Abstract
Materials and methods
Results and discussion
Acknowledgements
Author: H. L. TOOKEY, G. F. SPENCER, M. D. GROVE , J. A. DUKE
Pages: 49 to 57
Creation Date: 1975/01/01
Two varieties of opium poppy were grown under irrigation for one season and their capsules analysed for morphine content by gas chromatography. Harvest of the capsules at 29-33 days after flowering (mature seed) gave 6.70 kg/ha morphine as compared to 5.36 kg/ha morphine in capsules harvested at 4-6 days after flowering. With constant row spacing (30 cm between double rows on ridges 102 cm apart), a plant spacing of 6 cm gave 20 per cent more morphine than either 12- or 18-cm spacing. There was no statistical difference in morphine content between a blue- and a white-seeded variety.
* * *
Agronomic aspects of the opium poppy, Papaver somniferum L., are extensively documented in the foreign literature, as evidenced by a recent bibliography [ 5] . In contrast, corresponding studies in the United States are quite limited, because it is now illegal to grow opium poppies without rigorous licensing and security. A hundred years ago small experimental plots were cultivated in five states, with California and Arizona being the most promising areas [ 1] . Other agronomic work in the United States is virtually non-existent although Mika [ 7] did report a detailed study of the morphine content of poppies grown in a greenhouse under controlled temperature and photoperiods. He found that whereas green capsules had the highest morphine concentration 5 to 7 days after flowering, mature dry capsules collected 33 to 35 days after flowering had the largest total content of morphine.
In attempting to apply literature information on P. somniferum to a field situation, several problems arise. The most obvious ones are differences in soil, nutrients, climatic conditions, and methods used to assay for medicinal alkaloids. Somewhat less obvious is that much of the earlier work pertains to the production of gum opium. For several decades, increasing emphasis has been placed on industrial processes involving mechanical harvesting and direct extraction of mature capsular material or poppy straw [ 2] [ 4] [ 10] . Advantages of an extraction process include less hand labour and better control against diversion of alkaloids to illicit drug traffic. Analytical results may show either a decline in morphine as the capsule matures [ 6] [ 9] or an increase in morphine concentration [ 3] [ 8] . Such results are difficult to evaluate because they are often given in terms of alkaloid concentration rather than in absolute amount. When one considers a direct extraction process, the amount of morphine per unit area of land becomes important.
A single-season study was made of several factors that affect morphine content of P. somniferum grown under irrigation. We report here the effects of three different plant spacings and two stages of maturity of blue- and white-seeded poppies on the morphine content of their capsules.
Two varieties of P. somniferum, blue-seeded B-40-6-3 and white-seeded M-89, were planted on irrigated plots of silty clay loam in Arizona on 7 November 1973. The seed was drilled in double rows 30 cm apart on the tops of ridges with a between-ridge spacing of 102 cm. After germination and emergence, the plants were thinned on 14 December to stand 6, 12, or 18 cm apart within the rows. Irrigation was accomplished by flooding between the ridges. A total of approximately 91 cm of water was applied periodically during 17 weeks as dictated by stress, but not thereafter. Fertilizer was applied before planting at per hectare rates of 112.2 kg P 2O 5 and 56.1 kg N. Later applications were 56.1 kg N on 11 January, 56.1 kg N on 12 February, and 84.2 kg N on 7 March.
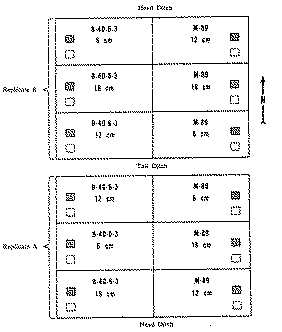
Arrangement of the 12 experimental plots (42.6 X 18.3-m each) is shown in figure 1. The positions of the varieties and replicates were assigned arbitrarily; spacing were chosen at random.
