Tolerance to cannabis
FIGURE 1
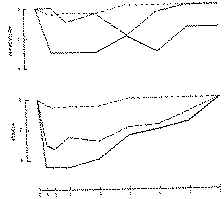
FIGURE 2 ---- 1ST DAY (THC, 5 MG/KG, I,V,) ---- 4TH DAY (THE AVERAGES OF 4 DOGS). ---- 8TH DAY
Dependence on cannabis
Summary
Bibliography
Author: Sükrü KAYMAKÇALAN
Pages: 39 to 47
Creation Date: 1973/01/01
It has generally been believed that cannabis produces neither tolerance nor physical dependence. Eddy, Halbach, Isbell and Seevers in their authoritative article described the characteristics of cannabis type drug dependence as follows: ( a) moderate to strong psychic dependence on account of the desired subjective effects, ( b) absence of physical dependence, so that there is no characteristic abstinence syndrome when the drug is discontinued, and ( c) little tendency to increase the dose and no evidence of tolerance (Eddy et al., 1965). Almost the same view of cannabis had been expressed by the WHO Expert Committee on Addiction Producing Drugs in its thirteenth report (1964).
One may find similar opinions even in the most recent textbooks of pharmacology. For example in the fifth edition of Goth's Medical Pharmacology it is stated that "neither physical dependence [on] nor tolerance [to marihuana] is known to develop" (Goth, 1970). Grollman and Grollman in the seventh edition of their Pharmacology and Therapeutics hold the view that "no great degree of tolerance to the drug develops and no abstinence syndrome follows withdrawal of the drug" (Grollman and Grollman, 1970). According to the fourth edition of the Pharmacological Basis of Therapeutics, edited by Goodman and Gilman, "there is general agreement that some degree of tolerance develops, but it is not associated with physical dependence, withdrawal symptoms or craving" (Goodman and Gilman, 1970).
Noting the paper by Eddy et al., 1965, concerning tolerance and physical dependence, the WHO Scientific Group on the Use of Cannabis observed that "these characteristics must now be re-examined because of subsequent findings".
Recent achievements in the synthesis of tetrahydrocannabinols made it possible to investigate the pharmacological properties of cannabis. Since delta-9-tetrahydrocannabinol (THC) produces in man the same physical as well as psychological effects as cannabis (Isbell, 1967) it is generally accepted that THC is the main active principle of cannabis. As a matter of fact, in cannabis preparations (marihuana or hashish) the concentration of THC is higher than in other cannabinoids (Mechoulam, 1970).
In this article, the author would like to mention some studies made during the last years on THC or cannabis in relation to tolerance and dependence and to discuss the results achieved.
Whether or not tolerance to cannabis develops in man has been a subject of debate for a long time. However it is known that in India some ganja and charas smokers consume, daily, very large amounts of cannabis and it is difficult to accept the consumption of such large doses unless some degree of tolerance developed (Report of a WHO Scientific Group, 1971). Recently Miras, leaving no place for doubt as to the development of tolerance in man, expressed his opinion on this subject with the following words:
"Hashish smokers I have known for twenty years are now able to smoke at least ten times as much as other people. If a beginner smoked the same quantity he would collapse" (Miras, 1970). According to Paton (1972), perhaps the best evidence for tolerance in man is still that in the LaGauardia report, where it was found that to produce a given degree of ataxia a dose three times as high was required for users compared with non-users.
During the last few years studies with cannabis extracts or THC disclosed the fact that tolerance to many effects of cannabis develops in different laboratory species.
Carlini (1968) found that cannabis extract strongly reduces the bar pressing behaviour and the rope climbing performance of rats; but after daily administrations, tolerance to these effects of cannabis developed in approximately two weeks. In our experiments with rats we have shown that THC produces analgesia (hot plate and phenyl-quinone tests), hypothermia and reduces the iodine (I 125) uptake of the thyroid glands. Tolerance developed to all these three effects in less than one week. We have observed that even after a single injection of THC (10mg/kg), tolerance to the analgesic effect lasted for more than one month (Kaymakçalan and Deneau 1971, 1972). (Figure 1.)

