Preparation of marijuana cigarettes
Production of marijuana smoke condensate samples
Monitoring of the smoke condensate
Characterization of marijuana cigarettes
Summary
Acknowledgements
Author: A. R. PATEL, Gio B. GORI
Pages: 47 to 54
Creation Date: 1975/01/01
The increase in the use of psychoactive drugs, especially the smoking of marijuana, in recent years has been of great concern to the health authorities in the United States. This circumstance has brought forward both the advocates and opponents for the legalization of the use of marijuana. The differences of opinion are based on the short- and long-term effects of the drug. Although the literature on marijuana is voluminous [ 1] , as often as not the reports are conflicting, often erroreous and are based on rumor, anecdote or second hand observation. The lack of reliable data about the pharmacological and clinical effects of marijuana is primarily due to the fact that until very recently little was known about the chemistry of marijuana [ 2] [ 3] [ 4]
Since 1969 the National Cancer Institute has been engaged in a program for the determination of carcinogenic potential of tobacco cigarette smoke condensate of different characteristics by skin painting in mice. Standard experimental protocols have been worked out for this program. In view of the widespread use of marijuana it seemed timely to study the carcinogenic potential of marijuana smoke. In this paper we are describing the preparation of standard marijuana cigarettes-85 mm long and 25 mm in circumference using medium porosity paper, with no filter, production of the smoke condensate samples from these and the analysis for the selected parameters.
A total of 5,800 lbs. of marijuana was furnished by the National Cancer Institute. Most of the material was seized in drug raids made by federal authorities. It was packed in small boxes in the form of foot long bricks. It was necessary to break up the bricks, which consisted of long stems, twigs, seeds, along with some powdery material. The material in its present form was unsuitable for making the cigarettes, since the stems would clog up the feeding system of the cigarette maker. Several approaches were considered in order to prepare the material of proper consistency.
First, a few stems and twigs were soaked in water for 72 hours to soften them prior to putting through the Thropp mill. Water had very little effect on this material. A waxy coating on the surface of the stems prevented penetration of water. Consequently, it was decided not to pass the material through the Thropp mill in order to flatten the stems and twigs prior to shredding.
In order to separate the fine material from the coarse one containing stems, twigs, seeds, etc., the marijuana was sifted manually using sifters fitted with onehalf inch wire mesh screen. The material was thus separated into two piles. In the preliminary experiments, a small pile of coarse marijuana was sprayed with steam and was divided into two parts. Part one was put through the Himoff shredder and part two was put through Fitz mill fitted with one-half inch screen. The product obtained from the Fitz mill resembled finely shredded tobacco. The coarse pile of marijuana was, therefore, passed through the Fitz mill. The resulting product was sifted manually to separate the fine material from the coarse one, which was once again put through the Fitz mill and the product collected in one pile. The three piles of finely shredded marijuana were then combined in order to obtain a uniform blend of marijuana for making the cigarettes.
In the preliminary run, about five hundred pounds of blended marijuana was sprayed with steam. The pile was covered with canvass and polyethylene sheets. The material was left overnight. Next morning, the moisture content of the material was determined. It had 16.5% moisture. This material was put through the cigarette maker in order to see how it would run and examine the quality of cigarettes produced. This run indicated that good quality of marijuana cigarettes can be made if the blended pile had about 16% moisture. About 1.29 million cigarettes were then made, and packed in large cardboard trays, which were stored at 0 °F or below until required for smoking. Throughout the production cycle, the cigarettes were sampled frequently in order to obtain sufficient number of cigarettes of uniform quality for analytical work. The average weight of the marijuana cigarette was 1.1 g. This represented approximately 50% yield of the product. Prior to smoking the cigarettes were equilibrated for moisture content in a controlled atmosphere at 75 °F and 60% relative humidity for 48 hours.
