Background
Etomidate is a short‑acting intravenous general anaesthetic used clinically for the induction of anaesthesia because of its relative cardiovascular stability [1,2]. However, etomidate is well‑established to inhibit adrenal steroid synthesis through suppression of 11β‑hydroxylase, leading to adrenocortical suppression [3]. This adverse effect limits its medical use and raises concerns in non‑medical contexts, especially with repeated exposure or routes of administration not intended for therapeutic dosing [3].
Etomidate’s pharmacological profile makes it attractive for misuse, particularly as a sedative‑hypnotic substance [4]. In recent years evidence of significant levels of misuse of etomidate and related compounds has emerged internationally, with detections increasingly reported across East and South‑East Asia and, more recently, North America, Europe and Oceania, particularly by administration from vaping devices [1,4].
Current reports indicate that etomidate has moved rapidly beyond isolated incidents, with repeated detections in drug samples, toxicology cases, and drug checking services, indicating sustained availability [1,4]. Following national controls on etomidate (e.g., in China), several structurally related analogues including metomidate, propoxate, and isopropoxate have been detected, suggesting market substitution in response to regulation [1].
National controls on etomidate have been announced or implemented in countries or territories such as China, Hong Kong, Taiwan, South Korea, Indonesia, Singapore, Japan, United Kingdom, Australia and New Zealand. Etomidate may be regulated under and/or misuse of drugs legislation [4].
Definition and description
tomidate analogues are structurally related compounds developed originally for medical research or optimisation of anaesthetic properties [5,6]. In illicit contexts, these compounds appear primarily as substitutes, including substances such as metomidate, propoxate, isopropoxate, sec‑butomidate and butomidate, as well as halogenated analogues (e.g. fluorinated derivatives). They are expected to share pharmacological features with etomidate, including activity at the GABAA receptor and the potential to affect adrenal function, although the magnitude and duration of adrenocortical suppression may vary between compounds.
![]()
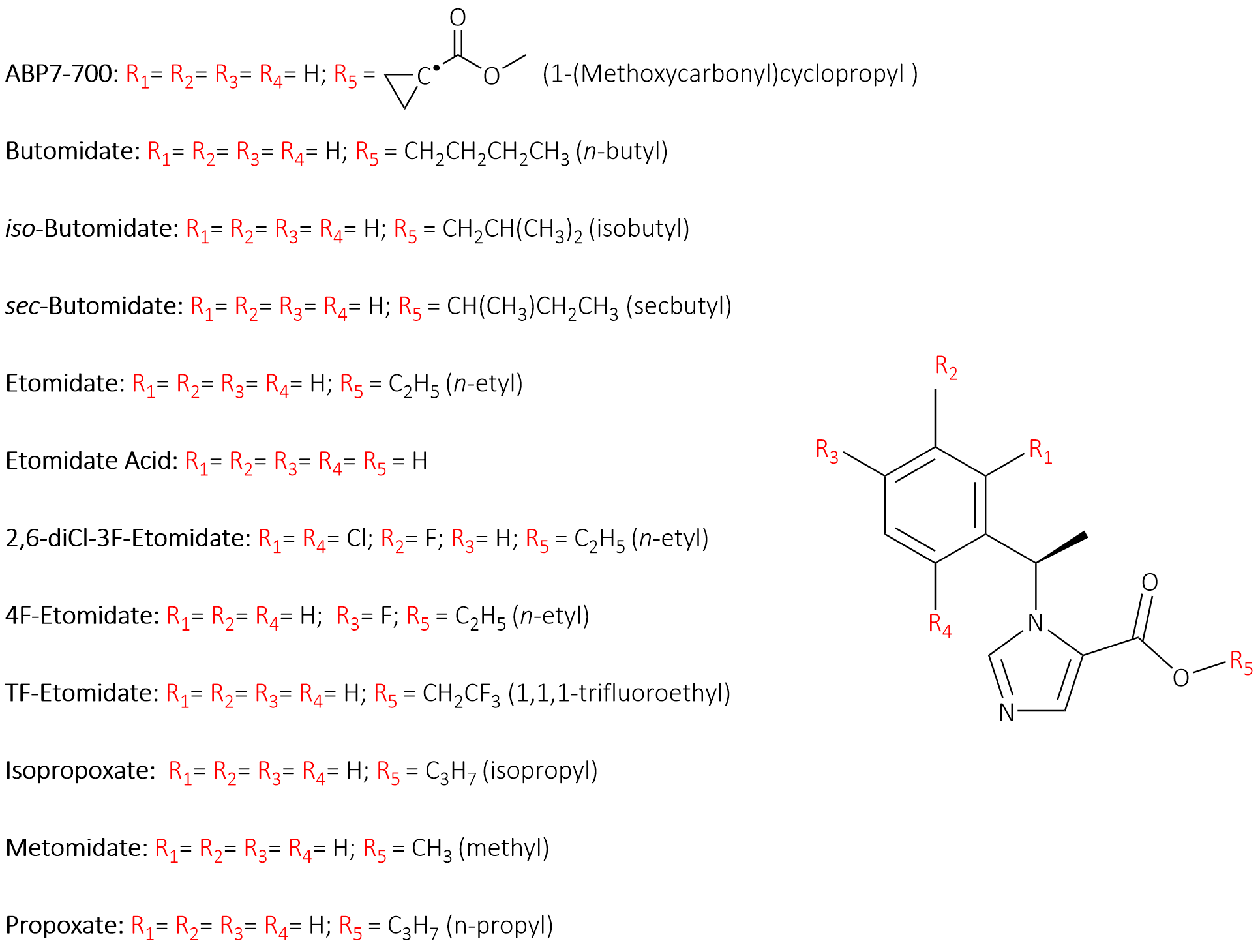
Figure 1: Core structure of etomidate and representative analogues.
