), "Marihuana: A factor in personality evaluation and Army maladjustment", J. Clin. Psychopathol., 7:765-782.
Freedman, I. and I. Peer (1968), "Drug addiction among pimps and prostitutes in Israel ", Int. J. Addictions, 3:271-300.
Gaskill, H. S. (1945), "Marihuana, an intoxicant ", Amer. J. Psychiat., 102:202-204.
Gill, E. W., W. D. M. Paton and R. G. Pertwee (1970)," Preliminary experiments on the chemistry and pharmacology of cannabis" Nature 228:134-136.
Grinspoon, L. (1969), "Marihuana ", Sci. Amer., 221:17-25.
Grinspoon, L. (1971), "Marihuana Reconsidered", Harvard University Press, Cambridge, Mass.
Haertzen, C. A. (1966), "Development of scales based on patterns of drug effect, using the Addiction Research Center Inventory (ARCI)", Psychol. Rep., 18:163-194.
HEW Report to Congress (1971), Marihuana and Health, U.S. Government Printing Office, Washington, D. C.Hepler, R. S. and I. Frank (1971), "Marihuana smoking and intraocular pressure ", J. Amer, Med. Ass., 217:1392.
Hepler, R. S. and I. Frank and J. T. Ungerleider (1971), "The effects of marihuana smoking on pupillary size ", Amer. J. Ophthal.
Hockman, C. H., R. G. Perrin and H. Kalant (1971), "Electroencephalographic and behavioral alterations produced by Δ-l-tetrahydrocannabinol ", Science, 172:968-970.
Hollister, L. E. (1968), Chemical Psychoses: L. S. D. and Related Drugs. Thomas, Springfield, Illinois.
Hollister, L. E. (1969), "Steroids and moods: Correlations in schizophrenics and subjects treated with lysergic acid diethylamide (LSD), mescaline, tetrahydrocannabinol, and synhexyl ", J. Clin. Pharmacol., 9:24-29.
Hollister, L. E. (1969), "Criminal laws and the control of drugs of abuse: An historical view of the law (or, it's the lawyer's fault)", J. Clin. Pharmacol., 9:345-348.
Hollister, L. E. (1970), "Tetrahydrocannabinol isomers and homologues: Contrasted effects of smoking", Nature, 227:968-969.
Hollister, L. E. (1971), "Marihuana in man: Three years later", Science, 172:21-24.
Hollister, L. E. (1971), "Hunger and appetite after single doses of marihuana, alcohol and detroamphetamine", Clin. Pharmacol. Ther., 12:44-49.
Hollister, L. E. (1971)," Actions of various marihuana derivatives ", Pharmacol. Rev., 23:349-358.
Hollister, L. E. and H. K. Gillespie (1969), "Similarities and differences between the effects of lysergic acid diethylamide and tetrahydrocannabinol in man ", in: Drugs and Youth, edited by J. R. Wittenborn, H. Brill, J.-P. Smith and S, A. Wittenborn. C. C. Thomas, Springfield, Illinois, pp. 208-211.
Hollister, L. E. and H. K. Gillespie (1970), "Marihuana, ethanol and dextroamphetamine: mood and mental function alterations", Arch. Gen. Psychiat. (Chicago), 23:199-203.
Hollister, L. E. and F. Moore (1967), "Urinary catecholamine excretion following lysergic acid diethylamide in man ", Psychopharmacologia (Berlin), 11:270-275.
Hollister, L. E. and F. Moore (1968), "Urinary catecholamine excretion after mescaline in man", Biochem. Pharmacol., 17:2105.
Hollister, L. E., F. Moore, S. L. Kanter and E. Noble (1970), "Δ-l-tetrahydrocannabinol, synhexyl and marihuana extract administered orally in man: Catecholamine excretion, plasma cortisol leveland platelet serotonin content", Psychopharmacologia, 17:354-360.
Hollister, L. E., R. K. Richards and H. K. Gillespie (1969), " Comparison of tetrahydrocannabinol and synhexyl in man", Clin. Pharmacol. Ther., 9:783-791.
Hollister, L. E., S. L. Sherwood and A. Cavasino (1970)," Marihuana and the human electroencephalogram ", Pharmacol. Res. Commun., 2:305-308.
Hollister, L. E. and B. J. Sjoberg (1964), "Clinical syndromes and biochemical alterations following mescaline, LSD, psilocybin and a combination of the 3 psychotomimetic drugs ", Compr., Psychiat., 5:170-178.
