Background
Synthetic cathinones are β-keto phenethylamines and chemically similar to amphetamine and methamphetamine [1]. Cathinone, the principal active ingredient in the leaves of the khat plant (catha edulis), can be considered as the prototype from which a range of synthetic cathinones have been developed. Internationally controlled substances in this group are cathinone, methcathinone, cathine and pyrovalerone. Cathinone and methcathinone are listed in Schedule I of the 1971 Single Convention on Psychotropic Substances, cathine in Schedule III and pyrovalerone in Schedule IV.
Synthetic cathinones appeared in drug markets in the mid-2000s. In 2005, methylone, an analogue of MDMA, was the first synthetic cathinone reported to the European Monitoring Centre on Drugs and Drug Addiction (EMCDDA). In 2007, reports of 4-methylmethcathinone (mephedrone) use emerged, first in Israel and then in other countries and regions, including Australia, Scandinavia, Ireland and the United Kingdom [2]. Mephedrone was reportedly first synthesized in 1929 [3].
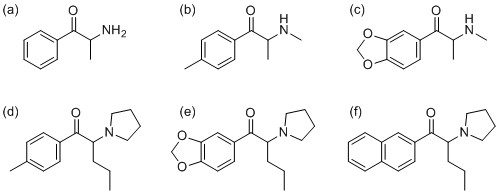
Figure 1 Synthetic cathinones: (a) cathinone, (b) 4-methylmethcathinone (mephedrone), (c) methylone,
(d) pyrovalerone, (e) 3,4-methylenedioxypyrovalerone, (f) naphyrone
Typically, synthetic cathinones have an amphetamine-type analogue, i.e. cathinone, ephedrone, and methylone are structurally related to amphetamine, methamphetamine and MDMA respectively. While little comprehensive pharmacological data is available for many of the synthetic cathinones, 4-methylmethcathinone (mephedrone) has been extensively studied[4-7].
Other synthetic cathinones recently identified in the drug market are analogues of pyrovalerone (3,4-methylenedioxypyrovalerone and naphyrone). For instance, 3,4-methylenedioxypyrovalerone (MDPV), first synthesized in 1969 [8], emerged in 2007 as a new psychoactive substance in Germany [9]. In 2008, it was first reported to the European Early Warning System by the United Kingdom and by Finland, after being associated with adverse health effects [10].
Other synthetic cathinones used as NPS from 2010 onwards, are methylone, butylone, 4-methylethcathinone, 4-fluoromethcathinone, naphyrone, 3-fluoromethcathinone, methedrone, and, to a lesser extent, 3,4-dimethyl- methcathinone, α-pyrrolidinopentiophenone (α-PVP), buphedrone, pentedrone and α-pyrrolidinopropiophenone (α-PPP).
While some synthetic cathinones such as methylone had been patented as antidepressant and antiparkinsonian agents [11], very few have been exploited clinically predominantly on account of their abuse and dependence potential. For instance, whereas diethylcathinone (amfepramone) is used as an appetite suppressant, pyrovalerone, first synthesized in 1964 and marketed for use as an appetite suppressant and in the treatment of chronic fatigue, was later withdrawn due to abuse and dependency in users [12]. Apart from cathinone, the only cathinone derivatives under international drug control are amfepramone, methcathinone and pyrovalerone [13].
Description
Synthetic cathinones are frequently found in products sold as ‘research chemicals’, ‘plant food’, ‘bath salts’ or ‘glass cleaner’ and are usually sold in powder, pill or capsule form. Mephedrone (‘m-cat’, ‘meph’, ‘drone’ or ‘miaow’) and methylone (‘explosion’ or ‘top cat’) are usually available as white or brown powders or in the form of pills that are often sold as ‘ecstasy’. Most synthetic derivatives are ingested but may be injected. Mephedrone is commonly nasally insufflated, injected, ingested by swallowing a powder wrapped in paper (‘bombing’), or mixed in a drink.