The marijuana cigarettes were smoked at periodic intervals on a large automatic cigarette smoking machine built for the National Cancer Institute by the Process and Instruments Corporation, New York. The smoking machine (fig. 1) was designed to automatically load, light, smoke and eject approximately 2,000 cigarettes per hour and take a maximum of 10 puffs per cigarette at the rate of one puff per minute. The machine consisted of a hopper (manually loaded) from which the cigarettes automatically feed to position for continuous automatic loading into appropriate holders spaced radially on the circumference of a drum rotating in a horizontal plane. The drum accommodated four banks of 90 cigarettes spaced one inch apart. The banks were spaced vertically four inches apart. On the inside of the drum (fig. 2) there were 10 stationary puffing stations arranged radially so as to puff on the cigarettes as they passed each station. A radiant lighter (projection lamp) lighted each cigarette during the first puff, and after the tenth puff each smoked cigarette was automatically ejected from the machine. A vacuum pump with. an absolute air filter between it and the traps provided the suction for puffing on the cigarettes. The smoking cycle for this study consisted of ten 35 ml puffs (including lighting) of two seconds' duration each with 58 seconds' pause between puffs.
The smoke condensate trapping system (fig. 3) was piped in downstream of the smoking machine. It consisted of four three-litre Pyrex reactions flasks with Teflon covers, glass and Teflon interconnecting piping and a leak-tight stainless stell tank with metal support frame for flasks. The assembled traps were housed in a refrigerated cabinet capable of sustained operation down to -30 °F. The traps were cooled down further to about -90 °F by immersion in a slurry of dry ice and isopropanol. The first two flasks were partially packed with Pyrex beads of 4 mm diameter. The last two flasks were loosely packed with Teflon filament. This arrangement was found highly satisfactory for the large scale collection of the smoke condensate samples.


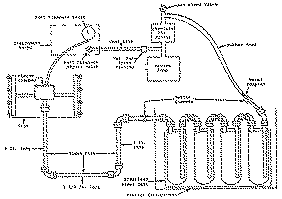
The smoke condensate from the trapping system was extracted with ACS certified reagent grade acetone, which had been freshly distilled through a vigreaux column. The acetone extract was concentrated under reduced pressure in a rotary evaporator at a temperature below 40 °C to yield a smoke condensate sample with less than 5% water content. The residue was mixed with a weighed amount of acetone and the water and the tetrahydrocannabinol contents of the mixture were determined by the gas chromatographic procedures. The smoke condensate data are given in tables 1 and 2.
The water in the smoke condensate was analysed by the vapor phase chromatographic procedure essentially similar to the one published by Schultz and Spears [ 5] . A Beckman GC 72-5 gas chromatograph with a thermal conductivity detector was used. A 3 ft by ½ inch O. D. copper column was packed with 120-150 mesh Porapak P. An injection port temperature of 220 °C, detector temperature of 250 °C and a column oven temperature of 168 °C (isothermal) were used. The helium flow rate was 40 ml/min, and the chart speed was one inch per minute. The injection volume for all samples was kept at 2 µl. The retention time for water was 0.4 min.
TABLE 1
Yield of marijuana cigarette smoke condensate under production conditions
|
Dry condensate yield |
|||||
|---|---|---|---|---|---|
|
Marijuana cig. lot No. |
No. cig. smoked |
No. puffs |
Butt length (mm) |
Total (g) |
mg/cig |
| 1 | 36 000 | 10 | 29.77 | 422 | 11.72 |
| 2 | 36 000 | 10 | 28.43 | 357 | 9.92 |
| 3 | 52 000 | 10 | 29.63 | 488 | 9.38 |
| 4 | 48 000 | 10 | 28.53 | 408 | 8.50 |
| 5 | 48 000 | 10 | 29.37 | 426 | 8.87 |
| 6 | 48 000 | 10 | 28.90 | 402 | 8.37 |
| 7 | 40 000 | 10 | 28.09 | 391 | 9.77 |
| 8 | 48 000 | 10 | 28.48 | 406 | 8.46 |
|
Mean
|
28.90 | 9.37 | |||
|
Standard deviation
|
0.58 | 1.05 | |||
The cannabinoids in the smoke condensate were analysed by the method of Lerner and Zeffert (6). A Beckman GC 65 gas chromatograph with a flame ionization detector was used. A 6 ft. be 2mm I.D. glass column was packed with 3% OV-17 on 100-200 mesh Gas-Chrom Q. An injection port temperature of 250°C, column oven temperature of 235°C and the detector temperature of 300 °C were used. The flow rates were: helium, 20ml/min; hydrogen ,45 ml/min; air, 250ml/min. The injection volume for all samples was kept at 1 μl. The relation times for cannabidiol, Δ 9tetrahydrocannabinol and cannabinol were 5.2, 7.3 and 9.3 minutes respectively.