The conserved etomidate scaffold shown in Figure 1 with variable substitution sites (R₁–R₄ on the aromatic ring and R₅ on the ester moiety), highlighting how systematic modifications, particularly at R₅ and on the phenyl analogues.
Commonly reported forms
Illicit etomidate use is most associated with vaping products, particularly e‑liquids and cartridges intended for inhalation via electronic vapourisers [1,4,5]. These products are often visually indistinguishable from other illicit vape liquids and may be marketed under informal street names, complicating identification and risk communication [1]. Etomidate has also been detected less frequently in tablets, powders and crystalline materials, including samples mis‑sold as pharmaceutical medicines or other controlled drugs [1,4].
Vaping products (e-liquids and cartridges):
Solid forms (tablets, powders, crystals):
tomidate is the most commonly detected compound in the group found in illicit products. Where analogues are detected, they are often present at much lower concentrations, consistent with trace‑level impurities [4], an important consideration for forensic interpretation. Forensic professionals should ensure they have the ability and capacity to analyse e-liquids (and other products designed for vaping) and detect etomidate and its related compounds.
Reported effects and toxicity
Etomidate and its analogues act primarily as central nervous system depressants, producing sedative and hypnotic effects that may be sought in illicit use, particularly through inhalation of vaping products [1,5]. Etomidate is also known to suppress adrenal steroid synthesis, a key toxicological concern, leading to reduced cortisol production [4,5,7]. Although some analogues may exhibit a reduced or shorter‑lasting effect compared with etomidate, residual adrenocortical suppression remains a clinically relevant risk, especially with repeated or frequent use [4]. Involuntary muscle movements, including myoclonus, are known reported adverse effects of etomidate and its analogue ABP-700 [2]. These movements may result from disinhibition of diencephalic pathways, but the exact mechanism remains unclear and requires further investigation.
Etomidate toxicity is of particular concern in individuals with renal impairment, as the drug is largely eliminated via the kidneys, and older adults may be at increased risk due to age‑related decline in renal function [4,5]. Overdose may occur with rapid or repeated dosing, leading to hypotension and cardiorespiratory depression, and there is no specific antidote. There are concerns about polysubstance exposure, noting that co‑use with opioids or other depressants or opioids may substantially increase the risk of adverse outcomes [4]. Uncertainties around dose, exposure patterns, and long‑term effects particularly via inhalation further complicate toxicological assessment and risk evaluation in illicit use contexts.
Analytical considerations
Analytically, illicit samples predominantly contain etomidate, with related analogues typically not detected or detected only at trace levels. Some minor analogue signals are consistent with known pharmaceutical impurities, an important consideration for forensic interpretation [4]. Various analytical techniques (e.g., GC-MS, LC-MS/MS) can be found in the literature on the determination of etomidate and its analogues in biological matrices such as blood, plasma, urine, and hair. Given its predominance, inclusion of etomidate in analytical screening particularly for vape liquids and unknown depressant‑type products should be considered. Laboratories should remain alert to analogue detections while avoiding over‑interpretation of trace findings. The continuing spread of etomidate on illicit markets, even where etomidate analogues are controlled, highlights the need for ongoing early‑warning surveillance and toxicology‑informed risk assessment.
References and further reading
[2] Valk, B. I., & Struys, M. M. R. F. (2021). Etomidate and its analogs: A review of pharmacokinetics and pharmacodynamics. Clinical Pharmacokinetics, 60 (10), 1253–1269. https://doi.org/10.1007/s40262-021-01038-6
[3] Karunarathna, I. (2025). Etomidate: Pharmacology, clinical applications, monitoring, and considerations in anesthetic practice. UVA Clinical Research Lab 2025 © UVA Clinical Anaesthesia and Intensive Care ISSN 2827-7198.
[5] Chen, Y., Wu, L., Lang, B., Zhang, W., & Chen, S. (2025). Recent progress in the development of etomidate analogues. Frontiers in Pharmacology, 16, 1614865. https://doi.org/10.3389/fphar.2025.1614865
[6] Jiang, X., Yin, Q., Deng, X. et al. (2024) Advance of a new etomidate analogue – methoxyethyl etomidate hydrochloride (ET-26) for anesthesia induction in surgical patients. APS 2, 22 (2024). https://doi.org/10.1007/s44254-024-00062-6