Synthetic cathinones act predominantly as central nervous system stimulants. Stimulants mediate the actions of dopamine, norepinephrine and/or serotonin, mimicking the effects of traditional drugs such as cocaine, amphetamine, methamphetamine, and ecstasy.
Reported adverse effects
Much of the current knowledge on health related effects comes from user reports and clinical observations. Further research is needed to provide evidence of short and long-term health risks and the addiction potential associated with the use of these substances.
Whereas cardiac, psychiatric, and neurological signs are some of the adverse effects reported by synthetic cathinone users, agitation, ranging from mild agitation to severe psychosis, is the most common symptom identified from medical observations [14]. Studies of patients under the apparent influence of mephedrone have also shown that synthetic cathinones present similar sympathomimetic effects (including tachycardia and hypertension as well as psychoactive effects) to similar amphetamine derivatives [15]. In a student survey, more than half of those who had taken mephedrone reported adverse effects associated with the central nervous system, nasal/respiratory system and cardiovascular system [16]. The first fatality related to the sole use of mephedrone, confirmed by toxicological analysis, was reported in Sweden in 2008 [17]. Most fatalities associated with the use of mephedrone involved the use of other substances [18]. Deaths associated with the use of other synthetic cathinones include two deaths related to methedrone [19] and two other deaths related to butylone [20].
The Finnish Poisons Information Centre reported 33 calls regarding exposures to MDPV during the period of January 2008 to October 2009. Post mortem toxicological analysis confirmed 6 deaths related to MDPV between 2009 and 2010, although in most of the cases the presence of other drugs was also detected [21]. A report from the United States provided details on the case of 35 patients who visited an Emergency Department over a 3-month-period after ingesting, inhaling or injecting substances sold as ‘bath salts’ and asserted that these products could contain stimulant compounds such as MDPV or mephedrone. One person was dead upon arrival at the emergency department. The toxicological analysis revealed a high level of MDPV, along with cannabis and prescription drugs, but the autopsy results revealed MDPV toxicity to be the primary factor contributing to death [22].
For further details regarding chemical structures, production and analysis see also the UNODC manual Recommended methods for the Identification and Analysis of Synthetic Cathinones in Seized Materials (click here).
References
[1] European Monitoring Centre for Drugs and Drug Addiction (EMCDDA), “Synthetic cathinones”, Drug Profiles accessed at www.emcdda.europa.eu.
[2] Kelly, J.P. “Cathinone derivatives: A review of their chemistry, pharmacology and toxicology”, Drug Testing and Analysis 3 (2011): 439-453.
[3] Saem de Burnaga Sanchez, J. “Sur un homologue de l’ephedrine”, Bulletin de la Société Chimique de France 45 (1929): 284-86. [4] Meyer, M.R., Wilhelm, J., Peters, F.T., Maurer, H.H. “Beta-ketone amphetamines: studies on the metabolism of the designer drug mephedrone and toxicological detection of mephedrone, butylone and methylone in urine using gas chromatography-mass spectrometry”, Analytical and Bioanlytical Chemistry 396 (2010):1225 – 1233.
[5] Winstock, A., Mitcheson L., Ramsey J., Davies S,. Puchnarewicz M., Marsden J. “Mephedrone: use, subjective effects and health risks.”, Addiction 106 (2011):1991-1996.
[6] Capriola, M., “Synthetic cathinone abuse”, Clinical Pharmacology: Advances and Applications 5 (2013):109-115.
[7] Pedersen, A.J., Reitzel, L.A., Johansen, S.S., Linnet, K. “In vitro metabolism studies on mephedrone and analysis of forensic cases.”, Drug Testing and Analysis 6 (2013):430-8.