TABLE 2
Analysis of marijuana smoke condensate samples
(Percentage)
|
Lot No. |
Δ9-tetrahydrocannabinol |
Cannabidiol |
cannabinol |
|---|---|---|---|
| 1 | 3.69 | 2.11 | 1.89 |
| 2 | 3.33 | 2.03 | 1.87 |
| 3 | 3.54 | 2.11 | 1.75 |
| 4 | 3.58 | 1.86 | 1.79 |
| 5 | 3.51 | 2.02 | 1.96 |
| 6 | 3.81 | 1.78 | 1.97 |
| 7 | 3.75 | 1.87 | 1.77 |
| 8 | 3.79 | 1.80 | 1.96 |
|
Mean
|
3.63 | 1.95 | 1.87 |
|
Standard deviation
|
0.15 | 0.13 | 0.08 |
The cigarettes to be smoked were first conditioned at 24 ± 1 °C and 60 ± 5% relative humidity for 48 hours. Several lots of 100 cigarettes each were selected at random. Using a Sartorium Model 2255 top-loading balance, each cigarette was weighed individually in order to determine the mean weight. Using a Filtrona pressure-drop tester model Mark 4LP, the pressure drop of each cigarette was also determined in order to obtain the mean pressure drop. The instrument operated by blowing air through the cigarette under test and then measuring the up-stream pressure relative to atmosphere when the flow rate was 17.5 ml per second.
The static burning rate of 100 cigarettes, temperature and humidity conditioned, and selected at random was determined in an essentially draft free enclosure. The time it took for the burning edge of the paper to move between two spaced pencil marks on the wrapper paper provided a measure of the free burning rate.
The combustion-zone temperature of cigarettes was determined by the method of Touey and Mumpower [ 7] using platinum-platinum/rhodium (87%-13%) thermocouple wires of 0.002 inch diameter. The temperature measurements were made by placing the thermojunction in the cigarettes at 15 and 55 mm from the lighted end along the axis. The temperature measurements were made on 10 cigarettes after conditioning them at 24 ± 1 °C and 60 ± 5% relative humidity.
The calcium, magnesium, potassium, sodium, cadmium and chromium in marijuana were determined by the atomic absorption spectroscopic method [ 8] . The nitrate content was determined by the procedure furnished by Dr. Markunas [ 9] . The nitrate was eluted from a column containing decolorizing mixture of celite, charcoal and florisil. The colorless extract was treated with sulfuric acid and brucine sulfate. The mixture was heated at 65 °C for 15 minutes, cooled, and the yellow color read at 400 mu. The reducing sugars were determined according to the classical method of Somogyi [ 10] . The hexane solubles in the ground marijuana were determined by a modification of the method of Bacot [ 11] .
Ground marijuana was weighed in triplicate samples of about two grams each in tared procelain dishes of 30 ml capacity. The samples were first heated in a muffle furnace at 230°C for 15 minutes and then at 650°C for 2 hours. The samples were cooled to room temperature in a dessicator and weighed. The increase in weight of the dishes was calculated as percentage total ash. About 2.5 g of ground marijuana was suspended in 50 ml of carbon dioxide-free distilled water. The suspension was stirred magnetically in nitrogen atmosphere for 1/2 hour and the pH of the aqueous extract was read on the pH meter.
The organic acids-citric, malic and oxalic acid-in marijuana were determined by a modification of the procedure of Palmer [ 12] . A Beckman GC 65 gas chromatograph with a flame ionization detector was used. A 4 ft. by 1/4 in. O. D. copper column was packed with 2.5% Carbowax 20M-TPA on 100-120 mesh, H. P. chromosorb G (AW-DMCS). An injection port temperature of 225 °C and the detector temperature of 275 °C were used. The column oven was maintained at 135 °C for one minute and then programmed from 135 °C to 210 °C at the rate of 25° per minute. The flow rates were: helium, 50 ml/min; hydrogen, 45 ml/ min; air, 250 ml/min. The injection volume for all samples was kept at 2 μl. The cannabinoids in the unsmoked marijuana were determined by the method of Lerner and Zeffert [ 6] . The analytical data are given in table 3.