For a pilot study of maturity, 10 terminal capsules plus 25-26 cm peduncles were cut from each of the 12-cm spacing plots of the M-89 variety.Capsules were first collected on the day of petal drop and at intervals thereafter.
The measurement of plant maturity used for the variety-spacing study was days after peak flowering, which was defined as the time when the number of buds equalled the number of petalless flowers. Capsules were hand cut with attached 16-18 cm peduncles over 9.3 m 2(100 sq. ft.) areas in each plot. For the first or opium-stage harvest, the areas were 3.05 X 3.05 m, 3.05 m in from the southwest corners of the B-40-6-3 plots or the southeast corners of the M-89 plots (figure 1). The areas for the second harvest, that with mature seeds, were 3.05 m north of the areas first harvested.
The opium, or green capsule, stage of the blue-seeded (B-40-6-3) variety was harvested on 3 April 1974, 6 days after peak flowering. The white-seeded (M-89) variety was cut on 6 April, 4 days after peak flowering. Because the opium-stage harvests were an idealized field trial, buds and flowers were not collected. The percentage of dry solids in the material ranged from 12.2 to 14.1 per cent. At the mature-seed stage all capsules within the selected areas were harvested on 30 April, 33 days after peak flowering for the B-40-6-3 variety and on 1 May, 29 days after peak flowering for the M-89 variety. These samples consisted of 60-70 per cent mature, dry capsules (plus peduncles) and 30-40 per cent green or partly dry capsules with some latex exuding from the cut end of the peduncles. Those plants left in the field reached complete maturity 10 days later.
|
mature-seed harvest area; within-row spacings for each replicate are given in centimetres.
|
|
opium-stage harvest area;
|
The capsules with attached peduncles from each harvest stage were placed in plastic bags and refrigerated with dry ice during air transportation to Peoria, I11. The samples were then stored frozen until used. To simulate field curing of the mature-seed samples, they were removed from storage, spread in a single layer, and dried at 31-33 C under approximately 2,000 foot-candles of light for 11 hr per day. Drying was complete within 3 days: After drying, the solids content ranged from 89.9 to 92.2 per cent.
Three random subsamples of 12-20 capsules each (plus peduncles) were taken from each harvest sample, except for the pilot study of maturity in which 10 terminal capsules were used. The subsamples from the opium-stage harvest (approximately 300 g) were placed in hot (70 C) acidified water (500 ml, made to pH 2 with HCl) and blended to a purée in a Waring Blendor. 1Mature-seed samples (approximately 60-80 g) were blended at first with 800 ml of acidified water. The purées were stirred, heated to 70 C, and then filtered through cheesecloth with suction. Three additional 20-min extractions with 400-450 ml of hot acidified water were made to ensure removal of all the alkaloids.
For our analyses, run in duplicate, a 1 per cent aliquot of the extract (20-25 ml) was placed on a column of Amberlite XAD-2 nonionic resin. The alkaloidal material was eluted with methanol and CHCl 3-methanol (3:1) (11). The eluate was concentrated to dryness under reduced pressure and redissolved in 1 ml of chloroform containing 1 mg of the gas-liquid chromatography (GLC) internal standard, methyl lignocerate. A few drops of N, N-dimethylformamide aided dissolution. A portion of this solution was withdrawn, partially concentrated under a stream of nitrogen, and treated with 150 μl of Regisil RC-1 [bis- N, N-(trimethylsilyl) trifluoroacetamide, Regis Chemical Co.]. The silylation mixture was allowed to stand at 50 C for 15 min. GLC analysis of the silylated alkaloids was made with a Bendix Model 2600 instrument equipped with glass columns (4 ft x 1/8 in. ID) packed with 3 per cent Dexil-300 on Gas Chrom Q (Applied Science Laboratories). The injection port was held at 240 C, the flame ionization detectors were kept at 285 C, and the column oven temperature was programmed as follows: Initial hold at 190 C for 15 min; 190 to 195 C at 0.5 C/min, and 195 to 280 C at 3 C/min. Peak areas were integrated by computer and the results were printed out directly as milligrams of alkaloid present. Chromatographic responses of individual alkaloids were previously determined by analysis of standard solutions of pure alkaloids at various concentrations compared to the internal standard. Identification of alkaloids was routinely based on their retention times relative to that of the internal standard. Identification was confirmed periodically by mass spectrometric analysis of the silylated alkaloids in the GLC column effluent. Although the five major alkaloids - morphine, codeine, thebaine, papaverine, and narcotine - could be adequately separated by this method, only data for the most important alkaloid, morphine, are presented here.