Tolerance to and dependence on cannabis 41
It is very well known that cannabis extracts produce ataxia in dogs and this response had been used for a long time as a biological assay in the activity control of cannabis preparations. Besides ataxia, general depression or drowsiness are other, easily observed signs of cannabis reaction in the dog. Giving scores for degrees of ataxia and depression, it is possible to evaluate these reactions in a quantitative way.
After daily administration of THC to dogs we have observed that, in 8 days, an almost complete tolerance developed to both ataxia and general depression (Kaymakçalan and Deneau, 1972). (Figure 2.)
The most characteristic effects of THC after a single administration in monkeys are ptosis of the eyelids, docility and loss of aggression. In chronic experiments tolerance to these effects developed in a few days. They reappeared after the dosage was increased, but lasted only for a short period of time (Deneau and Kaymakçalan, 1971).
Development of tolerance to THC has been demonstrated not only in mamals, but also in fish and birds. For example marihuana extract and THC inhibit the spontaneous aggressive behaviour of the male Siamese fighting fish ( Betta splendens). However after 8-10 exposures the fishes develop tolerance to the drugs (Gonzales et al., 1971). In pigeons conditioned to obtain food, THC causes dose-dependent decreases in the rates of key pecking and tolerance to this effect develops after a week of daily injections of THC (Frankenheim et al., 1970). It has been further demonstrated that a dose of THC, which completely disrupts food-reinforced responses in pigeons for many hours, can be increased more than 20-fold without disrupting this schedule controlled behaviour in tolerant birds (McMillan et al., 1970). These authors point out that the pattern of the development of tolerance to THC is similar to that seen in the same kind of behavioural experiments with morphine and methadone in pigeons. Moreover the degree of behavioural tolerance obtained in the pigeon was more similar to morphine tolerance than barbiturate tolerance, which is weaker. In neonatal chicks THC decreases locomotor activity, inhibits escape from a heat stimulus and produces hypothermia. Tolerance to all these behavioural and physiological effects develops in approximately two weeks time (Abel, 1972).
It is known that tolerance does not develop to all effects of morphine in the same degree of intensity. A similar pattern of tolerance might be applied also to THC. For example we have observed only a slight or questionable tolerance to the bradycardic effect of THC in rats after 16 days of daily administrations (Kaymakçalan and Sivil, 1972). It has been reported that following daily injections of THC in rats hypophagia and inhibition of normal growth last for at least three weeks (Manning et al., 1971). However in another study daily injections of cannabis in rats caused a maximum inhibition of food and water intake by the 4th and 6th day, and returned to its normal level on the llth day (Fernandes et al., 1971).
In dogs also there is no uniformity in all effects of tolerance. We have mentioned that an almost complete tolerance has developed to both ataxia and general depression in 8 days (figure 2). In these experiments only a slight or partial tolerance developed to the effects of THC on heart rate, respiration and body temperature in the same period (Kaymakçalan and Deneau, 1972).
Tolerance which develops to THC is not specific for cannabinoids. We have found that THC-tolerant rats present also tolerance to the analgesic effect of morphine (Kaymakçalan and Deneau, 1972). A similar cross-tolerance between alcohol and THC has also been reported (Newman et al., 1972). On the other hand subjects tolerant to LSD do not present cross-tolerance to THC (Isbell and Jasinski, 1969). These findings suggest that although THC in high doses may act as a hallucinogenic agent (Isbell at al., 1967), it is pharmacologically much closer to narcotic analgesics and alcohol than the other centrally acting substances and especially hallucinogens.
In chronic experiments with rhesus monkeys, not only tolerance, but also psychological and physical dependence to THC were clearly shown (Deneau and Kaymakçalan, 1971). Since these experiments are the first proofs for the possibility of physical dependence and withdrawal reactions due to cannabis in laboratory animals, some information will be given in this paper. The details of these experiments with monkeys have been presented elsewhere (Kaymakçalan, 1972).