TABLE 3
Determination of various analytical parameters of unsmoked blended marijuana and marijuana cigarettes
|
Unsmoked blended marijuana |
Value ± std. dev. |
Marijuana cigarette |
|---|---|---|
|
Ash
|
17.35 ± 005%
|
Average weight 1.114 g
|
|
Hexane solubles
|
7.82 ± 0.06%
|
Average pressure drop 14.75 cm. H20
|
|
Nitrate
|
0.31 ± 0.019%
|
Average moisture content 10.69
|
|
Reducing sugars
|
1.26 ± 0.039%
|
Static burning rate 3.96 mm/min.
|
|
Citric acid
|
1.06 ± 0.07
|
Fire zone temperature:
|
|
Malic acid
|
1.17 ± 0.02%
|
Thermo junction in center at:
|
|
Oxalic acid
|
1.88 ± 0.05%
|
15 mm mark 813 °C
|
|
Potassium
|
3.15 ± 0.02%
|
55 mm mark 830 °C
|
|
Sodium
|
0.04%
|
|
|
Calcium
|
2.86 ± 0.05%
|
|
|
Magnesium
|
0.51 ± 0.01%
|
|
|
Cadmium
|
0.43 ± 0.02 μg/g
|
|
|
Chromium
|
3.63 ± 0.04 μg/g
|
|
|
Δ9-tetrahydrocannabino
|
l.0.37 ± 0.02%
|
|
|
Cannabidiol
|
0.15 ± 0.01%
|
|
|
Cannabinol
|
0.32 ± 0.01%
|
|
|
pH
|
5.43 |
The objective of this study was the production of smoke condensate from the marijuana cigarettes on schedule for the bioassay study and involved quality control on each batch of smoke condensate. Examination of the condensate production data (tables 1 and 2) reveal that it is not possible to strictly control the butt length of cigarettes when smoked on a robot smoking machine thereby accounting for some variation in the yield of the dry particulate matter and A9-tetrahydrocannabinol. No A8-tetrahydrocannabinol was detected in any batch of the smoke condensate. Recently Hoffmann and co-workers [ 13] have reported that marijuana tar exhibited carcinogenic potential similar to tobacco cigarette smoke condensate in the dermal assay in mice.
We would like to thank Dr. M. Braude of the National Institute of Mental Health for furnishing pure cannabinoid standards. The work was conducted under U.S. Public Health Service Contract No. NIH 69-2084.
Anonymous, The Question of Cannabis, Bibliography U.N. Economic and Social Council, Committee on Narcotic Drugs Document E/CN.7/479/ 1965; Anonymous, Bulletin on Narcotics, III: I, p. 59, III: 2, p. 42 (1951).
002Neumeyer, J. L. and R. A. Shagoury, J. Pharm. Sci., 60, 1433 (1971).
003Marijuana: Chemistry, Pharmacology, Metabolism and Clinical Effects. R. Mechou-lam (ed.) Academic Press, New York (1973).
004Hollister, L. E., Science, 172, 21 (1971).
005Schultz, F. J. and A. W. Spears, Tobacco Science , 10, 75 (1966).
006Lerner, M. and J. T. Zeffert, Bulletin on Narcotics , XX: 2, p. 53 (1968).
007Touey, G. P. and R. C. Mumpower, II, Tobacco Science , 1, 33 (1957).
008Perkin-Elmer Corp., "Perkin-Elmer Analytical Methods for Atomic Absorption Spectrophotometry" (1971).
009Markunas, P. C., R. J. Reynolds Tobacco Co., Winston-Salem, North Carolina. Private communication.
010Somogyi, M., J. Biol. Chem ., 160, 61 (1965).
011Bacot, A.M., U.S. Dept. of Agr., Technical Bulletin No. 1225, 104 (1960).
012Palmer, A.M., Tobacco Science , 16, 83 (1972).
013Hoffmann, D., K. D. Brunnemann, E. Wynder and G. B. Gori. Paper given at the 14th annual symposium and meeting of the Phytochemical Society of North America held at Western Carolina University, Cullowhee, North Carolina, Aug. 7-9, 1974.