Hughes, J. E., L. P. Steahly and M. M. Bier (1970), "Marihuana and the diabetic coma ", J. Amer. Med. Ass., 214:1113-1114.
Indian Hemp Drug Commission (1969)," Report on Marihuana of the Indian Hemp Commission, 1893-1894", Thomas Jefferson Publishing Company, Silver Springs, Maryland
Isbell, H. (1971), "Clinical pharmacology ", Pharmacol. Rev., 23:337-338.
Isbell, H., G. W. Gorodetsky, D. Jasinski, U. Claussen, F, Spulak and F. Korte (1967)," Effects of ( - ) Δ 9 transtetrahydrocannabinol in man ", Psychopharmacologia, 11:184-188.
Isbell, H. and D. Jasinski (1969), "A comparison of LSD-25 with ( - ) Δ-9-transtetrahydrocannabinol (THC) and attempted cross tolerance between LSD and THC ", Psychopharmacologia, 14:115-123.
Johnson, S. and E. F. Domino (1971), "Some cardiovascular effects of marihuana smoking in normal volunteers ", Clin. Pharmacol. Ther. 12:762-786.
Jones, Ernest (1953), The Life and Work of Sigmund Freud. Basic Books, New York.
Jones, R. T. (1971)," Marihuana-induced ' high ': Influence of expectation, setting and previous drug experience ", Pharmacol. Rev., 23:359-370.
Jones, R. T. (1971), "Tetrahydrocannabinol and the marihuana-induced social ' high ' or the effects of the mind on marihuana ", Ann. N. Y. Acad. Sci., 191:155-165.
Jones, R. T. and G. C. Stone (1970), "Psychological studies of marihuana and alcohol in man ", Psychopharmacologia, 18:108-117.
Joyce, C. R. B. and S. H. Curry, eds. (1970), The Botany and Chemistry of Cannabis, J. & A. Churchill, London.
Kalant, H. (1969), "Marihuana and simulated driving", Science, 166:640.
Kaplan, H. S. (1971), "Psychosis associated with marijuana ", N. Y. St. J. Med. 71:433-435.
Kaplan, J. (1971)," Marihuana and drug abuse in Vietnam ", Ann. N. Y. Acad. Sci. 191:261-269.
Kiplinger, G. F. and J. E. Manno (1971), "Dose-response relationships to Cannabis in human subjects", Pharmacol. Rev., 23:339-348.
Korte, F. and H. Sieper (1965)," Recent results in hashish analysis ", In Hashish: Its Chemistry and Pharmacology, edited by G. E. W. Wolstenholme and J. Knight Little Brown and Co., Boston, Mass.
Lemberger, L., J. Axelrod and I. J. Kopin (1971), "Metabolism and disposition of Δ-9-tetrahydrocannabinol in man", Pharmacol. Rev., 23:371-380.
Lemberger, L., S. D. Silberstein, J. Axelrod, and I. J. Kopin (1970), "Marihuana: Studies on the disposition and metabolism of Δ-9-tetrahydrocannabinol in man ", Science, 170:1320-1322.
Lemberger, L., N. R. Tamarkin, J. Axelrod and I. J. Kopin (1971), "Δ-9-THC metabolism and disposition in long-term marihuana smokers ", Science, 173:72-74.
Lerner, M. and J. T. Zeffer, (1968), "Determination of tetrahydrocannabinol isomers in marihuana and hashish. Bulletin on Narcotics, XX, 2, pp. 53-54.
Lennard, H. L., L. Z. Epstein, A. Bernstein and D. C. Ransom (1971), "Mystification and Drug Misuse", Jossey-Boss. San Francisco.
Mann, P. E. G., A. B. Cohen, T. N. Finley and A. J. Ladman (1971)," Alveolar macrophages. Structural and functional differences between nonsmokers and smokers of marijuana and tobacco", Lab. Invest., 25:111-120.
Manno, J. E., G. F. Kiplinger, S. E. Haine, I. F. Bennett, and R. B. Forney, (1970), "Comparative effects of smoking marihuana or placebo on human motor and mental performance ", Clin. Pharmacol. Ther., 11:808-815.
Manno, J. E., G. F. Kiplinger, N. Scholtz and R. B. Forney (1971), "The influence of alcohol and marihuana on motor and mental performance", Clin. Pharmacol. Ther., 12:202-211.