Six rhesus monkeys have been prepared with intravenous catheters for self-administration of THC. The initial dose of THC was 0.1 mg/kg. During a period of 23 days, spontaneous self-administration of THC did not occur. Then the timer-controlled injections started and monkeys received the drug automatically four times a day. Gradually increasing the dose of THC from 0.1 mg/kg to 0.2 mg/kg and to 0.4 mg/kg in 24 days, monkeys were injected with the highest dose for 12 days. At the end of this period of 36 days' THC administration, the automatic injections were discontinued and all monkeys showed abstinence signs. Two of the six monkeys then initiated and maintained self-administration of THC
Following the first withdrawal of THC, one monkey was removed from the experiment for technical reasons (occlusion of the veins). For the remaining three monkeys which were not receiving THC, a cocaine solution was made available for self-administration. Two of them did not start to inject themselves with cocaine, but the third one initiated and maintained the cocaine self-administration. When cocaine was replaced by THC (0.4 mg/kg) in the drug reservoir of this animal, he continued to self-administer THC.
The three monkeys were allowed to self-administer THC for three to eight weeks. After the prevention of self-administration, all of them presented abstinence signs for the second time. The rate of self-administration of THC showed individual as well as daily fluctuations, varying in 24 hours between 5 to 30 times, mostly during the day. Abrupt cessation of drug availability caused characteristic withdrawal symptoms. Abstinence signs generally appeared within 12 hours and lasted for approximately five days.
The general picture of a THC-induced abstinence syndrome consists of the following symptoms: there is hyperirritability and increase in aggressiveness; tremors and twitches in the muscles are easily observed; sometimes the whole body of the animal shakes. Among the autonomic disturbances yawning, photophobia, piloerection and erection of the penis are most common. Several ejaculations may follow masturbation in male animals. During the whole period anorexia is striking. Sitting in propped-up positions, eating faeces or other unusual things, biting and licking fingers, pulling hair are other signs which are considered to be related to behavioural and probably sensory changes. Some reactions like staring in circles, grasping as if catching flies and slapping the cage wall, suggest the presence of hallucinations (figures 3-6). In three animals which were self-administering THC, pushing the button to receive the drug during the withdrawal period was repeatedly noticed.
The development of physical dependence on THC in monkeys might not seem to be applicable to man. It is common knowledge that marihuana smokers can easily give up marihuana, without showing any abstinence syndromes. However this may not be true for hashish users. As a matter of fact Fraser (1949) reported withdrawal symptoms in nine ganja smokers among Indian soldiers. They showed hyper-irritability, acute psychosis and masturbated almost continuously. At times most of them appeared to be visually and aurally hallucinated and the hallucinations were terrifying. According to Kielholz and Ladewig (1970), some hashish smokers who take hashish over a long period show withdrawal symptoms such as sweating, disturbed sleep, fine tremors, hypotony, anxiety and depression lasting for 5 to 7 days.
Research conducted on many different laboratory animals, including fish, birds, rats, dogs and monkeys, show that tolerance to THC does develop. These findings are in agreement with observations in man, especially in the heavy cannabis smokers in India and Greece.
Physical dependence on THC has been demonstrated in monkeys. A review of the literature indicates that withdrawal symptoms may also occur in heavy cannabis smokers.




Abel, E.L. (1972). Studies of tolerance to l-Δ 9-tetrahydrocannabinol (Δ 9-THC) in neonatal chicks. Fed. Proc. 31: 505 Abs.
Carlini, E. A. (1968). Tolerance to chronic administration of Cannabis sativa (marihuana) in rats. Pharmacology 1: 135.
Deneau G.A., and Kaymakçalan, S. (1971). Physiological and psychological dependence to synthetic Δ 9-tetrahydrocannabinol (THC) in rhesus monkeys. The Pharmacologist 13: 246.
Eddy, N.B., Halbach, H., Isbell, H., and Seevers, M.H. (1965). Drug dependence: Its significance and characteristics . Bull. Wld. Hlth. Org. 32: 721.
