Background
The appearance of ‘herbal highs’ in the market is not a new phenomenon. Such products usually consisted of plant mixtures with little psychoactive effects. Since 2004, however, the composition of these herbal products seems to have substantially changed to include potent new psychoactive compounds known as synthetic cannabinoids.
Research on the mechanism of cannabis activity dates back several decades when molecules with similar behaviour to Δ9-tetrahydrocannabinol (THC) were first examined. A synthetic analogue of THC, ‘HU-210’, was first synthesized in Israel in 1988 [1] and is considered to have a potency of at least 100 times that of THC. Due to its similar chemical structure to THC, ‘HU-210’ is regarded as a ‘classical cannabinoid’ and has been found in synthetic cannabinoids sold in the United States and other countries.
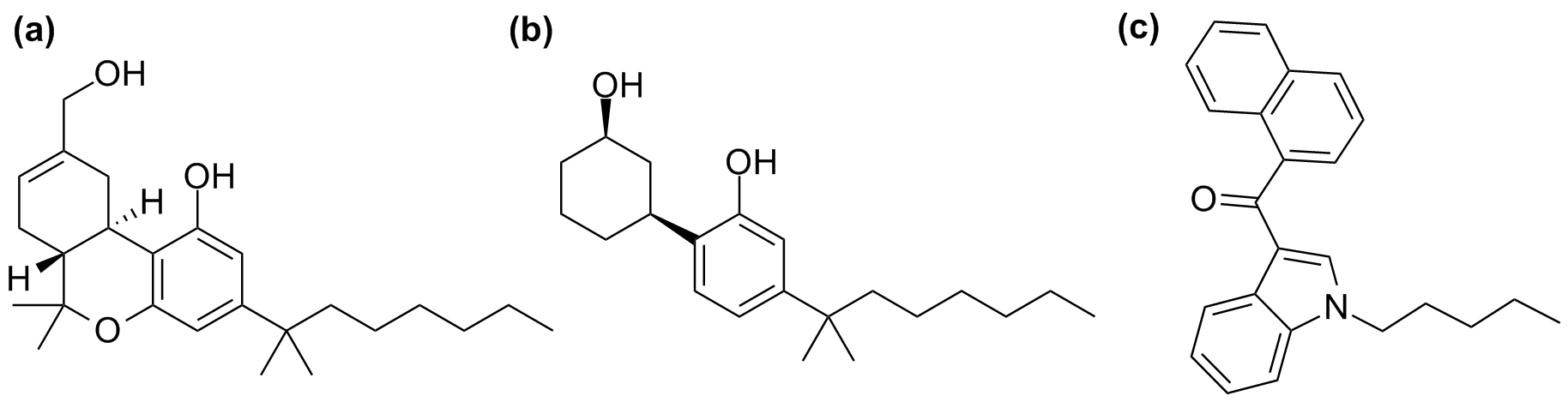
Figure 1 Depiction of the most common synthetic cannabinoids: (a) HU-210 (classical cannabinoid), (b) CP-47,497 (non-classical cannabinoid), (c) JWH-018 (aminoalkylindoles)
Another group of synthetic cannabinoids termed ‘non-classical’ include cyclohexylphenols or 3-arylcyclohexanols (‘CP’compounds). ‘CP’ compounds, were developed as potential analgesics by Pfizer, a pharmaceutical company, in the 1980s.
Other structurally dissimilar varieties of synthetic cannabinoids unrelated to THC have also emerged on the market. These include aminoalkylindoles, such as naphthoylindoles (e.g. JWH-018), phenylacetylindoles (e.g. JWH-250), and benzoylindoles (e.g. AM-2233) [2]. JWH-018, arguably the most widely known synthetic cannabinoid, belongs to the group of aminoalkylindoles and is considered to be three times as potent as THC. Aminoalkylindoles are by far the most prevalent compounds found in herbal products laced with synthetic cannabinoids. This is due to the fact that syntheses of aminoalkylindoles are less elaborate and complicated than syntheses of classical, non-classical or hybrid cannabinoids. The JWH-compounds had been previously developed as test compounds in the research of receptor-drug interactions by Professor John William Huffman [3] and his team in the United States.
Synthetic cannabinoids that have emerged more recently show an even greater structural diversity, e.g APINACA (AKB-48), an adamantyl indazole carboxamide, and AB-PINACA, an aminocarbonyl indazole carboxamide. It is possible that these newer compounds were developed to bypass some of the national legislative responses that had been put in place to counter the ‘earlier generations’ of synthetic cannabinoids.
While cannabis and THC are controlled under the international drug control treaties, none of the synthetic cannabinoids are currently under international control. However, several have been subject to control measures at the national level.