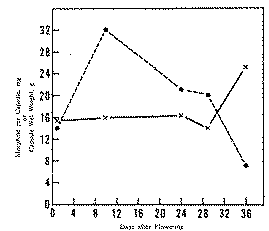
The pilot study of maturation of terminal capsules (figure 2) shows that morphine content per capsule remains relatively constant for at least 24 days after flowering. At the mature, dry stage harvested on day 36, however, morphine content increased more than 50 per cent. The significance of this increase in terminal capsules is not clear and bears further study since a comparable increase was not observed in the variety-spacing studies. Instead, morphine content per capsule increased only 5.8 per cent in the variety-spacing samples from these same plots when all capsules were analysed.
1 The mention of firm names or trade products does not imply that they are endorsed or recommended by the U.S. Department of Agriculture over other firms or similar products not mentioned.
Effects of maturity and plant spacing on morphine content 53
Morphine was analysed in both blue- and white-seeded varieties planted with 6-, 12-, and 18-cm spacing and harvested at two stages of maturity (tables 1 and 2). The times of harvest correspond to those selected by Mika (7). The percentage of buds and flowers not collected at the opium-stage harvest is given in table 3. Although the buds and flowers were rejected in order to have a more homogeneous sample, they might be collected in an actual harvest. In a calculation to show how much morphine could be lost by their exclusion, it was assumed that the buds and flowers contain no more morphine than the harvested capsules (6). This calculated maximum amount of morphine was added to kg/ha values of table 1 to give the adjusted morphine yield in table 3.
At the second harvest, the capsules contained mature seed, which comprised about half the total weight. For convenience, the seed was included in the extraction and analysis. To assure ourselves that the seed did not contribute morphine to the results, seed was separated from some samples and subjected separately to extraction and analysis. The GLC curve from the seed alone did show a peak at the retention time for morphine. However, mass spectrometry demonstrated that the peak was not due to morphine. Nonetheless, inclusion of the seed peak in the routine calculation would, for example, add 1.62 mg "morphine" to the 228 mg morphine found in the corresponding capsular material. Another experiment gave 0.49 mg "morphine" from seed and 296 mg morphine from the corresponding capsular material. Since these values indicate that inclusion of the seed material as morphine introduced an error of less than 0.5 per cent, seed was not separated before extraction.
An analysis of variance of the morphine data showed the greatest variability between replicate plots, suggesting some unknown non-random effect associated with the plots. The results are conveniently grouped in figure 3, where significant differences are shown at P <0.01. Since this experiment is factorial, the means shown in figure 3 are averaged over all levels of factors not under consideration. For example, the colour means are averaged over all conditions of spacing and maturity. The "adjusted morphine" from table 3 is not shown; however, that mean value for morphine is 6.01 kg/ha, which is still significantly lower than the mean of 6.70 kg/ha at the mature-seed stage (P < 0.05).
