The Problem of Morphine Synthesis
Author: David Ginsburg
Pages: 15 to 17
Creation Date: 1956/01/01
The techniques used by the organic chemist to elucidate the constitution of a given molecule of simple or complex structure are, in principle, the same. After the compound under investigation is isolated in pure form and its empirical constitution is established by elementary analysis, the organic chemist applies degradative chemical reactions in which he chops the molecule into simpler fragments whose constitution it is easier to determine. The degradative technique allows one to reason from the structures established for the simpler fragments and from prior chemical knowledge, and thus deduce which structure would seem reasonable for the original, more complex molecule.
It is clear that the new tools available to the organic chemist, particularly those of ultraviolet and infrared spectroscopy, make the task - once so formidable - of determining the structure of a complex molecule simpler in the present generation. It could be said that infrared spectroscopy has supplied the average chemist today with an additional pair of eyes. True, it has not made a genius out of him, but it has revolutionized the technique of organic chemistry, and the average chemist today can accomplish more than his average counterpart in the previous generation. Further, he can carry more complex problems to their successful solution, not only because of the availability of these physical tools, but because of the obvious progress in his field due to his predecessors of the previous generations.
The organic chemist is not satisfied with the assignment of a structure to a molecule, no matter how logical his deductions may seem on the basis of the degradative evidence. Unsuspected rearrangements may have occurred during degradative reactions. Fine stereochemical points may still be obscure, and fully convincing deductions are sometimes impossible until synthesis of the molecule has been accomplished through a series of known reactions from compounds of unequivocal structure. Such an approach should appeal to the pure logician. It is unfortunate, however, that the organic chemist cannot always fulfil the rigid demands of his self-imposed logic during his lifetime. With certain complex structures several life-spans may elapse between isolation and final synthesis. In some cases, new and unprecedented reactions may creep into the complex synthesis, thus somewhat tarnishing the purity of the required logical discipline.
The morphine story is a good case in point and can be used as a case-history to illustrate the above statements.
Morphine was first isolated by Sertürner in 1805. In 1925, Gulland & Robinson summarized the results of a century of degradative work and proposed the correct formulation of the alkaloid. The circle was completed in 1952, when Gates and his co-workers succeeded in synthesizing this complex molecule. On the heels of this first synthesis of morphine, a second synthesis was published in 1954.
Since the syntheses of morphine by both the Gates group and the Ginsburg group include steps which were at least partly unexpected, the rigid logician might be doubtful as to whe- ther the stamp of final approval should be placed on the closure of the morphine story. However, the organic chemist who is aware of the trials and tribulations involved in the degradation and synthesis of complex natural products, is usually more charitable than the rigid logician and accepts morphine into the ranks of substances whose structure is unequivocally established.
Now, when we inspect the Robinson formula for morphine, we are prejudiced through our human frailty, by our previous fields of interest. We look at the spatial configuration of the morphine molecule from different vantage points and from different angles.
The chemist who is primarily interested in steroids sees morphine as a steroid which has somehow gone astray. Three of the four rings present in the steroid molecule are present in morphine, but ring D has disappeared and has been replaced by a piperidine ring fused to the carbocyclic system in an unorthodox way. Yet the reactions of certain morphine derivatives are analogous to those of steroids (cf. bromination of coprostanone and dihydrothebainone ([1] )).
The chemist working in the field of polycyclic aromatic compounds will certainly note the reduced phenanthrene system present in morphine. The furan chemist will observe that morphine is a furan or a reduced dibenzofuran derivative. The chemist interested in piperidine derivatives will not fail to note the N-methyl piperidine portion of the morphine molecule. Indeed, it was only when this viewpoint was taken that the synthetic compounds based on the morphine structure began to have analgesic activity. The chemist interested in isoquinolines will observe that morphine can be viewed as an isoquinoline derivative. When biogeneric arguments are brought into play, one can see that a substituted 1-benzylisoquinoline portion is present in the morphine molecule.
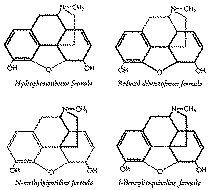
One cannot under-estimate the importance of the various viewpoints, angles of observation and vantage points. It cannot be a coincidence that the Gates synthesis of morphine involves a napthaquinone starting material when one knows of the earlier association between Dr. Gates and Professor Fieser. Grewe has published an excellent article ([2] ) on the biogenetic considerations (first pointed out by Robinson ([3] )) which led him to the synthesis of N-methylmorphinan. The latter synthesis has been described in a beautifully written series of papers ([4] ) which should be made required reading for all graduate students of organic chemistry. The interest of Koelsch in the Michael condensation led him to apply this reaction to the synthesis of morphine, thus far culminating in the synthesis of N-methylmorphinan ([5] ). Surely it is no original discovery that persons of varied chemical interests apply their expert knowledge in specific fields and attempt to extend it to other fields.
It is not necessary to give a full review of the many varied approaches to the morphine structure, as this field has been reviewed recently ([6] ). Certain developments in the field have nevertheless rendered some aspects of the recent review obsolete. Therefore, several of the more successful approaches will be discussed here in a general way. The details of each synthetic route may be obtained by consulting the original publications.
The Gates synthesis ([7] ) may be termed the naphthalene approach. Rings A and B of morphine are performed in a suitably chosen naphthalene derivative which bears in the 4-position the embryo of the piperidine ring. Ring C is at-