Mayor's Committee on Marihuana (1944), The Marihuana Problem in the City of New York - Sociological, Medical, Psychological and Pharmacological Studies. Cattell, Lancaster, Pennsylvania.
McGlothin, W. H., D. O. Arnold and P. K. Rowan (1970), "Marihuana use among adults ", Psychiatry, 4:433.
Melges, F. T., J. R. Tinkelberg, L. E. Hollister and H. K. Gillespie (1970), "Marihuana and temporal disintegration ", Science, 168:1118-1120.
Melges, F. T., J. R. Tinkelberg, L. E. Hollister and H. K. Gillespie (1970), "Temporal disintegration and depersonalization during marihuana intoxication ", Arch. Gen. Psychiat., (Chicago), 23:204-210.
Meyer, R. E., R. C. Pillard, L. S. Shapiro and S. M. Mirin, (1971), "Administration of marihuana to heavy and casual marijuana users ", Amer. J. Psychiat., 128:2
Miras, C. J. (1969)," Experience with chronic hashish smokers ", In: Drugs and Youth, edited by J. R. Wittenborn, H. Brill, J. P. Smith and S. A. Wittenborn. Charles C. Thomas, Springfield, pp. 191-198.
Miras, C., S. Simos and J. Kiburis (1964), "Comparative assay of the constituents from the sublimate of smoked cannabis with that from ordinary cannabis. Bulletin on Narcotics XVI, 1, pp. 13-15.
Mirin, S. M., L. M. Shapiro, R. E. Meyer, R. C. Pillard, and S. Fisher (1971)," Casual vs heavy use of marihuana, a redefinition of the marihuana problem ", Amer. J. Psychiat., 127:1134-1140.
Mohan, H. and G. C. Sood (1964), "Conjugate deviation of the eyes after Cannabis indica intoxication", Brit. J. Ophthal., 48:160-161.
Moreau, J. J. (1845), Du hachish et de l'aliénation mentale: études psychologiques 34, Libraire de Fortin, Masson, Paris.
Myers, S. A. and D. F. Caldwell (1969), "The effects of marihuana on auditory and visual sensation: a preliminary report ", The New Physician, 18:212-215.
Newman, R. H., W. L. Jones and R. W. Jenkins (1969), "Automatic device for the evaluation of total mainstream cigarette smoke ", Anal. Chem., 41:543-545.
Palmer, E. L. (1949), Fieldbook of Natural History, McGraw-Hill Book Company, Inc., New York.
Parker, C. S. and F. J. Wrigley (1950), "Synthetic Cannabis preparations in psychiatry: Syn-hexyl ", J. Ment. Sci., 96:276-279.
Pillsbury, H. C., C. C. Bright, K. J. O'Connor and F. W. Irish (1969), "Tar and nicotine in cigarette smoke", J. Assoc. Offic. Anal. Chem., 52:458-462.
Pivik, T., V., Zarcone, L. E. Hollister and W. Dement (1969): "The effects of hallucinogenic agents on sleep ", Psychophysiology, 6:261.
Podolsky, S., C. G. Pattavina and M. A. Amaral (1971), "Effect of marihuana on glucose tolerance test ", Ann. N. Y. Acad. Sci., 191:54-60.
President's Commission on Law Enforcement and Administration of Justice, N. de B. Katzenbach, Chairman (1967), Task Force Report: Narcotics and Drug Abuse. U.S. Government Printing Office, Washington, D.C.
Renault, P. F., C. R. Schuster, R. Heinrich and D.X. Freeman (1971)," Marihuana: Standardized smoke administration and dose effect curves on heart rate in humans ", Science, 174:589-591.
Rodin, E. and E. F. Domino (1970), "Effects of acute marihuana smoking on the electroencephalograms ", Electroenceph. Clin. Neurophysiol., 29:321.
Rodin, E. A., E. F. Domino and J. P. Porzak (1970), "The marihuana-induced social high: Neurological and electroencephalograph concomitants", J. Amer. Med. Ass., 213:1300-1302.
Roland, J. L. and M. Teste (1958), "Le cannabisme au Maroc", Maroc- Med., 387:694-703. This same article appears under the name of Benabud, A. (1957), "Psycho-pathological aspects of the cannabis situation in Morocco: Statistical data for 1956", Bulletin on Narcotics, IX, 4, pp. 1-16.