|
Morphine |
|||||
|---|---|---|---|---|---|
|
Variety and
spacing
|
Number of capsules harvested
|
Weight
(as is),
kg
|
Yield,
kg/ha a
|
Per capsule analysed,
mg
|
Dry
solids,
(percentages)
|
|
B-40-6-3 Blue b
c
|
|||||
|
6 cm Ac
|
555 | 8 051 | 5.91 | 13.9 | 0.59 |
|
Bc
|
476 | 7 825 | 6.11 | 15.1 | 0.60 |
|
12 cm Ac
|
453 | 7 711 | 5.36 | 13.6 | 0.55 |
|
B
|
554 | 9 526 | 5.07 | 10.2 | 0.47 |
|
18 cm A
|
334 | 6 464 | 3.45 | 12.3 | 0.42 |
|
B
|
439 | 8 278 | 6.19 | 15.8 | 0.61 |
|
M-89 White d
d
|
|||||
|
6 cm A
|
601 | 8 732 | 5.68 | 13.5 | 0.47 |
|
B
|
592 | 8 448 | 5.99 | 11.1 | 0.56 |
|
12 cm A
|
496 | 8 023 | 5.32 | 13.2 | 0.45 |
|
B
|
531 | 7 768 | 4.96 | 11.1 | 0.45 |
|
18 cm A
|
433 | 7 088 | 4.38 | 13.3 | 0.48 |
|
B
|
510 | 7 768 | 5.96 | 13.4 | 0.55 |
a Each value is the average of duplicate determinations of 3 subsamples.
b Harvested 3 April 1974, 6 days after peak flowering.
c These harvest areas were moderately lodged.
d Harvested 6 April 1974, 4 days after peak flowering.
|
Morphine |
|||||
|---|---|---|---|---|---|
|
Variety and
spacing
|
Number of capsules harvested
|
Weight
(as is),
kg
|
Yield,
kg/ha a
|
Per capsule
Analysed,
mg
|
Dry
solids, b
(percentages)
|
|
B-40-6-3 Blue c
c
|
|||||
|
6 cm A
|
640 | 2672 | 7.44 | 14.4 | 0.33 |
|
B
|
555 | 2812 | 7.67 | 15.5 | 0.33 |
|
12 cm A
|
554 | 2838 | 5.52 | 11.4 | 0.23 |
|
B
|
525 | 2623 | 6.08 | 12.0 | 0.28 |
|
18 cm A
|
467 | 2539 | 6.61 | 15.8 | 0.30 |
|
B
|
527 | 2670 | 6.16 | 13.6 | 0.25 |
|
M-89 Whited
d
|
|||||
|
6 cm A
|
686 | 2474 | 8.05 | 13.7 | 0.39 |
|
B
|
737 | 2757 | 7.54 | 12.7 | 0.32 |
|
12 cm A
|
554 | 2060 | 6.07 | 13.6 | 0.35 |
|
B
|
637 | 2643 | 6.30 | 12.1 | 0.28 |
|
18 cm A
|
540 | 2063 | 5.60 | 12.2 | 0.32 |
|
B
|
596 | 2420 | 7.38 | 13.8 | 0.36 |
a Each value is the average of duplicate determinations of 3 subsamples.
b Seeds are present; for approximate percentage morphine in the capsular material without seed, multiply this value by 2.
c Harvested 30 April 1974, 33 days after peak flowering.
d Harvested 1 May 1974, 29 days after peak flowering.
|
Variety and
spacing
|
Buds and flowers
not picked
percentage of capsules
harvested
|
Adjusted
morphine, a
kg/ha
|
|
B-40-6-3 Blue
|
||
|
6 cm A
|
15.3 | 6.81 |
|
B
|
8.8 | 6.65 |
|
12 cm A
|
11.7 | 5.98 |
|
B
|
8.3 | 5.49 |
|
18 cm A
|
19.5 | 4.12 |
|
B
|
18.5 | 7.34 |
|
M-89 White
|
||
|
6 cm A
|
3.2 | 5.86 |
|
B
|
10.6 | 6.62 |
|
12 cm A
|
15.7 | 6.16 |
|
B
|
8.5 | 5.38 |
|
18 cm A
|
17.1 | 5.14 |
|
B
|
11.2 | 6.62 |
a Assuming morphine/bud or flower = morphine/capsule; for details, see text.