tached through a Diels-Alder reaction. The piperidine ring forms "of its own accord". The latter phrase is placed in quotation marks because most organic chemists will accept the adage that "God helps those who help themselves".
Professor Woodward made a statement at the recent Gordon Conference on Steroids and Related Natural Products ([8] ) which I am taking the liberty of paraphrasing : "lucky" cyclizations take place when groupings are so placed by the synthetic chemist that Lady Luck is in a favourable position to exert her influence. It is clear that it was not by chance that Gates placed a cyanomethyl group at the 4-position of his naphthaquinone starting material. Once the tetracyclic ring system present in morphine had been prepared, the synthesis of morphine was, in principle, certain to succeed. I do not wish to under-estimate the subsequent steps of the Gates synthesis, the execution of which was brilliant. The major factor, however, is that a double bond was present in ring C, designed to be present as the key or the wedge for insertion of other groups necessary to convert the first tetracyclic intermediate of trans- stereochemisty to the final goal - morphine. Knowledge of various reactions and electronic mechanistic principles certainly helped in the final stages, but the point I wish to make is that the beauty of the Gates synthesis is in starting from a very well chosen set of materials which already has built in the features for further synthetic elaboration - the nitrogen atom for the piperidine ring and the double bond resulting from the diene reaction in a position to serve as key for the introduction of further necessary features into the molecule.
The Grewe synthesis of N-methylmorphinan and tetrahydrodesoxycodeine ([4] ) follows the presumed biogenetic route used by the opium poppy. The starting materials are compounds of which analogues are present in the plant, and although the latter uses enzyme systems to accomplish the key addition of a hydrogen atom of the benzene ring to the double bond of the octahydroisoquinoline intermediate, it is necessary to use much more stringent conditions in the laboratory (long heating with concentrated phosphoric acid).

The difficulty in the Grewe approach lies in preparing suitable intermediates for preparing morphine itself. A key to ring C is lacking. Despite the fact that Grewe and his associates did not synthesize morphine itself, they were the first to construct the tetracyclic skeletal system present in morphine and to demonstrate the identity of their synthetic tetrahydrodesoxycodeine with that of the product derived from natural sources by various degradative reactions.
The Ginsburg approach to morphine may be termed the phenylcyclohexane approach. In contrast to the Gates method which constructs the carbocyclic rings of morphine from AB+C, this approach starts with AC and adds on B.
The nitrogen atom required for the piperidine ring is either present in the grouping which enters the AC system through Michael addition, or is introduced subsequently into the ABC system by the use of a carbonyl group acting as the key to
ring B. Here also the nitrogen containing ring is cyclized through an unprecedented reaction, made possible, however, by the presence of a second carbonyl group whose function it is to serve as a key to ring C. The synthesis culminates with dihydrothebainone since this ketone was previously converted to morphine by Gates and his associates. Again, the success of this approach can be traced to the presence

of activating groups at points required for further synthetic elaboration. A somewhat similar approach by Horning and associates ([9] ) failed because such keys were lacking. The same may be said for the Fieser-Holmes AB + C approach ([10] ) preceding the Gates work, in which groups introduced as potential keys failed to turn the lock.
In their present stage of development, neither of the two published syntheses of morphine is practical from the commercial point of view. The Grewe approach has been modified and various synthetic analgesics are being manufactured and marketed, e.g., 3-hydroxy-N-methylmorphinan. The major importance of work done on the synthesis of a complex molecule does not lie merely in the successful achievement of the synthesis. The fun involved in the work should not be under-estimated; nor should the satisfaction in successful completion of a long and difficult road. Surely the major importance lies in the development and application of synthetic methods and in the cross-fertilization of different fields of research in organic chemistry. In general, the more complex the molecule under attack, the broader the interest and the more numerous are the theoretically possible approaches and vantage points. The true value of a synthesis of a complex molecule is not merely in the completion of the circle of organic chemical logic which requires the structure deduced by degradation to be proved by synthesis, but in the application to other fields of the progress made through that synthesis.
GINSBURG, U. N. Bulletin of Narcotics, 1954, VI : 1, p. 32.
002GREWE, Naturwiss., 1946, 33, 333.
003ROBINSON, Proceedings Univ. Durham Phil. Soc., 1927-1928, 8, part 1, 14.
004GREWE, et al., Ber., 1948, 81, 279; Ibid., 1951, 84, 527; Ann. 1949, 564, 161.
005KOELSCH & ALBERTSON , J.A.C.S., 1953 , 75, 2095.
006STERN, Quart. Rev., 1951, 5, 405.
007GATES, et al., J.A.C.S., 1948, 70, 2261; Ibid., 1950, 72: 1141, 4839; Experientia, 1949, 5, 285; J.A.C.S., 1952, 74, 1109.
008New Hampton, New Hampshire, 6 August 1954.
009HORNING, et al., J.A.C.S ., 1948, 70, 2072, 2941, 2945; Ibid., 1949, 71, 1359.
010FIESER & HOLMES, J.A.C.S., 1936, 58, 2319; Ibid., 1938, 60, 2548.