Fernandes, M., Rating, D., and Kluwe, S. (1971). The influence of subchronic tetrahydrocannabinol- and cannabis-treatment on food- and water-intake, body weight and body temperature of rats. Acta Pharmacol. et Toxicol. 29: Suppl. 4: 89.
Frankheim, J. M., McMillan, D. E., and Harris, L.S. (1970). Effects of l-Δ 9-and 1-Δ 9-trans-tetrahydrocannabinol on schedule-controlled behaviour of the pigeon . Fed. Proc. 29: 619.
Gonzalez, S. C., Matsudo, V. K. R., and Carlini, E. A. (1971). Effects of marihuana compounds on the fighting behaviour of siamese fighting fish (Betta splendens). Pharmacology 6: 186.
Goodman, L. S., and Gilman, H. (1970). The Pharmacological Basis of Therapeutics (1970). Fourth edition, p. 300. The McMillan Co., New York.
Goth, A. Medical Pharmacology (1970). Fifth edition, p. 282. The C.V. Mosby Co., Saint Louis.
Grollman, A., and Grollman, E. F. (1970). Pharmacology and Therapeutics (1970). Seventh edition, p. 218. Lea and Febiger, Philadelphia.
Isbell, H., Gorodetsky, C. W., Jasinski, D., Claussen, U., v. Spulak, F., and Korte, F. (1967). Effects of (-)- Δ 9-trans-tetrahydrocannabinol in man. Psychophormacologia 11: 184.
Isbell, H., and Jasinski, D. R. (1969). A comparison of LSD-25 with (-)-Δ 9-trans-tetrahydrocannabinol and attempted cross-tolerance between LSD and THC. Psychopharmacologia 14: 115.
Kaymakçalan, S. (1972). Physiological and psychological dependence on THC in rhesus monkeys. In: Cannabis and its derivatives, pp. 142-146. Edited by W. D. M. Paton and June Crown, Oxford University Press, London, 1972.
Kaymakçalan, S., and Deneau, G.A. (1971). Some pharmacological effects of synthetic Δ 9-tetrahydrocannabinol (THC). The Pharnmcologist 13: 247.
Kaymakçalan, S., and Deneau, G. A. (1972). Some pharmacologic properties of synthetic Δ 9-tetrahydrocannabinol (THC). Acta Medica Turcica, Supplementum: 1.
Kaymakçalan, S., and Sivil, S. (1972). The effects of acute and chronic administrations of THC on the heart rate in rat. Presented to the 22nd National Turkish Medical Congress, Oct. 3-7, Ankara.
Manning, F. J., Jr., MacDonough, J. H., Elsmore, T. F., Saller, C., and Sodetz, F. J. (1971). Inhibition of normal growth by chronic administration of Δ 9-tetrahydrocannabinol . Science 174: 424.
McMillan, L. S., Harris, L. S., Frankenheim, J. M., and Kennedy, J. S. (1970). 1-Δ 9-tetra-hydrocannabinol in pigeons: Tolerance to the behavioral effects. Science 169: 501.
Mechoulam, R. (1970). Marihuana Chemistry. Science 168: 1159.
Miras, C. J. (1970). In The Botany and Chemistry of Cannabis, p. 203. Edited by C. R. B. Joyce and S. H. Curry. J. and A. Churchill, London.
Newman, L. M., Lutz, M. P., Gould, M. H., and Domino, E. F. (1971). Δ 9-tetrahydro-cannabinol and ethyl alcohol: Evidence for cross-tolerance in the rat. Science 175: 1022.
Paton, W. D. (1972). Guide to Drugs 5: Cannabis. Drugs and Society 1: No. 9: 17.
Report of a WHO Scientific Group (1971). The use of cannabis. Wld. Hlth. Org. Tech. Rep. Ser. 478, p. 33.
WHO Expert Committee on Addiction-Producing Drugs (1964). Thirteenth Report, Wld. Hlth. Org. Techn. Rep. Ser. 273.