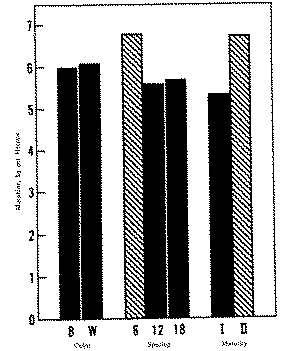
Essentially, there is no difference between the blue- and white-seeded varieties as grown under our conditions (figure 3). There is some advantage to having the denser plant population (6-cm spacing), although the 12- and 18-cm spacings cannot be statistically distinguished. There is, however, 25 per cent more morphine per hectare if plants are left to mature in the field rather than being harvested at an opium stage. A comparison of all the morphine-per-capsule values shows little increase from first to second harvest (13.0 mg/capsule v. 13.4 mg/capsule or 3 per cent). Thus the major contribution to the increase in morphine content is the 17.5 per cent increase in the number of capsules between the two harvests. An apparent decrease from 0.52 to 0.31 per cent in the average morphine content as a percentage of dry solids is somewhat misleading. Since the material harvested at maturity is approximately 50 per cent seed by weight, after separation of the seed the remaining capsular material would have a morphine concentration of about 0.62 per cent.
The mature-seed harvest has several distinct advantages over an earlier harvest: first, the material is dry and easy to harvest with conventional equipment; second, dry material offers minimum storage problems; and third, valuable seeds may be separated as a by product.
Appreciation is expressed to Dr. Q. Jones for helpful discussions and guidance, Dr. W. F. Kwolek for performing the analysis of variance, Mr. J. A. Park for growing the plants, and Mr. R. D. Plattner for developing the computer programme.
Anon. The opium poppy. Agric. Rep. 1870, U.S. Dept. Agric., Washington, D.C., pp. 206-210, 1871.
002O. L. Brekke, H. G. Maister, G. C. Mustakas, L. Van Ermen, M. C. Raether, and C. T. Langford. Morphine recovery from a 2-butanol extract of opium poppy meal. Ind. Eng. Chem . 50:1733-1736, 1958.
003E. S. Bunting, Changes in the capsule of Papaver somniferum between flowering and maturity. Ann. Appl. Biol . 51:459-471, 1963.
004J. A. Duke, Utilization of Papaver. Econ. Bot . 27(4):390-400, 1973.
005J. A. Duke, C. R. Gunn, E. E. Leppik, C. F. Reed, M. L. Solt, and E. E. Terrell. Annotated bibliography on opium and oriental poppies and related species. ARS-NE-28, U.S. Dept. Agric., Washington, D.C., 349 pages, 1973.
006K. Heydenreich, R. Miram, and S. Pfeifer. The alkaloid metabolism of Papaver somniferum L. Sci. Pharm . 29(4):221-249, 1961.
007E. S. Mika, Studies on the growth and development and morphine content of opium poppy. Bot. Gaz . 116:323-339, 1955.
008R. Miram and S. Pfeifer. Fluctuations in the content of alkaloids of poppy plants during a period of vegetation. Sci. Pharm . 27(1-2):34-53, 1959.
009M. Penka. Changes in water economy and morphine content in irrigated and non-irrigated plants of Papaver somniferum L. Acta. Univ. Agric., Brno Fac. Agron . 16:579-790, 1968.
010P. Popov, Y. Dimitrov, S. Georgiev, and L. S. Iliev. Indigenous and foreign poppy varieties characterized by the morphine content of their dry capsules. Bulletin on Narcotics XXV: 3, 51-56, 1973.
011D. Smith, D. W., T. H. Beasley, R. L. Charles, and H. W. Ziegler. J. Pharm. Sci . 62:1691-1694, 1973.