Description
Synthetic cannabinoids are usually added to plant material by soaking or spraying, but in some cases their solid form (crystalline powder) was added to plant material, leading to an inhomogeneous mixture. They are sold as ‘Spice Gold’, ‘Spice Silver’, ‘Spice Diamond’, ‘K2’, ‘Bliss’, ‘Black Mamba’, ‘Bombay Blue’, ‘Blaze’, ‘Genie’, ‘Zohai’, ‘JWH -018, -073, -250’, ’Kronic’, ‘Yucatan Fire’, ‘Skunk’, ‘Moon Rocks’, ‘Mr. Smiley’. They are usually smoked, but oral use has also been reported.
Synthetic cannabinoids act as Synthetic Cannabinoid Receptor Agonists (SCRA). These substances bear structural features that allow binding to one of the known cannabinoid receptors, and produce effects similar to those of delta-9-tetrahydrocannabinol (THC), the only known psychoactive component in cannabis. Synthetic Cannabinoids are often laced onto herbal products.
Reported adverse effects
While side effects of cannabis are well documented [4], data on human toxicity related to the use of synthetic cannabinoids remains limited. As with other NPS, products sold as synthetic cannabinoids often contain several chemicals in different concentrations, making it very difficult to determine substance-specific effects. Available knowledge on the toxicity of these compounds comes from scientific reports and clinical observations.
Health-related problems associated with the use of synthetic cannabinoids include cardiovascular problems and psychological disorders [5], and it appears that there may be carcinogenic potential with some of the metabolites of the substances contained in these products [6].
A study published in 2011 on the severe toxicity following synthetic cannabinoid ingestion suggested that JWH-018 could lead to seizures and tachyarrhythmia (irregular heartbeat) [7]. In a review of clinical reports from 2010, addiction and withdrawal symptoms similar to those seen with cannabis abuse were also linked to the use of synthetic cannabinoids [8]. An analysis of synthetic cannabinoids in ‘spice-like’ herbal blends highlighted the increasing number of reports on suicides associated with preceding use of these products [9].
For further details regarding chemical structures, production and analysis see also the UNODC manual Recommended methods for the Identification and Analysis of Synthetic Cannabinoid Receptor Agonists in Seized Materials (click here).
References
[1] Mechoulam, R., Lander, N., Breuer, A. and Zahalka, J. ‘Synthesis of the individual, pharmacologically distinct, enantiomers of a tetrahydrocannabinol derivative’, Tetrahedron: Asymmetry 1.5 (1990): 315-18.
[2] United Nations Office on Drugs and Crime (UNODC), Synthetic cannabinoids in herbal products, Vienna, 2011: 5; see also Hudson, S. Ramsey, J. ‘The emergence and analysis of synthetic cannabinoids’, Drug Testing and Analysis 3 (2011): 466–478.
[3] John W. Huffman is a US chemist and a retired professor of organic chemistry at Clemson University in the United States, whose research led to the synthesis of non-cannabinoid cannabimimetrics in the 1990s. Dr Huffman’s research group focussed on the synthesis of analogues and metabolites of THC with the aim to develop new pharmaceutical products for medical treatment.
[4] For more information see Hall, W. and Solowij, N., “Adverse effects of cannabis” Lancet 352 (Nov 1998): 1611-6; Ashton, C. H., “Adverse effects of cannabis and cannabinoids”, British Journal of Anaesthesia 83 (1999): 637-49.
[5] H. Müller, et.al., “Panic attack after spice abusein patient with ADHD”, Pharmacopsychiatry 43.4 (2010): 152-153; A. Mir, et.al., “Myocardial infarction associated with use of the synthetic cannabinoid K2”, Journal of Pediatrics 128.6 (2011): 1622-1627; S. Every-Palmer, “Synthetic cannabinoid JWH-018 and psychosis: an explorative study”, Drug and Alcohol Dependence 117 (2011): 152-157.
[6] C.Y. Lin, et.al., “Toxicity and metabolism of methylnaphthalenes: comparison with naphthalene and 1- Nitronaphthalene”, Toxicology 260 (2009): 16-27.
[7] J. Lapoint, et.al., “Severe toxicity following synthetic cannabinoid ingestion”, Clinical Toxicology (Philadelphia) 49 (2011): 760-64.
[8] I. Vardakou, C. Pistos and C.H. Spiliopoulou, “Spice drugs as a new trend: mode of action, identification and legislation”, Toxicology Letter 197 (2010): 157-162.
[9] Ludger, E., et.al., “Synthetic cannabinoids in ‘spice-like’ herbal blends: first appearance of JWH-307 and recurrence of JWH-018 on the German market”, Forensic Science International 222.1 (2012): 216-222.