Scher, J. M. (1970), "The marihuana habit ", J. Amer. Med. Assoc., 214:1120.
Schick, J. F., D. E. Smith and F. H. Meyers (1968), "The use of marijuana in the Haight-Ashbury subculture ", J. Psychedelic Drugs, 2:49-66.
Schofield, M. (1971), The Strange Case of Pot, Penguin Books, Middlesex, England.
Seevers, M. H. (1970), "Drug dependence and drug abuse, a world problem ", Pharmacologist, 12:172-181.
Smith, D. E. (1968), "Acute and chronic toxicity of marijuana ", J. Psychedelic Drugs, 2:37-41.
Snyder, S. H. (1970), "What have we forgotten about pot?" New York Times Magazine, December 13, p. 26.
Snyder, S. H. (1971), Uses of Marijuana, Oxford University Press, New York.
Soueif M. I. (1967), "Hashish consumption in Egypt with special reference to psychosocial aspects. Bulletin on Narcotics XIX, 2, pp. 1-12.
Talbott, J. A. and J. W. Teague, (1969)," Marihuana psychosis: Acute toxic psychosis associated with the use of cannabis derivatives ", J. Amer. Med. Ass., 210:299-302.
Tart, C. T. (1969), Altered States of Consciousness, J. Wiley & Sons, New York.
Tart, C. T. (1970), "Marihuana intoxication: Common experiences", Nature, 226:701-704.
Tart, C. T. and H. J. Crawford (1970), "Marijuana intoxication: Reported effects on sleep, Psychophysiol. 7:348.
Tennant, F. S., Jr., M. Preble, T. J. Prendergast and P. Ventry (1971)," Medical manifestations associated with hashish", J. Amer. Med. Ass., 216:1965-1969.
Tinklenberg, J. R., F. T. Melges, L. E. Hollister and H. K. Gillespie (1970), "Marihuana and immediate memory ", Nature, 226:1171-1172.
Volavka, J., R. Dornbush, S. Feldstein, G. Clare, A. Zaks, M. Fink and A. M. Freedman (1971), "Marijuana, EEG and Behavior ", Ann., N.Y. Acad. Sci., 191:206-215.
Waller, C. W. (1970), Supplies for the Marihuana Program - Report to the Committee on Problems of Drud Dependence. Natl. Acad. Sci., NRC, 6394-6396.
Waskow, I. E., J. E. Olsson, C. Salzman, and M. M. Katz (1970), "Psychological effects of Δ-9-THC", Arch. Gen. Psychiat. 22:97-107.
Weil, A. T. (1969), "Cannabis", Science J. (London), 5A:36-42.
Weil, A. T. (1970), "Adverse reactions to marihuana: Classification and suggested treatment ", New Eng. J. Med., 282:997-1000.
Weil, A. T. and N. E. Zinberg (1969), "Acute effects of marihuana on speech ", Nature, 222: 434-437.
Weil, A. T., N. E. Zinberg and J. M. Nelson (1968), "Clinical and psychological effects of marihuana in man ", Science, 162:1234-1242.
White, R. B., H. Goolishian and E. S. Barratt (1970), "Dilemmas encountered by the marihuana researcher", Paper presented at the 46th Annual Conference of the Central Neuropsychiatric Association, Galveston, Texas, October 16.
Wikler, A. (1970)," Clinical and social aspects of marihuana intoxication ", Arch. Gen. Psychiat., (Chicago), 22:320-325.
Williams, E. G., C. K. Himmelsbach, A. Wikler, D. C. Ruble and B. J. Lloyd, Jr. (1946)," Studies on marihuana and pyrahexl compound ", Public Health Rep., 61:1059.
Wilson, C. W. M. (1968), Adolescent drug dependence, Pergamin Press, London.
Wilson, C. W. M. and A. Linken (1968), "The use of Cannabis in relation to the adolescent in Adolescent drug dependence ", PP. 93-138, Pergamin Press, London, England.
WHO Expert Committee on Addiction - Producing Drugs (1964), Thirteenth Report, World Health Organization Technical Report Series No. 273, Geneva.
Yardley, D. C. M. (1968), "Legal aspects of drug dependence in relation to student drug abuse ", In: The Pharmacological and Epidemiological Aspects of Adolescent Drug Dependents, edited by C. W. M. Wilson. Pergamon Press, Oxford.