[8] “Boehringer Ingelheim Patent for MDPV” accessed at http://catbull.com/alamut/Bibliothek/Boehringer_MDPV_Patent.htm)
[9] In 2007, MDPV was first identified in a seizure in Germany. See also Westphal, F., et. al., “Mass and NMR spectroscopic characterization of 3,4-methylenedioxypyrolvalerone: a designer drug with apyrrolidinophenone structure”, Forensic Science International 190 (2009): 1-8.
[10] European Monitoring Centre for Drugs and Drug Addiction (EMCDDA) and the European Police Office (EUROPOL), “EMCDDA–Europol 2010 Annual report on the implementation of Council Decision 2005/387/JHA”, Lisbon, May 2011.
[11] Jacob, P., Shulgin, A.T., (Neurobiological Technologies Inc, USA) Patent WO9639133 (1996), 19 pp. CA 126: 117961.
[12] Meltzer, P., Butler, D., Deschamps, J.R. and Madras, B.K., “(4-methylphenyl)-2-pyrrolidin-1-yl-pentan-1-one (Pyrovalerone) analogues: a promising class of monoamine uptake inhibitors”, Journal of Medicinal Chemistry 49 (2006): 1420-32; other cathinone derivatives, such as amfepramone and bupropion are or have also been used as active pharmaceutical ingredients.
[13] Cathinone and methcathinone are listed in Schedule I, and amfepramone and pyrovalerone in Schedule IV, of the 1971 United Nations Convention on Psychotropic Substances.
[14] J.M. Prosser and L.S. Nelson, “The toxicology of bath salts: a review of synthetic cathinones”, The Journal of Medical Toxicology 2012, 8(1): 33-42.
[15] The term sympathomimetic refers to a pharmacologic agent that mimics the effects of stimulation of organs and structures by the sympathetic nervous system. It functions by occupying adrenergic receptor sites and acting as an agonist or by increasing the release of the neurotransmitter norepinephrine at postganglionic nerve endings; European Monitoring Centre for Drugs and Drug Addiction (EMCDDA), “Synthetic cathinones”, Drug Profiles accessed at www.emcdda.europa.eu.
[16] Dargan, P.I., Albert, S. and Wood, D.M., “Mephedrone use and associated adverse effects in school and college/university students before the UK legislation change”, Oxford Journal of Medicine 103.10 (2010): 875-9.
[17] Gustavsson, D. and Escher, C., “Mephedrone – internet drug which seems to have come and stay. Fatal cases in Sweden have drawn attention to previously unknown substance”, Lakartidningen 1.6.43 (2009): 2769-71.
[18] The death of a 46-year old man in the UK was caused by a combination of mephedrone and heroin. Other cases reported from Scotland revealed the presence of other substances along with mephedrone. See also Dickson, A.J., Vorce, S.P., Levine, B. and Past M.R., “Multiple-drug toxicity caused by the coadministration of 4-methylmethcathinone (mephedrone) and heroin”, Journal of Analytical Toxicology 34.3 (2010): 162-8; Torrance, H. and Cooper, G., “The detection of mephedrone (4-methylmethcathinone) in 4 fatalities in Scotland”, Forensic Science International 202 (2010): 62-3.
[19] Wikström, M., Thelander, G., Nyström, I. and Kronstrand, R, ‘Two fatal Intoxications with the New Designer Drug Methedrone (4-Methoxymethcathinone)’, Journal of Analytical Toxicology, 2010, 34, 594-98.
[20] N. Carter, et.al., “Deaths associated with MBDB misuse”, Journal of Legal Medicine 2000, 113: 168–70.
[21] National Institute for Health and Welfare (THL), “MDPV in Finland”, Finland, 2010 accessed at https://ewsd.wiv-isp.be/Publications%20on%20new%20psychoactive%20substances/MDPV/MDPV%20facts%20from%20Finland.pdf
[22] Centers for Disease Control and Prevention (CDC), “Emergency Department visits after use of a drug sold as “bath salts”, Morbidity and Mortality Weekly Report (MMWR) May 20, 2011, vol. 60(19): 624-627.